SEPSIS AND MENINGOCOCCEMIA Assoc Prof Dr Suat Bier

SEPSIS AND MENINGOCOCCEMIA Assoc. Prof. Dr. Suat Biçer Faculty of Medicine, Yeditepe University

Purposes of this lecture: Definition of SIRS, sepsis, severe sepsis, septic shock, and MODS Diagnosis of sepsis, septic shock, and meningococcemia Treatment of sepsis, septic shock, and meningococcemia

Introduction: Sepsis remains a major cause of morbidity and mortality among children. Sepsis-associated mortality in children decreased from 97% in 1966 (7) to 9% among infants in the early 1990 s (8). 3. Hazelzet JA, Risseeuw-Appel IM, Kornelisse RF, et al: Age-related differences in outcome and severity of DIC in children with septic shock and purpura. Thromb Haemost 1996; 76: 932– 938 4. Anderson MR, Blumer JL: Advances in therapy for sepsis in children. Pediatr Clin North Am 1997; 44: 179– 205 5. Martinot A, Leclerc F, Cremer R, et al: Sepsis in neonates and children: Definitions, epidemiology, and outcome. Pediatr Emerg Care 1997; 13: 277– 281 6. Despond O, Proulx F, Carcillo JA, et al: Pediatric sepsis and multiple organ dysfunction syndrome. Curr Opin Pediatr 2001; 13: 247– 253 8. Stoll BJ, Holman RC, Schuchat A: Decline in sepsis-associated neonatal and infant deaths in the United States, 1979 through 1994. Pediatrics 1998; 102: e 18

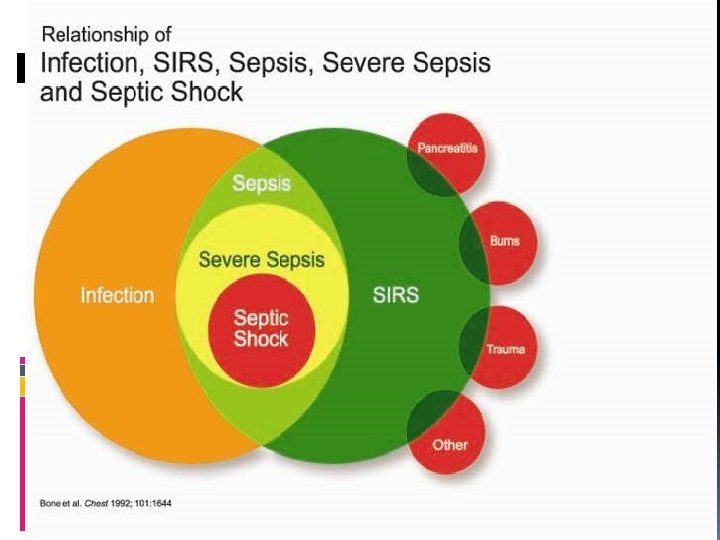
Nomenclature: SIRS (Systemic Inflammatory Response Syndrome), infection, sepsis, severe sepsis, septic shock, multiple organ dysfunction syndrome (MODS)


Relevant questions: 1. How should the pediatric age groups affected by sepsis be delineated? 2. What are the specific definitions of pediatric SIRS, infection, sepsis, severe sepsis, and septic shock? 3. What are the specific definitions of pediatric organ failure and the validity of pediatric organ failure scores?

Pediatric age groups for severe sepsis definitions: Newborn 0 days to 1 wk Neonate 1 wk to 1 mo Infant 1 mo to 1 yr Toddler and preschool 2– 5 yrs School age child 6– 12 yrs Adolescent and young adult 13 to 18 yrs

