September 8 2014 Have FDA Expedited Programs Shortened

September 8, 2014 Have FDA Expedited Programs Shortened Drug Development Timelines? An analysis of newly approved therapies and how FDA expedited programs impacted drug development timelines Artisan Healthcare Consulting, Inc. +1 781 -221 -5656 phone info@artisan-consulting. com www. artisan-consulting. com

Methodology (1 of 2) 1. We collected key dates for new molecular entities (NME) and biologics approved over the past 6 years. ü We collected the following dates for each NME approved from 2009 to 2014: ü IND(investigational new drug) submission date of the first approved indication ü Date of the FDA end of phase 2 (EOP 2) meeting where available or the start date for the pivotal trials ü NDA (new drug application)/BLA (Biologics License Application) filing dates ü NDA/BLA approval dates ü We stratified the NMEs by year of approval, and type of FDA expedited program used. ü We categorized each NME by therapeutic area: Allergy, Autoimmune, Cardiovascular, Central Nervous System (CNS), Diabetes/ Metabolism, Diagnostic, Infectious disease, Musculoskeletal, Oncology, Ophthalmology, Rare diseases, Respiratory system, Sexual Dysfunction, Skin, Supportive care, and Women’s Health. 2. We looked at the 4 major FDA programs used to expedite clinical development to understand impact on time to market ü Fast Track is introduced to accelerate the approval for medications treating serious conditions with unmet medical needs or a drug that has been designated as a qualified infectious disease product. ü Accelerated Approval allows for approval of medication treating serious conditions based on surrogate endpoints. ü Priority Review is for medications that offer significant improvements in the safety or effectiveness of the treatment, diagnosis, or prevention of serious conditions when compared to standard applications. ü Breakthrough therapy designation is for medications that treat serious or life threatening disease and the medications may demonstrate substantial improvement on a clinically significant endpoint(s) over available therapies. . 3. We compiled analyzed the findings to determine the effect of FDA expedited programs on drug development timelines.

