Serostim Treatment of Short Bowel Syndrome NDA 21

Serostim ® Treatment of Short Bowel Syndrome NDA 21 -597 Division of Gastrointestinal and Coagulation Drug Products Hugo E. Gallo-Torres, M. D. , Ph. D, PNS Medical Team Leader

Outline • Introduction • Medical Literature • Study IMP 20317 • Outstanding Issues – – replicability generalizability validity of endpoints exploration of dosing Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 2

Proposed Indication Serostim® – Treatment of Short Bowel Syndrome (SBS) in patients receiving specialized nutritional support – In conjunction with optimal management of SBS Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 3

Short Bowel Syndrome Treatment • Nutritional management and replacement of fluid and electrolyte losses • IPN requirements vary depending on the presence or absence of: ileocecal valve, jejunum, functional colon and length of residual bowel • Patients with residual small bowel 100 cm or less frequently require chronic administration of IPN Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 4

Complications of Long-Term Parental Nutrition • • Cholelithiasis Catheter Sepsis Hepatic Dysfunction Nutrient Deficiencies Bone Demineralization Central Vein Thrombosis Glucose Metabolism Disorders Progressive Renal Insufficiency Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 5

Known Complications of Growth Hormone • • Edema Arthralgia Headache Hypothyroidism Antibody Formation Glucose Metabolism Disorders Possible Association with Leukemia Intracranial Hypertension with Papilledema Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 6
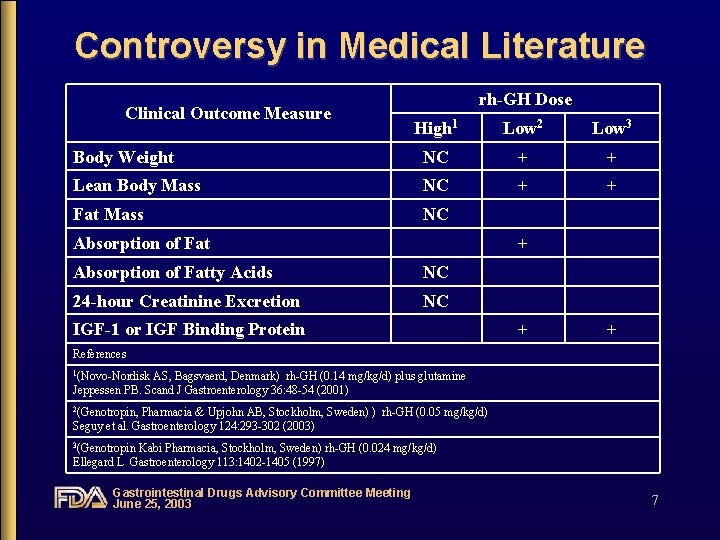
Controversy in Medical Literature Clinical Outcome Measure rh-GH Dose High 1 Low 2 Low 3 Body Weight NC + + Lean Body Mass NC + + Fat Mass NC Absorption of Fat + Absorption of Fatty Acids NC 24 -hour Creatinine Excretion NC IGF-1 or IGF Binding Protein + + References 1(Novo-Nordisk AS, Bagsvaerd, Denmark) rh-GH (0. 14 mg/kg/d) plus glutamine Jeppessen PB. Scand J Gastroenterology 36: 48 -54 (2001) 2(Genotropin, Pharmacia & Upjohn AB, Stockholm, Sweden) ) rh-GH (0. 05 mg/kg/d) Seguy et al. Gastroenterology 124: 293 -302 (2003) 3(Genotropin Kabi Pharmacia, Stockholm, Sweden) rh-GH (0. 024 mg/kg/d) Ellegard L Gastroenterology 113: 1402 -1405 (1997) Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 7

Study IMP 20317 Design • Evaluation of Recombinant Human Growth Hormone (rh-GH) and Glutamine, Singly and as Co-therapy, in the Improvement of Residual Gut Absorptive Function in Patients with Short Bowel Syndrome – – – phase III dose: (0. 1 mg/kg) subcutaneous daily randomized double-blind controlled parallel-group (3 -arm) Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 8

Treatment Arms • GROUP A: – rh-GH + (GLN Placebo) • GROUP B: – rh-GH + GLN • GROUP C: (control group) – (rh-GH placebo) + GLN Specialized Oral Diet, received by all patients: Components: fluid, oral cal. , protein, carbohydrates, fat Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 9

