Serine proteases Named because they use a serine
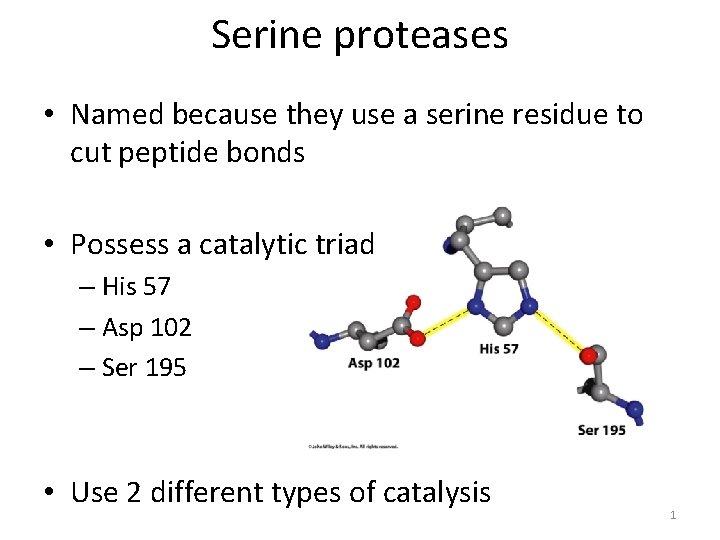
Serine proteases • Named because they use a serine residue to cut peptide bonds • Possess a catalytic triad – His 57 – Asp 102 – Ser 195 • Use 2 different types of catalysis 1
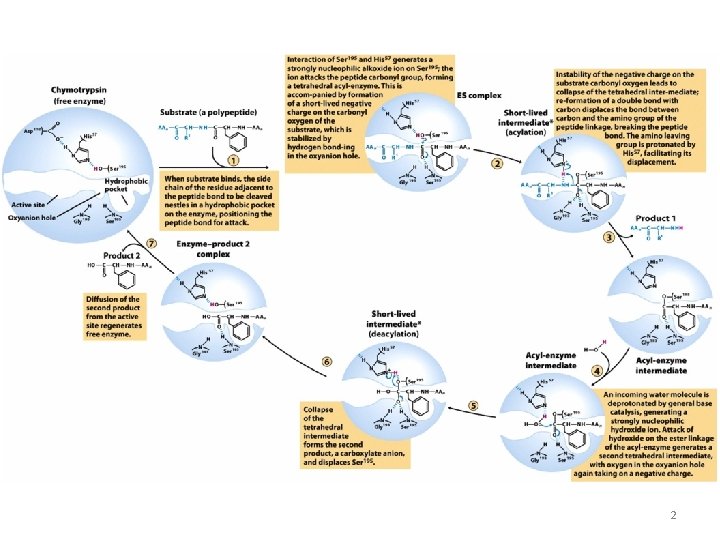
2
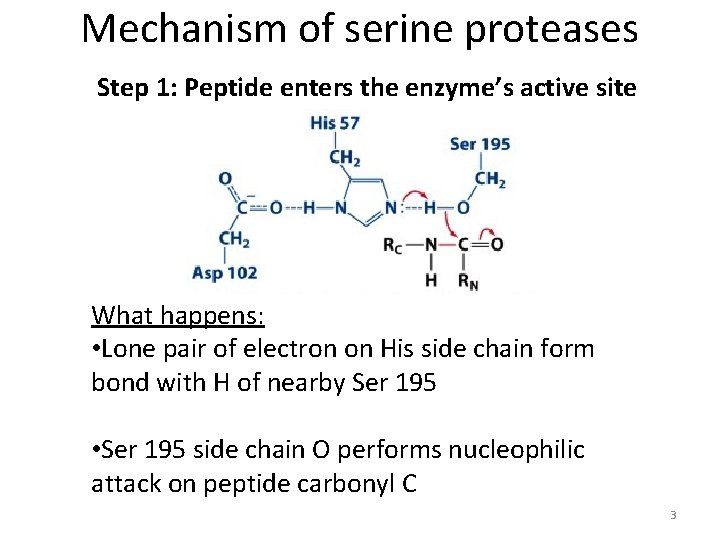
Mechanism of serine proteases Step 1: Peptide enters the enzyme’s active site What happens: • Lone pair of electron on His side chain form bond with H of nearby Ser 195 • Ser 195 side chain O performs nucleophilic attack on peptide carbonyl C 3