Methodology (2 of 2) Excluded NMEs from the analysis u Lumizyme and Corifact: Excluded entirely from the analysis as IND filings and EOP 2 meeting data are not available u 2014 approved NMEs Belsomra, Cerdelga, Jardiance, Jublia, Orbativ, Otezla, Pledridy, Sivextro, Striverdi Respimat, Tanzeum, Zydelig and Zykadia: Excluded as full FDA documentation is not published Sources used u IND filing: As reported in FDA filings - where information not available dates extracted from the Federal Register u End of Phase 2 meeting/beginning of pivotal trial -As reported in FDA filings – where the information was not available start dates of the pivotal trial extracted from www. clinicaltrials. gov u NDA/BLA filing: As reported in FDA filings u NDA/BLA approval: As reported in FDA filings *Sources: “Approved drugs 2013”, “ 2011 Novel New Drugs”, “ 2012 Novel New Drugs Summary”
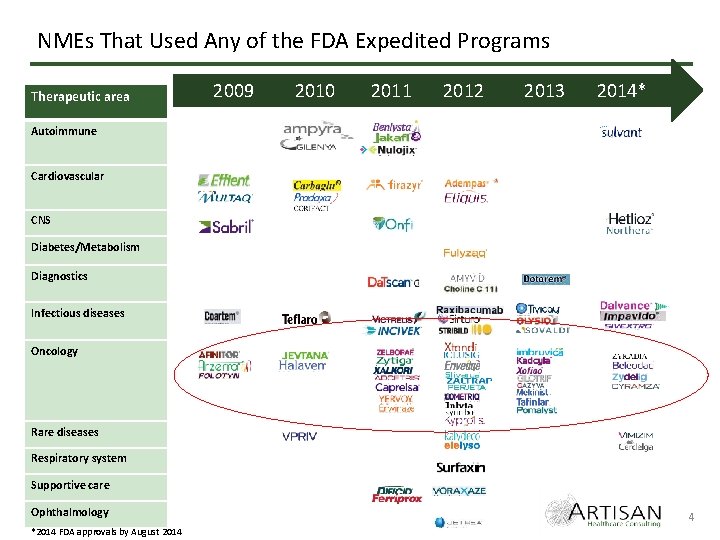
NMEs That Used Any of the FDA Expedited Programs Therapeutic area 2009 2010 2011 2012 2013 2014* Autoimmune Cardiovascular CNS Diabetes/Metabolism Diagnostics Infectious diseases Oncology Rare diseases Respiratory system Supportive care Ophthalmology *2014 FDA approvals by August 2014 4
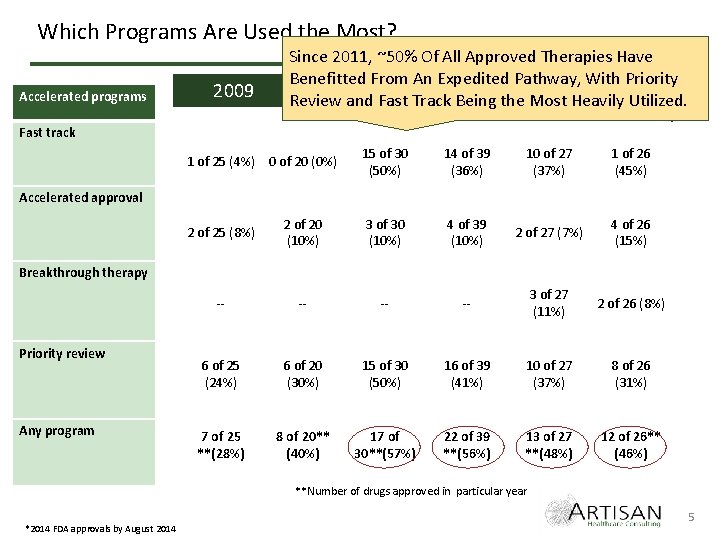
Which Programs Are Used the Most? Accelerated programs 2009 Since 2011, ~50% Of All Approved Therapies Have Benefitted From An Expedited Pathway, With Priority 2010 and 2011 2014* Review Fast Track 2012 Being the 2013 Most Heavily Utilized. Fast track 1 of 25 (4%) 0 of 20 (0%) 15 of 30 (50%) 14 of 39 (36%) 10 of 27 (37%) 1 of 26 (45%) 2 of 25 (8%) 2 of 20 (10%) 3 of 30 (10%) 4 of 39 (10%) 2 of 27 (7%) 4 of 26 (15%) -- -- 3 of 27 (11%) 2 of 26 (8%) 