September 2021 Shapes of Molecules LO Predict shapes
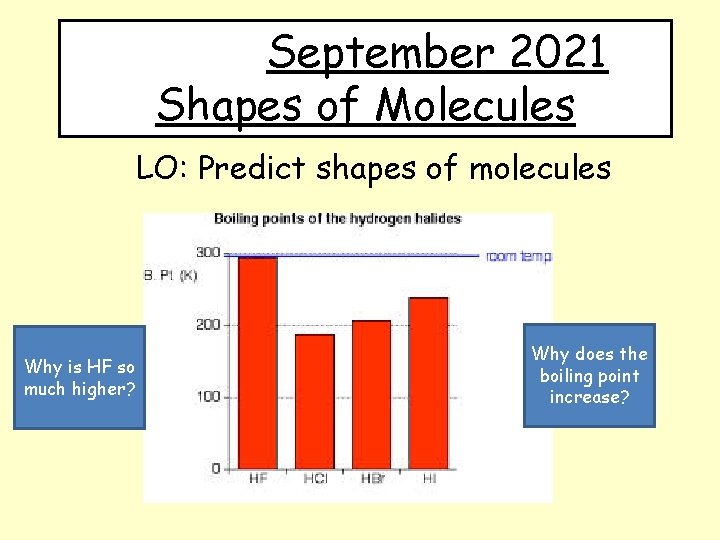
September 2021 Shapes of Molecules LO: Predict shapes of molecules Why is HF so much higher? Why does the boiling point increase?

Learning Objectives • Describe electron pair repulsion theory • Explain how the number of electron pairs around an atom affects the shape of the molecule • Describe the effect lone pairs have on shapes

Electron Pair Repulsion Theory • Pairs of electrons will repel other electron pairs • The electron pairs will move as far away from each other as possible

First task • Draw the dot and cross diagrams to find the number of bonded and lone pairs of electrons – Be. Cl 2 – BF 3 – CH 4 – PCl 5 – SF 6

Molymods • Make as many of them with the molymods as you can – Be. Cl 2 – BF 3 – CH 4 – PCl 5 – SF 6
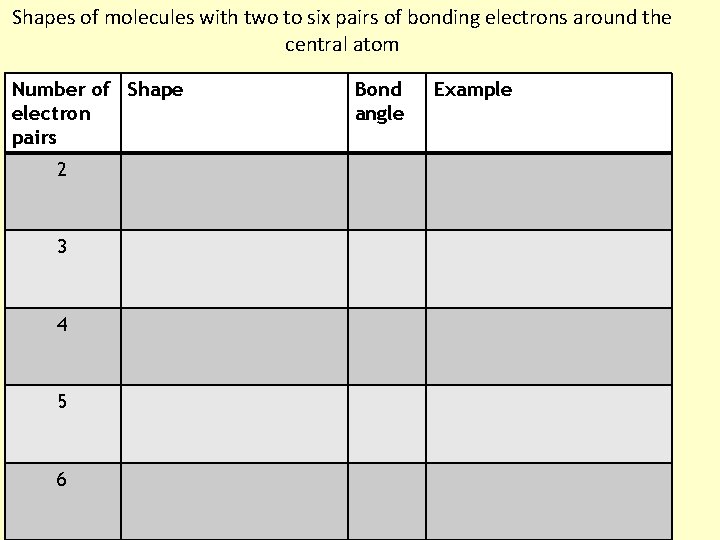
Shapes of molecules with two to six pairs of bonding electrons around the central atom Number of Shape electron pairs 2 3 4 5 6 Bond angle Example
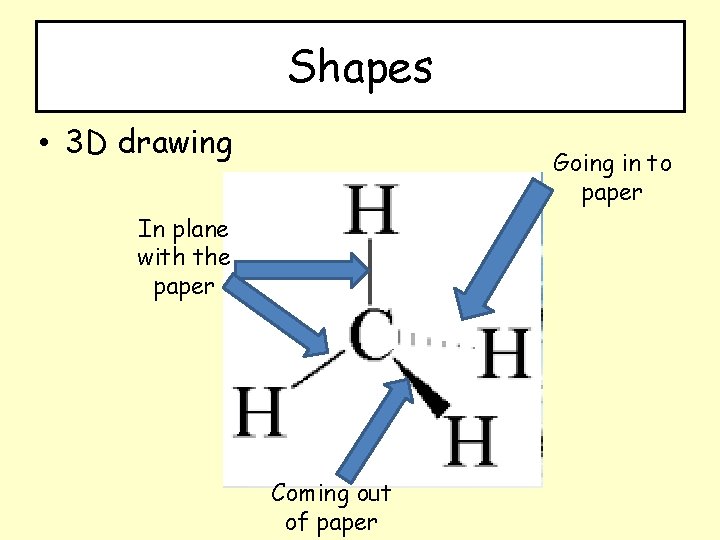
Shapes • 3 D drawing Going in to paper In plane with the paper Coming out of paper

