Serial Dilution By EID ALATAWI Introduction Many of

Serial Dilution By: EID ALATAWI

Introduction: � Many of the laboratory procedures involve the use of dilutions. � It is important to understand the concept of dilutions, since they are a handy tool used throughout all areas of the clinical laboratory. DEFINITIONS: �Solutions: homogeneous mixture of two or more substances physically mixed together in a uniform way. �Solute: substance being dissolved. �Solvent: part of a solution doing the dissolving. �Soluble: when a substance dissolves in another substance. �Aliquot: a measured sub-volume of original sample. �Diluent: material with which the sample is diluted.

�Dilution: - It is the process of reducing the concentration of a solute in solution, usually simply by mixing with more solvent. To dilute a solution means to add more solvent without the addition of more solute. � A serial dilution: - It is any dilution where the concentration decreases by the same quantity in each successive step. � Serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. � Serial dilutions are multiplicative, so each successive dilution would be multiplied by the dilution factor.

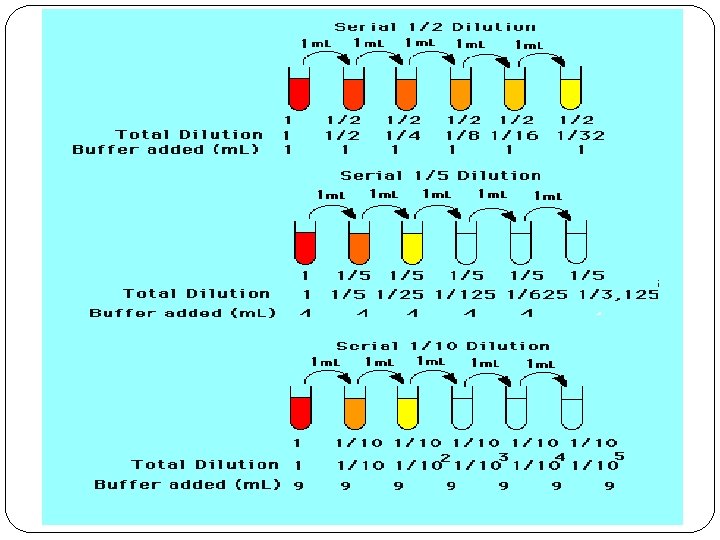
Doubling Dilutions �“Doubling dilutions” are very popular. � This is a series of ½ dilutions. Each successive tube will ½ the amount of the original concentrated solution. �If this is done 6 times this is what you would end up with:

Doubling Dilution 6 Times � 1 st dilution = 1 /2 � 2 nd dilution = 1 /2 x 1 /2 = 1/4 � 3 rd dilution = 1/4 x 1 /2 = 1/8 � 4 th dilution = 1/8 x 1 /2 = 1/16 � 5 th dilution = 1/16 x 1 /2 - 1/32 � 6 th dilution = 1/32 x 1 /2 = 1/64 �This results in a series of dilutions, each a doubling dilution of the previous one

v. Other dilutions: �The dilution can also be 1/3, 1/4, 1/5, …, 1/100, etc. �If a solution has a 1/10 dilution the number represents 1 part of the patient sample added to 9 parts of diluent. �This represents 1 part patient sample added to 9 parts of diluent.

Dilution Calculations: v To calculate a dilution factor: - Dilution factor (DF): ratio of final volume/aliquot volume (final volume = aliquot + diluent). - EXAMPLE: What is the dilution factor if you add 0. 1 m. L aliquot of a specimen to 9. 9 m. L of diluent? - Dilution factor (DF) = Final volume Aliquot volume - The final volume = the aliquot volume + the diluent volume: 0. 1 m. L + 9. 9 m. L = 10 m. L - DF = 10 ml = 1 : 100 dilution 0. 1 ml

üPractice �What is the dilution factor when 0. 2 m. L is added to 3. 8 m. L diluent?

§ Answer: �dilution factor = final volume/aliquot volume � 0. 2 +3. 8 = 4. 0 total volume � 4. 0/0. 2 = 1: 20 dilution

Remember that each successive dilution would be multiplied by the dilution factor. �In the previous dilution all successive tubes would have 3. 8 m. Ls of diluent. �You would then transfer 0. 2 of the initial diluted sample into the next tube, mix transfer 0. 2, mix and so on. �If you had 4 tubes what would be the final dilution of tube 4?
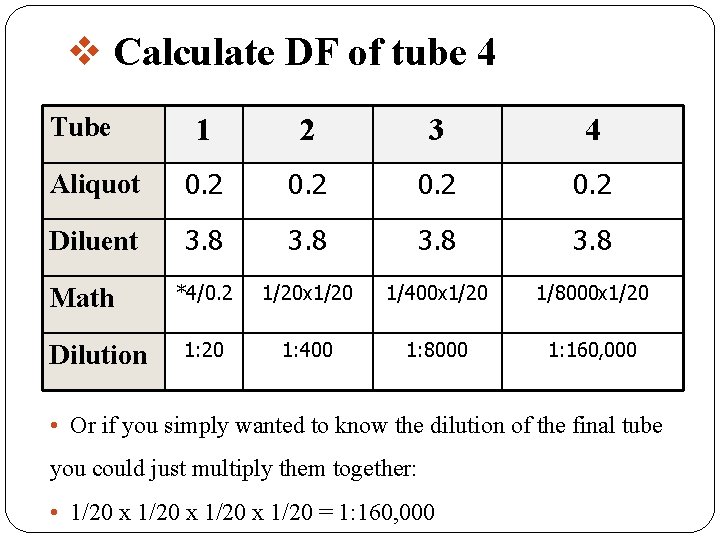
v Calculate DF of tube 4 Tube 1 2 3 4 Aliquot 0. 2 Diluent 3. 8 *4/0. 2 1/20 x 1/20 1/400 x 1/20 1/8000 x 1/20 1: 400 1: 8000 1: 160, 000 Math Dilution • Or if you simply wanted to know the dilution of the final tube you could just multiply them together: • 1/20 x 1/20 = 1: 160, 000

v To Calculate the Concentration of a Solution: In the initial and diluted solution, � the moles of solute are the same � the concentrations and volumes are related by the following equations: For percent concentration C 1 V 1 = Concentrated solution C 2 V 2 Diluted solution V 1 x C 1 = V 2 x C 2 � C 1 = Initial concentration. V 1 = Initial volume. C 2 = Final concentration. 13 V 2 = Final volume.