6 of 25 (24%) 6 of 20 (30%) 15 of 30 (50%) 16 of 39 (41%) 10 of 27 (37%) 8 of 26 (31%) 7 of 25 **(28%) 8 of 20** (40%) 17 of 30**(57%) 22 of 39 **(56%) 13 of 27 **(48%) 12 of 26** (46%) Accelerated approval Breakthrough therapy Priority review Any program **Number of drugs approved in particular year *2014 FDA approvals by August 2014 5
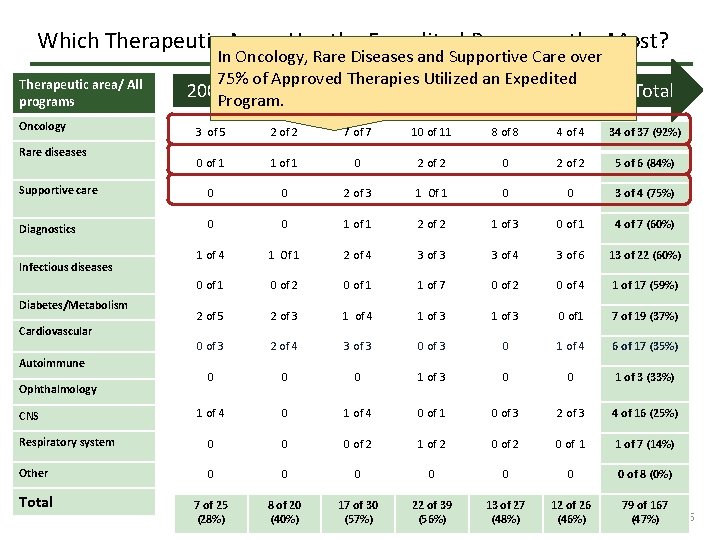
Which Therapeutic Areas Use the Expedited Programs the Most? Therapeutic area/ All programs Oncology In Oncology, Rare Diseases and Supportive Care over 75% of Approved Therapies Utilized an Expedited 2009 Program. 2010 2011 2012 2013 2014 Total 3 of 5 2 of 2 7 of 7 10 of 11 8 of 8 4 of 4 34 of 37 (92%) 0 of 1 1 of 1 0 2 of 2 5 of 6 (84%) Supportive care 0 0 2 of 3 1 Of 1 0 0 3 of 4 (75%) Diagnostics 0 0 1 of 1 2 of 2 1 of 3 0 of 1 4 of 7 (60%) 1 of 4 1 Of 1 2 of 4 3 of 3 3 of 4 3 of 6 13 of 22 (60%) 0 of 1 0 of 2 0 of 1 1 of 7 0 of 2 0 of 4 1 of 17 (59%) 2 of 5 2 of 3 1 of 4 1 of 3 0 of 1 7 of 19 (37%) 0 of 3 2 of 4 3 of 3 0 1 of 4 6 of 17 (35%) 0 0 0 1 of 3 (33%) 1 of 4 0 of 1 0 of 3 2 of 3 4 of 16 (25%) Respiratory system 0 0 0 of 2 1 of 2 0 of 1 1 of 7 (14%) Other 0 0 0 0 of 8 (0%) Total 7 of 25 (28%) 8 of 20 (40%) 17 of 30 (57%) 22 of 39 (56%) 13 of 27 (48%) 12 of 26 (46%) 79 of 167 (47%) Rare diseases Infectious diseases Diabetes/Metabolism Cardiovascular Autoimmune Ophthalmology CNS 6
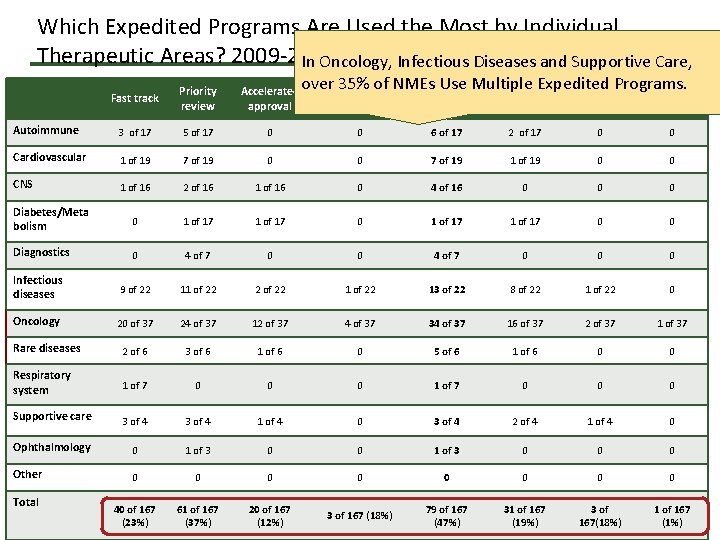
Which Expedited Programs Are Used the Most by Individual Therapeutic Areas? 2009 -2014 In Oncology, Infectious Diseases and Supportive Care, Fast track Priority review Accelerated approval Autoimmune 3 of 17 5 of 17 Cardiovascular 1 of 19 CNS over. Breakthrough 35% of NMEs Use Multiple Expedited Programs. Any single 3 All 4 therapy Program 0 0 6 of 17 7 of 19 0 0 1 of 16 2 of 16 1 of 16 Diabetes/Meta bolism 0 1 of 17 Diagnostics 0 Infectious diseases Oncology 2 programs 2 of 17 0 0 7 of 19 1 of 19 0 0 0 4 of 16 0 0 0 1 of 17 0 0 4 of 7 