September 4 2009 HITSP 2009 TC Timeline Overview
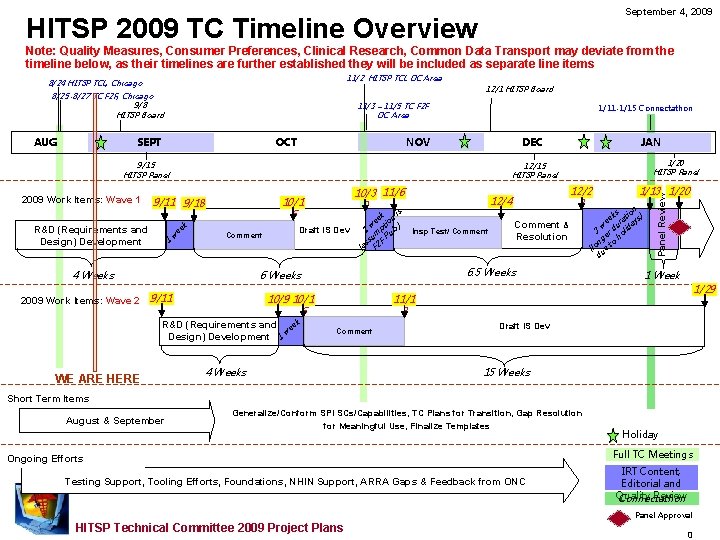
September 4, 2009 HITSP 2009 TC Timeline Overview Note: Quality Measures, Consumer Preferences, Clinical Research, Common Data Transport may deviate from the timeline below, as their timelines are further established they will be included as separate line items 8/25 -8/27 TC F 2 F, Chicago 9/8 HITSP Board AUG 12/1 HITSP Board 11/3 – 11/5 TC F 2 F DC Area SEPT OCT 1/11 -1/15 Connectathon DEC NOV 9/15 HITSP Panel 2009 Work Items: Wave 1 1 w ee k 10/3 11/6 0 10/1 6 Comment Draft IS Dev is k ee tion ) w 1 mp ub. u P s s F (a F 2 9/11 Insp Test/ Comment WE ARE HERE Comment & Resolution 6. 5 Weeks 10/9 10/1 6 R&D (Requirements and eek Design) Development 1 w 12/2 3 12/4 6 Weeks 4 Weeks 2009 Work Items: Wave 2 1/20 HITSP Panel 12/15 HITSP Panel 9/11 9/18 R&D (Requirements and Design) Development JAN 1/13 1/20 n ks tio ) ee ura ays w 3 er d olid h ng to (lo ue d Panel Review 11/2 HITSP TCL DC Area 8/24 HITSP TCL, Chicago 1 Week 1/29 11/1 3 Draft IS Dev Comment 4 Weeks 15 Weeks Short Term Items August & September Generalize/Conform SPI SCs/Capabilities, TC Plans for Transition, Gap Resolution for Meaningful Use, Finalize Templates Ongoing Efforts Testing Support, Tooling Efforts, Foundations, NHIN Support, ARRA Gaps & Feedback from ONC Holiday Full TC Meetings IRT Content, Editorial and Quality Review Connectathon Panel Approval HITSP Technical Committee 2009 Project Plans 0

2009 Work Items Split Up by Waves 4 Activities During Wave 1 4 Activities During Wave 2 – Quality Measures (Pop) – Common Data Transport** (SPI) – Newborn Screening (Pop) ONC Priorities – Consumer Preferences** (SPI/Cons) – Clinical Research (Pop) – General Lab Orders (Prov) – Medication Gaps (Cons) – Prior-Authorization (Admin) 4 Rationale – ONC high-priority items – For the purpose of proper resource utilization, the Program Management Team selected additional work items to include in Wave 