SESAMI Trial Sirolimus Stent vs Bare Stent in

SESAMI Trial Sirolimus Stent vs. Bare Stent in Acute Myocardial Infarction Trial Presented at The Euro. PCR meeting Paris, France May 2006 Presented by Dr. Maurizio Menichelli

SESAMI Trial: Background • The goal of the trial was to evaluate primary/rescue percutaneous coronary intervention (PCI) using a sirolimus-eluting stent compared with a bare metal stent among patients with acute myocardial infarction. www. Clinical trial results. org Presented at Euro. PCR May 2006
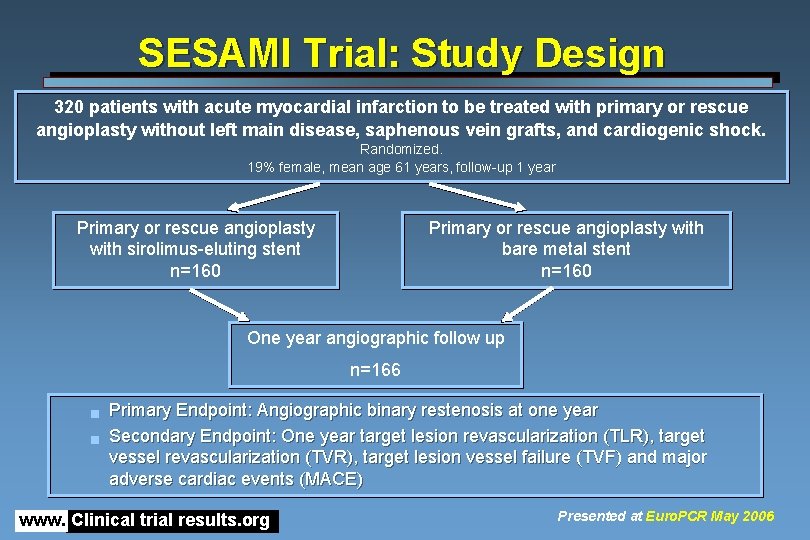
SESAMI Trial: Study Design 320 patients with acute myocardial infarction to be treated with primary or rescue angioplasty without left main disease, saphenous vein grafts, and cardiogenic shock. Randomized. 19% female, mean age 61 years, follow-up 1 year Primary or rescue angioplasty with sirolimus-eluting stent n=160 Primary or rescue angioplasty with bare metal stent n=160 One year angiographic follow up n=166 g g Primary Endpoint: Angiographic binary restenosis at one year Secondary Endpoint: One year target lesion revascularization (TLR), target vessel revascularization (TVR), target lesion vessel failure (TVF) and major adverse cardiac events (MACE) www. Clinical trial results. org Presented at Euro. PCR May 2006

SESAMI Trial: Baseline Characteristics • Baseline characteristics were balanced between groups, with primary PCI performed in 82% of patients and rescue PCI in 18% of patients. • In approximately half of the patients, the infarct– related artery was the left anterior descending artery. • In the majority of patients, Abciximab was used in either the emergency room or the cath lab. • Post-procedural TIMI grade 3 flow was present in 95% of patients. www. Clinical trial results. org Presented at Euro. PCR May 2006

SESAMI Trial: Primary Endpoint Incidence One year binary restenosis p<0. 05 www. Clinical trial results. org • The primary endpoint of one year binary restenosis on angiography occurred less often in the sirolimus-eluting stent group vs. the bare metal stent group (9. 3% vs. 21. 3%, relative risk reduction [RRR] 56%, p<0. 05). • Likewise, clinically driven restenosis was also lower in the sirolimus-eluting stent group (5. 6% vs. 17. 2%, RRR 64%, p<0. 05). Presented at Euro. PCR May 2006
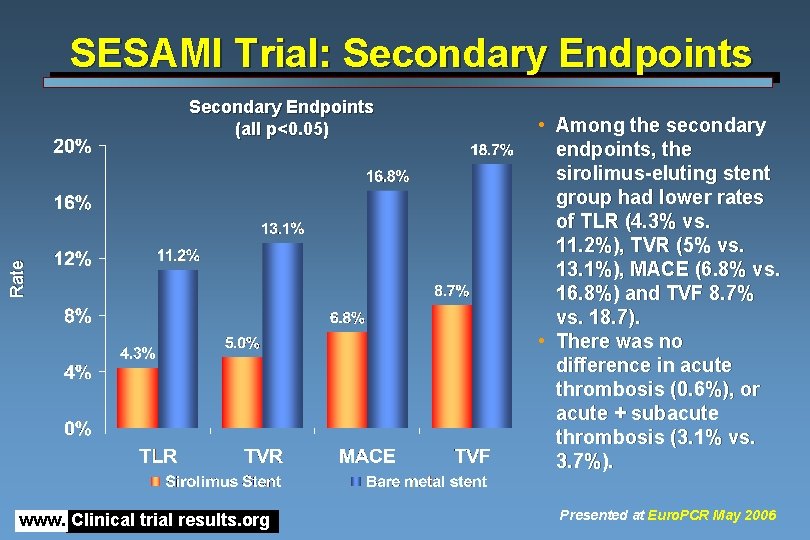
SESAMI Trial: Secondary Endpoints Rate Secondary Endpoints (all p<0. 05) www. Clinical trial results. org • Among the secondary endpoints, the sirolimus-eluting stent group had lower rates of TLR (4. 3% vs. 11. 2%), TVR (5% vs. 13. 1%), MACE (6. 8% vs. 16. 8%) and TVF 8. 7% vs. 18. 7). • There was no difference in acute thrombosis (0. 6%), or acute + subacute thrombosis (3. 1% vs. 3. 7%). Presented at Euro. PCR May 2006

SESAMI Trial: Summary • Among patients with acute myocardial infarction undergoing primary or rescue PCI, use of sirolimus-eluting stent was associated with a reduction in binary restenosis at one year compared with use of a bare metal stent. • Results of the present single center trial are similar to those seen in the multi-center TYPHOON trial, which showed a reduction in target vessel failure at one year with sirolimus-eluting stents compared with bare metal stents in PCI for acute MI, driven primarily by a reduction in TLR. • The PASSION trial, however, did not demonstrate a reduction in TLR with paclitaxel-eluting stents over bare metal stents among STEMI patients. • A cost-effectiveness analysis from the present trial is underway. www. Clinical trial results. org Presented at Euro. PCR May 2006
- Slides: 7