0 0 0 9 of 22 11 of 22 2 of 22 13 of 22 8 of 22 1 of 22 0 20 of 37 24 of 37 12 of 37 4 of 37 34 of 37 16 of 37 2 of 37 1 of 37 Rare diseases 2 of 6 3 of 6 1 of 6 0 5 of 6 1 of 6 0 0 Respiratory system 1 of 7 0 0 0 Supportive care 3 of 4 1 of 4 0 3 of 4 2 of 4 1 of 4 0 Ophthalmology 0 1 of 3 0 0 0 Other 0 0 0 0 40 of 167 (23%) 61 of 167 (37%) 20 of 167 (12%) 3 of 167 (18%) 79 of 167 (47%) 31 of 167 (19%) 3 of 167(18%) 1 of 167 (1%) 7 Total
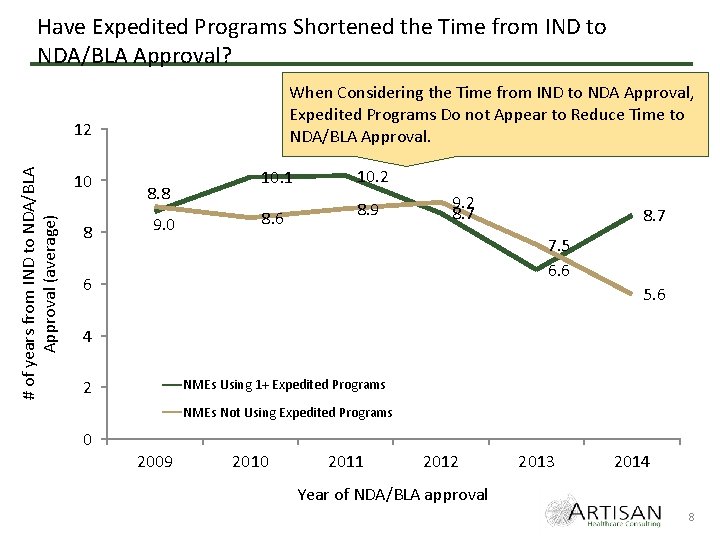
Have Expedited Programs Shortened the Time from IND to NDA/BLA Approval? When Considering the Time from IND to NDA Approval, Expedited Programs Do not Appear to Reduce Time to NDA/BLA Approval. # of years from IND to NDA/BLA Approval (average) 12 10 8 8. 8 9. 0 10. 1 8. 6 10. 2 8. 9 9. 2 8. 7 7. 5 6. 6 6 5. 6 4 NMEs Using 1+ Expedited Programs 2 NMEs Not Using Expedited Programs 0 2009 2010 2011 2012 2013 2014 Year of NDA/BLA approval 8
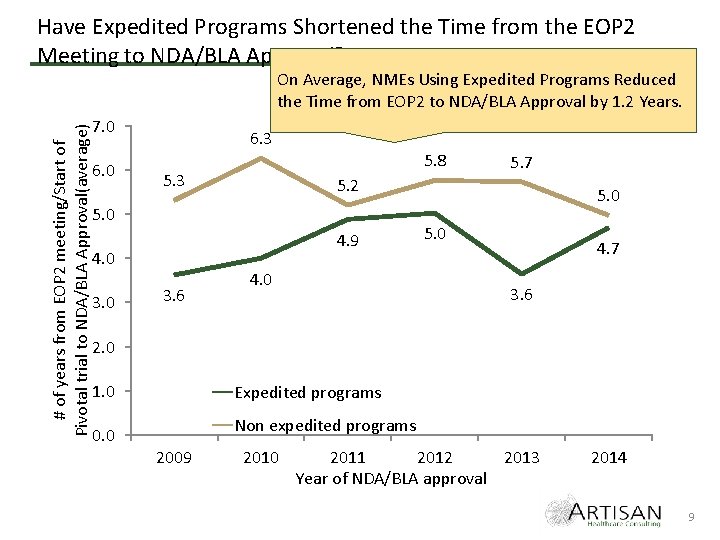
Have Expedited Programs Shortened the Time from the EOP 2 Meeting to NDA/BLA Approval? # of years from EOP 2 meeting/Start of Pivotal trial to NDA/BLA Approval(average) On Average, NMEs Using Expedited Programs Reduced the Time from EOP 2 to NDA/BLA Approval by 1. 2 Years. 7. 0 6. 3 5. 8 5. 3 5. 2 5. 0 4. 9 4. 0 3. 6 5. 7 4. 0 5. 0 4. 7 3. 6 2. 0 1. 0 0. 0 Expedited programs Non expedited programs 2009 2010 2011 2012 2013 Year of NDA/BLA approval 2014 9
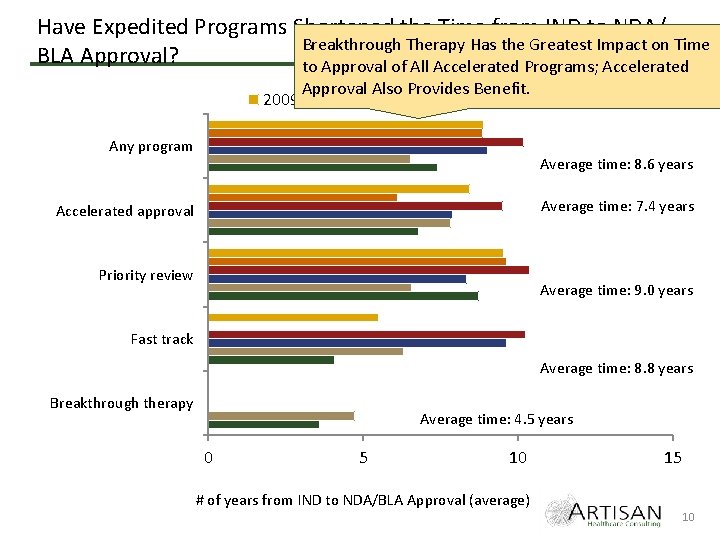
Have Expedited Programs Shortened the Time from IND to NDA/ Breakthrough Therapy Has the Greatest Impact on Time BLA Approval? to Approval of All Accelerated Programs; Accelerated 2009 Approval Also Provides Benefit. 2010 2011 2012 2013 Any program 2014 Average time: 8. 6 years Average time: 7. 4 years Accelerated approval Priority review Average time: 9. 0 years Fast track Average time: 8. 8 years Breakthrough therapy Average time: 4. 5 years 0 5 10 15 # of years from IND to NDA/BLA Approval (average) 10