Test your-self 1 a) Draw dot and cross diagram, showing only the outer electrons for: – – Al. Cl 3 PF 5 Si. H 4 BCl 3 b) Predict the shape and bond angle for the molecules above.
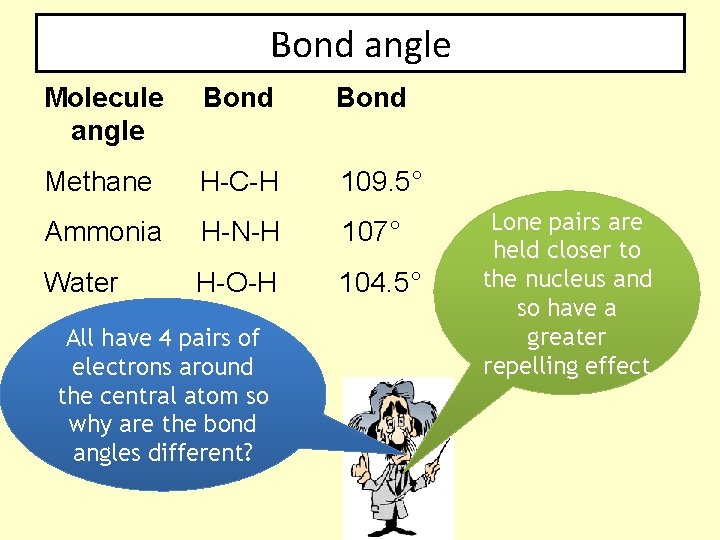
Bond angle Molecule angle Bond Methane H-C-H 109. 5° Ammonia H-N-H 107° Water H-O-H 104. 5° All have 4 pairs of electrons around the central atom so why are the bond angles different? Lone pairs are held closer to the nucleus and so have a greater repelling effect
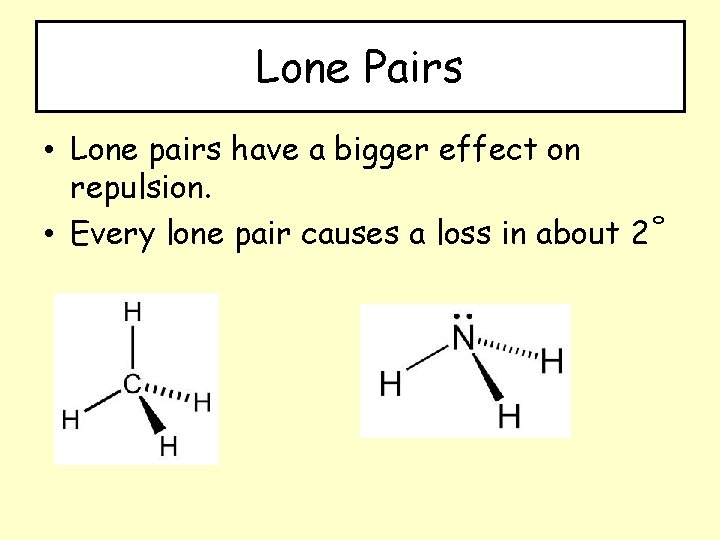
Lone Pairs • Lone pairs have a bigger effect on repulsion. • Every lone pair causes a loss in about 2˚
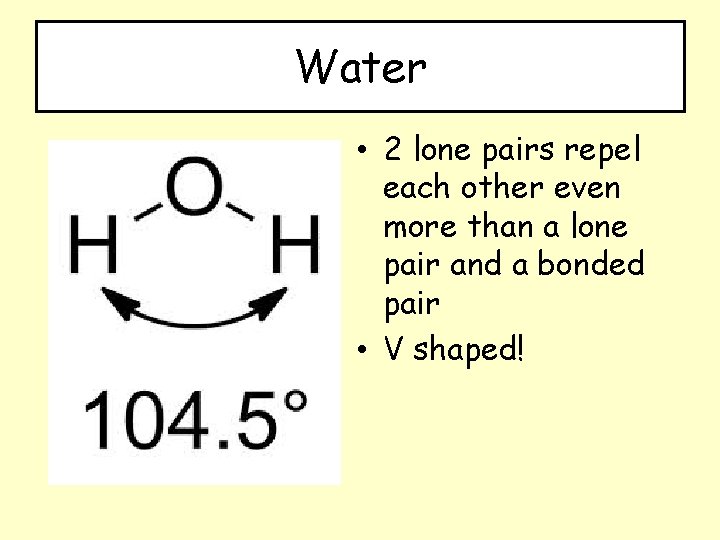
Water • 2 lone pairs repel each other even more than a lone pair and a bonded pair • V shaped!
Dative Bonding Effect

Chlorine Tetraflouride ion • Cl. F 4 • The lone pairs repel each other as far away as possible leaving the 4 bonded pairs in the middle • They form a square planar

Test your-self 1. Draw dot and cross diagram, for CH 4 and H 2 O a) Explain why the bond angle H-C-H is 109. 5⁰ in CH 4 and the bond angle H-O-H is 104. 5⁰ and not 180⁰. b) Why is the bond angle in H 2 O less than 109. 5 ⁰? c) Suggest a name for the shape of the H 2 O molecule. 2. Make predictions about shapes and bond angles for H 3 O+, BF 4, NH 4+.

Past paper questions: Jun 2009

Past paper questions: Jun 2010 a) HCH in methane, CH 4 b) FSF in sulfur hexafluoride, SF 6 c) FOF in oxygen difluoride, OF 2

Past paper questions: Jan 2010 and Jun 2009

Past paper questions: Jan 2010

2008 -2012 shapes • These are the shapes that came up between 2008 and 2012 in exam papers. Pupils were asked for dot and cross diagrams, 3 d diagram, shape name and bond angles CH 2 CH 3+ BF 3 H 2 S NH 4+ CCI 2 F 2 CIF 3 BF 4– Cl. F 2+ As. F 5 As. H 3 NH 2 H 2 Se NH 3 PH 4+ Br. F 3 Br. F 4 NHF 2 BF 3 H 3 O +
- Slides: 19