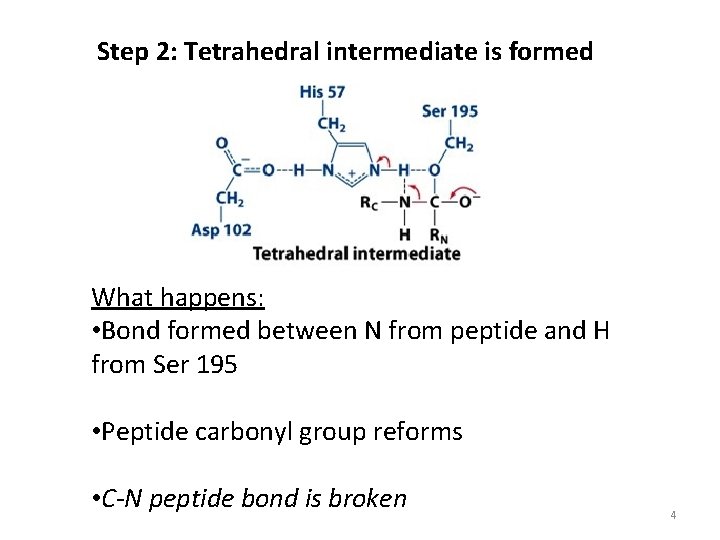
Step 2: Tetrahedral intermediate is formed What happens: • Bond formed between N from peptide and H from Ser 195 • Peptide carbonyl group reforms • C-N peptide bond is broken 4
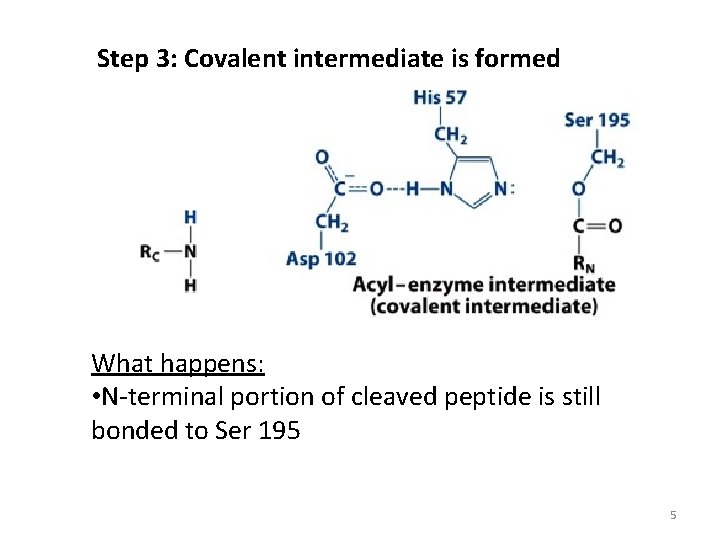
Step 3: Covalent intermediate is formed What happens: • N-terminal portion of cleaved peptide is still bonded to Ser 195 5
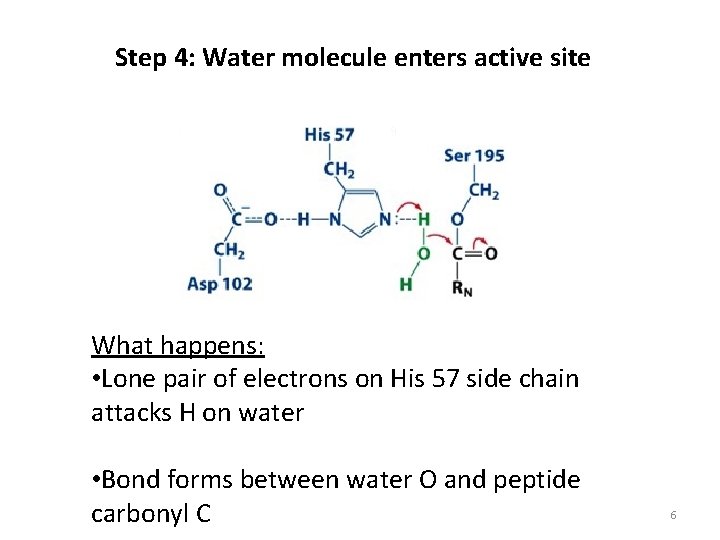
Step 4: Water molecule enters active site What happens: • Lone pair of electrons on His 57 side chain attacks H on water • Bond forms between water O and peptide carbonyl C 6