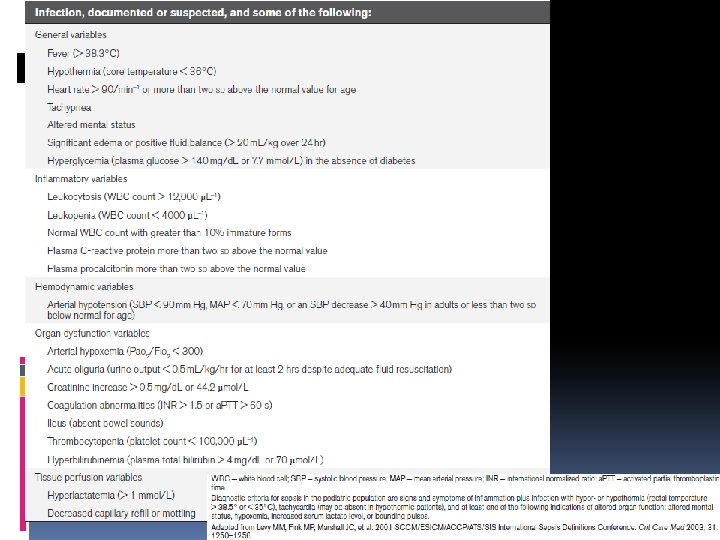
Definitions: SIRS (Systemic inflammatory response syndrome): The presence of at least two of the following four criteria, (one of which must be abnormal temperature or leukocyte count): Core temperature of > 38. 5°C or < 36°C. Tachycardia, defined as a mean heart rate >2 SD above normal for age in the absence of external stimulus, chronic drugs, or painful stimuli; or otherwise unexplained persistent elevation over a 0. 5 - to 4 -hr time period or for children <1 yr old: bradycardia, defined as a mean heart rate <10 th percentile for age in the absence of external vagal stimulus, β-blocker drugs, or congenital heart disease; or otherwise unexplained persistent depression over a 0. 5 -hr time period. Mean respiratory rate > 2 SD above normal for age or mechanical ventilation for an acute process not related to underlying neuromuscular disease or the receipt of general anesthesia. Leukocyte count elevated or depressed for age (not secondary to chemotherapy-induced leukopenia) or >10% immature neutrophils.

Infection: A suspected or proven (by positive culture, tissue stain, or polymerase chain reaction test) infection caused by any pathogen OR a clinical syndrome associated with a high probability of infection. Evidence of infection includes positive findings on clinical exam, imaging, or laboratory tests (e. g. , white blood cells in a normally sterile body fluid, perforated viscus, chest radiograph consistent with pneumonia, petechial or purpuric rash, or purpura fulminans)

Sepsis: SIRS in the presence of or as a result of suspected or proven infection. Severe sepsis: Sepsis plus one of the following: • Cardiovascular organ dysfunction OR • Acute respiratory distress syndrome OR • Two or more other organ dysfunctions. Septic shock: Sepsis and cardiovascular organ dysfunction.
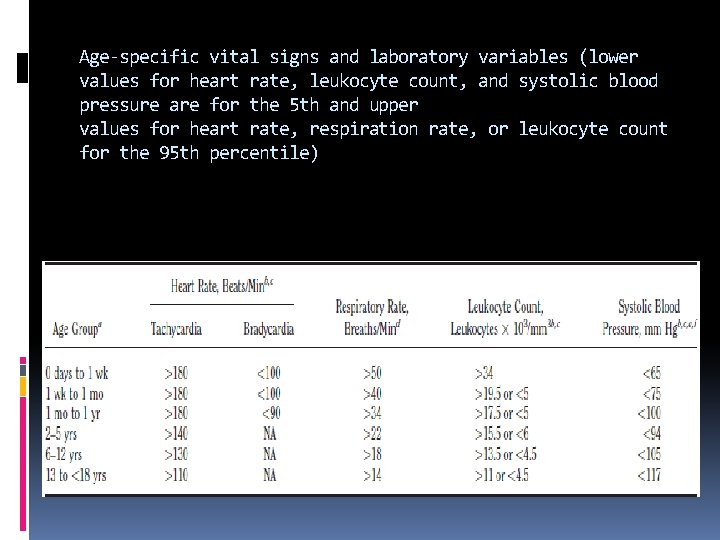
Age-specific vital signs and laboratory variables (lower values for heart rate, leukocyte count, and systolic blood pressure are for the 5 th and upper values for heart rate, respiration rate, or leukocyte count for the 95 th percentile)

Tachycardia and tachypnea are common presenting symptoms of many pediatric disease processes. Therefore, the major difference in the definition of SIRS between adults and children is that the diagnosis of pediatric SIRS requires that temperature or leukocyte abnormalities be present (SIRS should not be diagnosed if a pediatric patient exhibits only elevated heart and respiratory rates).