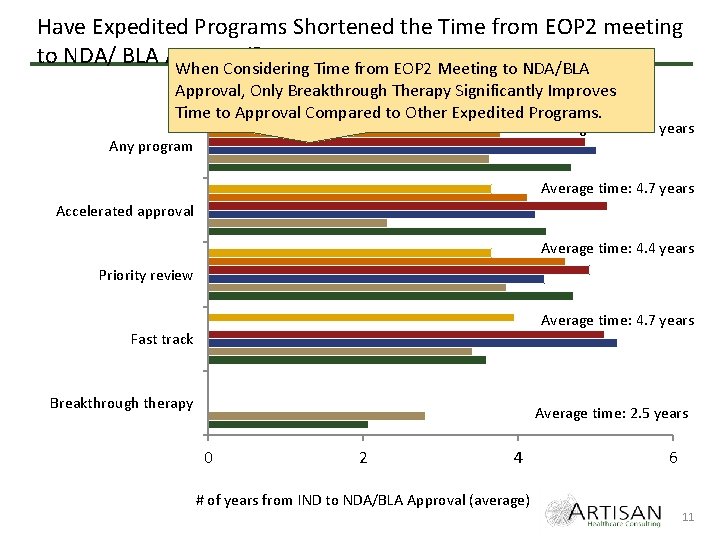
Have Expedited Programs Shortened the Time from EOP 2 meeting to NDA/ BLA Approval? When Considering Time from EOP 2 Meeting to NDA/BLA Approval, Only Breakthrough Therapy Significantly Improves 2009 2010 2011 2012 2013 2014 Time to Approval Compared to Other Expedited Programs. Average time: 4. 5 years Any program Average time: 4. 7 years Accelerated approval Average time: 4. 4 years Priority review Average time: 4. 7 years Fast track Breakthrough therapy Average time: 2. 5 years 0 2 4 6 # of years from IND to NDA/BLA Approval (average) 11
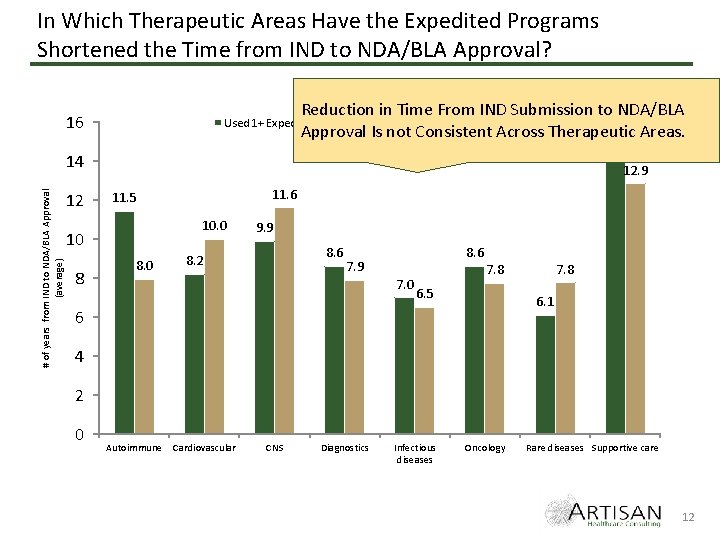
In Which Therapeutic Areas Have the Expedited Programs Shortened the Time from IND to NDA/BLA Approval? Reduction in Time From IND Submission to NDA/BLA No Expedited Programs Utilized 15. 0 Areas. Approval Is not Consistent Across Therapeutic 16 Used 1+ Expedited Program # of years from IND to NDA/BLA Approval (average) 14 12 12. 9 10. 0 10 8 11. 6 11. 5 8. 0 9. 9 8. 6 8. 2 8. 6 7. 9 7. 0 7. 8 6. 5 6. 1 6 4 2 0 Autoimmune Cardiovascular CNS Diagnostics Infectious diseases Oncology Rare diseases Supportive care 12
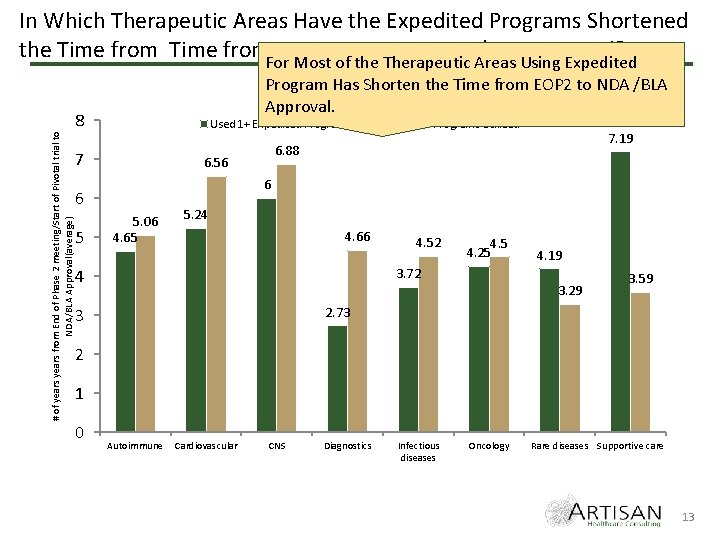
# of years from End of Phase 2 meeting/Start of Pivotal trial to NDA/BLA Approval(average) In Which Therapeutic Areas Have the Expedited Programs Shortened the Time from. For EOP 2 meting to NDA/BLA approval? Most of the Therapeutic Areas Using Expedited Program Has Shorten the Time from EOP 2 to NDA /BLA Approval. 8 Used 1+ Expedited Program 7 7. 19 6. 88 6. 56 6 6 5 No Expedited Programs Utilized 5. 06 4. 65 5. 24 4. 66 4. 52 4. 5 4. 25 3. 72 4 4. 19 3. 29 3. 59 2. 73 3 2 1 0 Autoimmune Cardiovascular CNS Diagnostics Infectious diseases Oncology Rare diseases Supportive care 13

Conclusions ✔ ✔ Since 2011, ~50% Of All Approved Therapies Have Benefitted From An Expedited Pathway, With Priority Review and Fast Track Being the Most Heavily Utilized. When Considering Time from EOP 2 meeting to NDA Approval FDA Expedited Programs Reduced the Time from EOP 2 to Approval by 1. 