Primary Endpoint • Change in total IPN volume: – IPN volume plus – supplemental lipid emulsion (lipids) plus – intravenous hydration fluid • Total IPN volume requirements were captured on a daily basis Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 10

Secondary Endpoints • Mean change in total IPN calories – carbohydrate – protein – fat • Mean change in IPN or lipid frequency – number of days per week of IPN or – lipids if greater than 200 kcal – intravenous hydration Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 11

Sponsor’s Exploratory Analysis • Subjects who demonstrated a “complete response” at Week 6 – complete wean from IPN, lipids and wean from intravenous hydration – complete wean from IPN, and lipids intravenous hydration allowed • Results in these study populations summarized descriptively Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 12
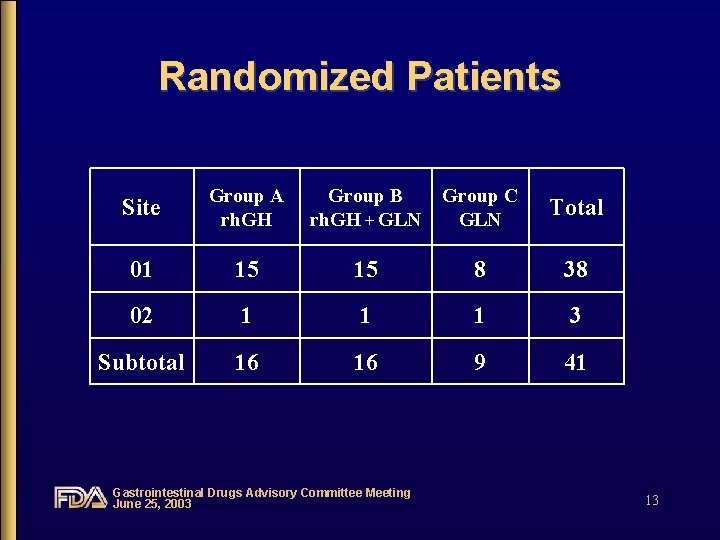
Randomized Patients Site Group A rh. GH Group B rh. GH + GLN Group C GLN Total 01 15 15 8 38 02 1 1 1 3 Subtotal 16 16 9 41 Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 13

Study Population • 41 randomized patients – age 20 to 75 years • < 65 years (n = 33) • >= 65 years (n = 8) – Caucasian (n = 32), non-Caucasian (n = 9) – females (n = 29), males (n = 12) • baseline characteristics were similar between treatment groups: i. e. length of residual bowel, IPN requirements, duration of therapy Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 14
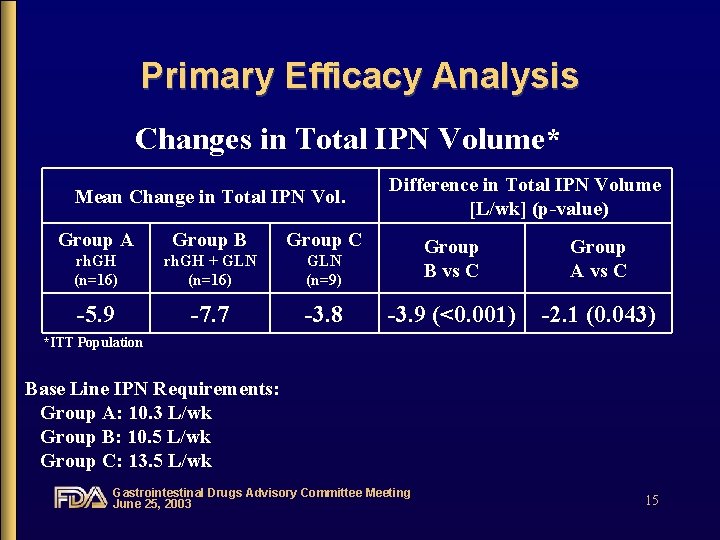
Primary Efficacy Analysis Changes in Total IPN Volume* Mean Change in Total IPN Vol. Difference in Total IPN Volume [L/wk] (p-value) Group A Group B Group C rh. GH (n=16) rh. GH + GLN (n=16) GLN (n=9) Group B vs C Group A vs C -5. 9 -7. 7 -3. 8 -3. 9 (<0. 001) -2. 1 (0. 043) *ITT Population Base Line IPN Requirements: Group A: 10. 3 L/wk Group B: 10. 5 L/wk Group C: 13. 5 L/wk Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 15
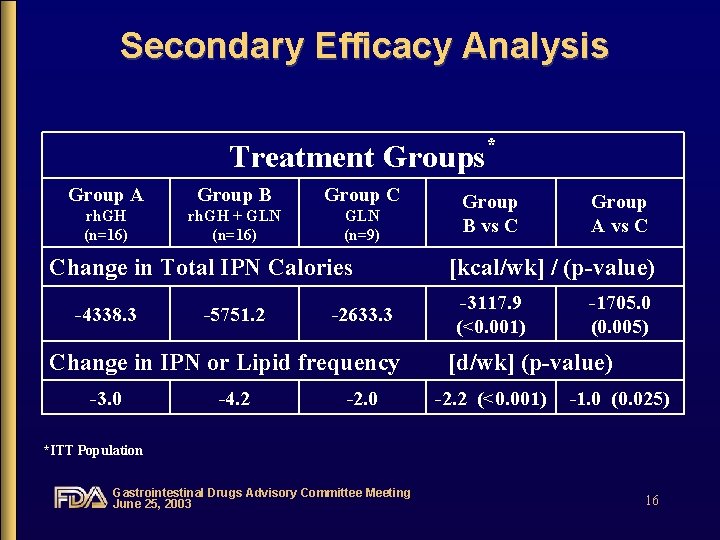
Secondary Efficacy Analysis Treatment Groups Group A Group B Group C rh. GH (n=16) rh. GH + GLN (n=16) GLN (n=9) Change in Total IPN Calories -4338. 3 -5751. 2 -2633. 3 Change in IPN or Lipid frequency -3. 0 -4. 2 -2. 0 * Group B vs C Group A vs C [kcal/wk] / (p-value) -3117. 9 (<0. 001) -1705. 0 (0. 005) [d/wk] (p-value) -2. 2 (<0. 001) -1. 0 (0. 025) *ITT Population Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 16