Guide to Calculating Dilution Quantities 14

Example of Dilution Calculations Using Percent Concentration % What volume of a 2. 00% (m/v) HCl solution can be prepared by diluting 25. 0 m. L of 14. 0% (m/v) HCl solution? STEP 1 Prepare a table: C 1 = 14. 0% (m/v) V 1 = 25. 0 m. L C 2 = 2. 00% (m/v) V 2 = ? STEP 2 Solve dilution expression for the unknown C 1 V 1 = C 2 V 2 = V 1 C 1 C 2 STEP 3 Set up the problem using known quantities: V 2 = V 1 C 1 = (25. 0 m. L)(14. 0%) = 175 m. L C 2 2. 00% 15

v Applications of dilution: Why we need to perform a dilution? � Serial dilutions are often used in microbiology, biotechnology, toxicology, immunology and in chemistry classes. � Serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. * � Sometimes when analyzing a blood sample the results are above the linearity (or accuracy) of the machine. � When this happens the blood must be diluted, retested, then the result multiplied by the dilution factor.
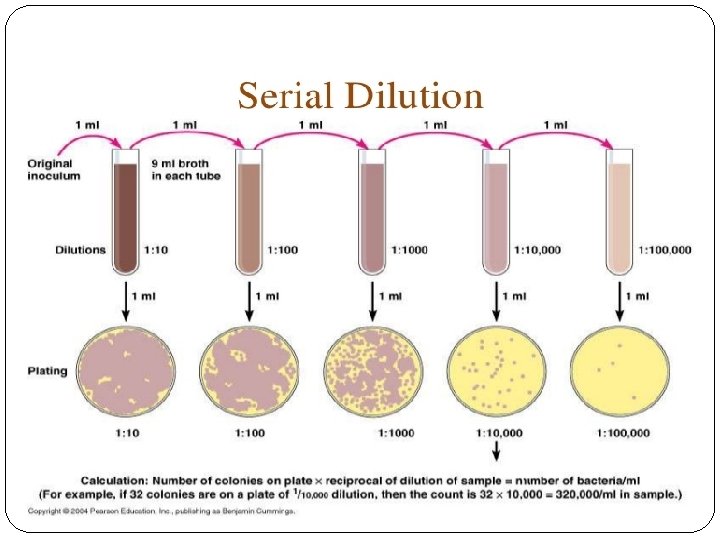

Example: �A blood glucose of 800 mg/d. L was obtained. �According to the manufacturer the highest glucose result which can be obtained on this particular instrument is 500 mg/d. L. �The sample must be diluted.

�The serum was diluted 1: 10 and retested. �The result is 80 mg/d. L. �THIS IS NOT THE REPORTALBE RESULT! �You must multiply by the dilution factor of 10. � 10 x 80 = 800 mg/d. L

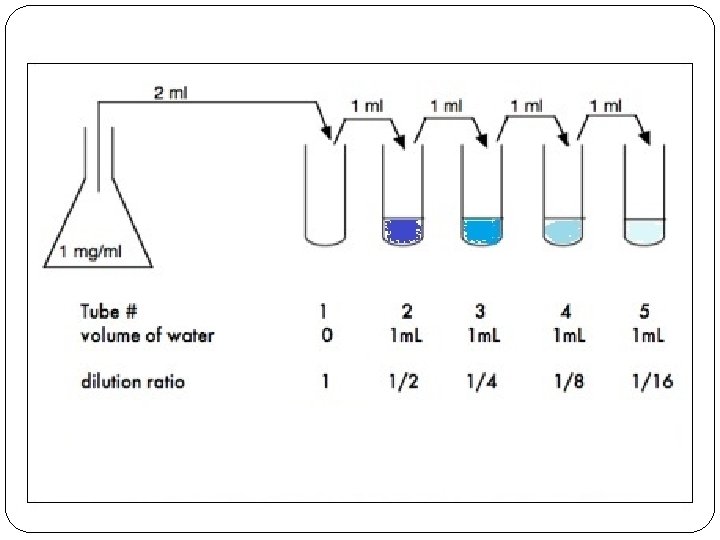
v Procedure: � Prepare all materials are needed for this experiment. � Label tubes for serial dilutions as follows: 1 (1: 2); 2 (1: 4); 3 (1: 8); 4 (1: 16); 5 (1: 32) � Using a micropipettor, pipet 1 m. L of distilled water into tubes 1, 2, 3, 4, 5. � Be sure cap is firmly closed and mix the dye solution by inverting the tube. Using a micropipettor, pipet 1 m. L of blue dye into tube 1. Mix gently by drawing the solution up and down 3 times (3 X). � Transfer 1 m. L of solution from tube 1 into tube 2. Mix gently 3 X. � Transfer 1 m. L of solution from tube 2 into tube 3. Mix gently 3 X. Continue to transfer and mix through tube 5. � Discard the last 1 m. L from tube 5. � Examine the tube dilutions. Note that the color decreases with increasing tube number.

�Time To Practice. . .
- Slides: 22