1, supplemental to ONC identified priorities so that each TC would have a work item in Wave 1. CMHR/Data Architecture was not included as they have other prioritized items to complete HITSP Technical Committee 2009 Project Plans (Nominal Risk) – Clinical Encounter Notes (CMHR) – Common Device Connectivity (Cons) – Long Term Care (Prov) – Maternal and Child Health (Pop) – Medical Home: Co-Morbidity & Registries (Cons) – Order Sets (Prov) – Scheduling (Admin) 4 Wave 3 (Greater Risk) – Consumer Adverse Event Reporting (Pop) ** = May be on different schedule – beginning in August 1
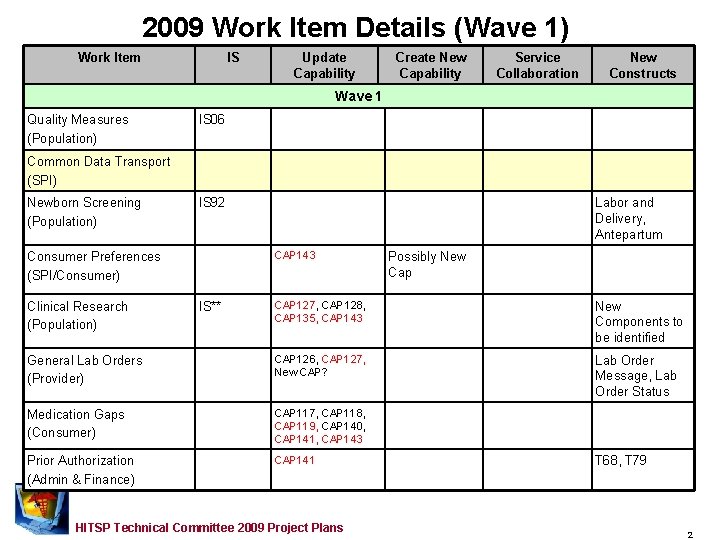
2009 Work Item Details (Wave 1) Work Item IS Update Capability Create New Capability Service Collaboration New Constructs Wave 1 Quality Measures (Population) IS 06 Common Data Transport (SPI) Newborn Screening (Population) IS 92 Labor and Delivery, Antepartum CAP 143 Consumer Preferences (SPI/Consumer) Possibly New Cap CAP 127, CAP 128, CAP 135, CAP 143 New Components to be identified General Lab Orders (Provider) CAP 126, CAP 127, New CAP? Lab Order Message, Lab Order Status Medication Gaps (Consumer) CAP 117, CAP 118, CAP 119, CAP 140, CAP 141, CAP 143 Prior Authorization (Admin & Finance) CAP 141 Clinical Research (Population) IS** HITSP Technical Committee 2009 Project Plans T 68, T 79 2
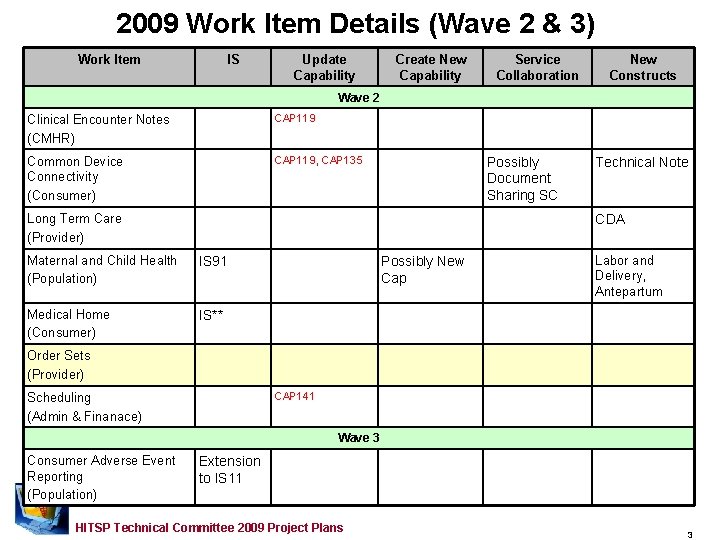
2009 Work Item Details (Wave 2 & 3) Work Item IS Update Capability Create New Capability Service Collaboration New Constructs Wave 2 Clinical Encounter Notes (CMHR) CAP 119 Common Device Connectivity (Consumer) CAP 119, CAP 135 Possibly Document Sharing SC Long Term Care (Provider) Technical Note CDA Maternal and Child Health (Population) IS 91 Medical Home (Consumer) IS** Possibly New Cap Labor and Delivery, Antepartum Order Sets (Provider) CAP 141 Scheduling (Admin & Finanace) Wave 3 Consumer Adverse Event Reporting (Population) Extension to IS 11 HITSP Technical Committee 2009 Project Plans 3
- Slides: 4