Effects of Covariates on Primary Endpoint • Total IPN volume was significantly influenced by: – patients' weight • the higher the body weight the greater the reductions in IPN volume – length of residual bowel • the longer the residual bowel the greater the reduction in IPN volume – IPN volume history • the higher the IPN volume requirements the greater the decrease in IPN volume during the treatment period – race • Caucasians responded to treatment better than non-Caucasians Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 17

Significance of Treatment Effect After Adjusting for Covariates • Pairwise Comparison of Group B to C – Maintained significant difference in total IPN volume after adjusting for covariates • Pairwise Comparison of Group A to C – Only reached a significant difference in total IPN volume when weight was used as a covariate Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 18

Effects of Covariates on Secondary Endpoints • Total IPN calories for the ITT population were not influenced by any of the covariates • Only weight influenced the treatment results for frequency of administration of IPN or lipids • Covariate analyses for the Evaluable for Efficacy population yielded similar results to the ITT population Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 19

Changes in Specialized Oral Diet • The greater the reduction in total IPN the greater the increase in oral diet – with exception of oral fluids, a larger increase in oral intake occurred in Groups A & B compared to Group C (control group) • As nutritional status improved subjects appetite increased Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 20

Exploratory Analysis Complete Responders Group A Group B Group C rh. GH (n=16) rh. GH + GLN (n=16) GLN (n=9) Complete Wean from IPN, Lipids and IV Hydration 4 4 Complete Wean from IPN and Lipids (IV Hydration Acceptable) 5 7 Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 1 1 21

Adverse Events • One or more AE • groups A&B: • control group: 100% 89% • Most Common AEs • tissue edema • fatigue • gastrointestinal disorders • No Deaths Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 22

Adverse Events • No SAE were considered drug related • Safety profile in SBS population similar to rates reported in Serostim® package insert • No clinically significant difference in laboratory values for the 3 treatment groups Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 23

Conclusions • Study IMP 20317: A single center, 41 patient study demonstrated subcutaneous rh-GH (0. 1 mg/kg) in cotherapy with GLN and SOD reduces the total IPN volume requirement in patients with SBS • Clinical relevance of the primary endpoint (reduction in Total IPN requirement per week) is uncertain Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 24

Outstanding Issues • Replicability – Essentially a one center, single study trial enrolling 41 -patients • Generalizability – Can one center be representative of the US SBS population? • Validity of Primary Endpoint of Efficacy – reduction in Total IPN Volume requirements – should primary endpoint be complete wean from IPN and lipid? – durability of response • Further exploration of Dosing – Is low-dose rh-GH more effective? Gastrointestinal Drugs Advisory Committee Meeting June 25, 2003 25
- Slides: 25