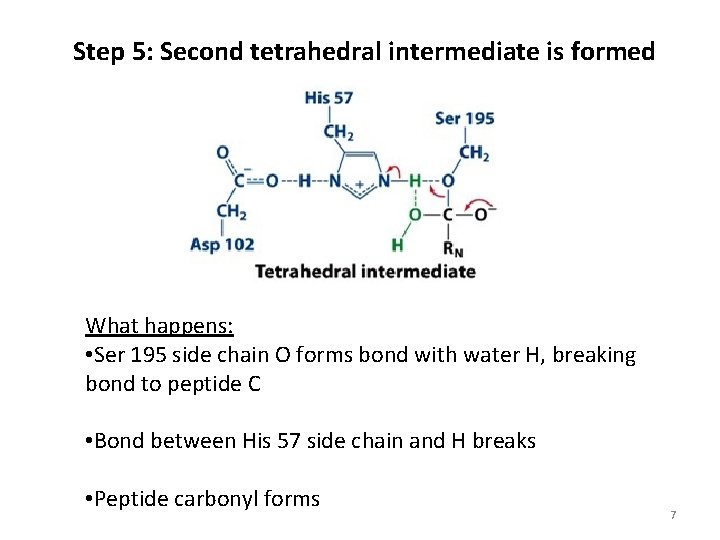
Step 5: Second tetrahedral intermediate is formed What happens: • Ser 195 side chain O forms bond with water H, breaking bond to peptide C • Bond between His 57 side chain and H breaks • Peptide carbonyl forms 7
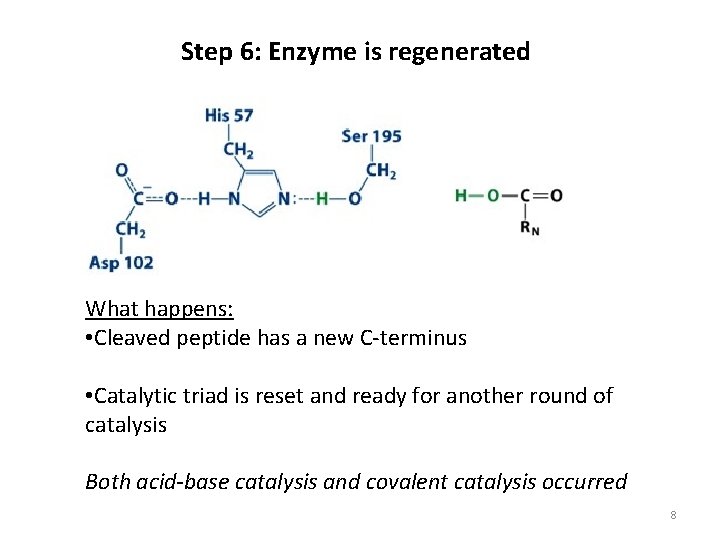
Step 6: Enzyme is regenerated What happens: • Cleaved peptide has a new C-terminus • Catalytic triad is reset and ready for another round of catalysis Both acid-base catalysis and covalent catalysis occurred 8
- Slides: 8