2 Years. The Expedited Programs Do not Shorten the Time from IND Submission to NDA/BLA Approval. Breakthrough Therapy Was introduced the Latest but Seems to Have the Biggest Impact on Approval Time Compared to Other Expedited Programs. In Oncology, Rare Diseases and Supportive Care over 75% of Approved Therapies Utilized an Expedited Program. In Oncology, Infectious Diseases and Supportive Care, over 35% of NMEs Use Multiple Expedited Programs. 14

Brad Payne - Bio Brad Payne is a Team Leader for Artisan Healthcare Consulting. He has over 5 years experience in the healthcare consulting space, with expertise in domestic and international reimbursement and market access. • Lead of the global market access practice • Extensive knowledge of national and regional payers in Europe, Canada, Latin America, and Asia/Pacific • Advanced understanding of U. S. market access, with a proven track record of developing strategy that influenced national and regional payer coverage and payment decisions • Proven ability to develop market access strategies utilizing experience across a number of therapeutic areas including oncology, immunology, infectious diseases, rare diseases, and devices • Understanding of health economics, with extensive expertise in Health Technology Assessment (HTA) analyses Prior to joining Artisan, Brad was a consultant at Trinity Partners, a pharmaceutical and biotech consulting firm, where he provided insight on forecasting projects, licensing and acquisition opportunities, and provided recommendations for managed care contracting strategies. Brad graduated with a degree in economics Cum Laude from Harvard University and received an MBA from the W. P. Carey School of Business at Arizona State University. Brad Payne Team Leader +1 781. 996. 7338 office +1 702. 250. 3136 cell bpayne@artisan-consulting. com 15
- Slides: 15