Fever Children with core temperatures of ≥ 38°C may be considered to have fever. (38, 39) However, a temperature of ≥ 38. 5°C improves specificity and reflects clinical intensive care unit practice. A core temperature by either rectal, bladder, oral, or central catheter probe is required. Temperatures taken via the tympanic, toe, or axillary route are not sufficiently accurate. Hypothermia (< 36°C) may also indicate serious infection, especially in infants. (38, 40, 41) 38. Baraff LJ: Management of the febrile child: A survey of pediatric and emergency medicine residency directors. Pediatr Infect Dis J 1991; 10: 795– 800 39. Baraff LJ, Bass JW, Fleisher GR, et al: Practice guideline for the management of infants and children 0 to 36 months of age with fever without source. Pediatrics 1993; 92: 1– 12 40. Kline MW, Lorin MI: Bacteremia in children afebrile at presentation to an emergency department. Pediatr Infect Dis J 1989; 6: 197– 198 41. Bonadio WA: Incidence of serious infections in afebrile neonates with a history of fever. Pediatr Infect Dis J 1987; 6: 911– 914

Biochemical markers of inflammation may one day prove to be more objective and reliable than physiologic variables. Elevated sedimentation rate, C reactive protein, base deficit, interleukin-6, and procalcitonin levels have been reported as potential biochemical markers of SIRS. (42– 51) However, although some markers are sensitive they lack specificity, and no biochemical markers have been confirmed to be robust enough to add to the general definition at this time.

Infection could be of bacterial, viral, fungal, or rickettsial origin. Examples of clinical findings indicating an infection include petechiae and purpura in the setting of hemodynamic instability; fever, cough, and hypoxemia in the setting of leukocytosis and pulmonary infiltrates; or distended tympanitic abdomen with fever and leukocytosis associated with a perforated bowel.
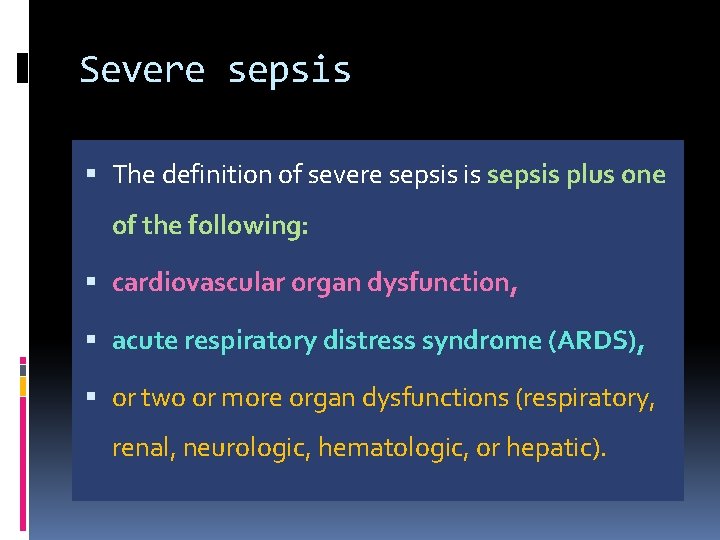
Severe sepsis The definition of severe sepsis is sepsis plus one of the following: cardiovascular organ dysfunction, acute respiratory distress syndrome (ARDS), or two or more organ dysfunctions (respiratory, renal, neurologic, hematologic, or hepatic).
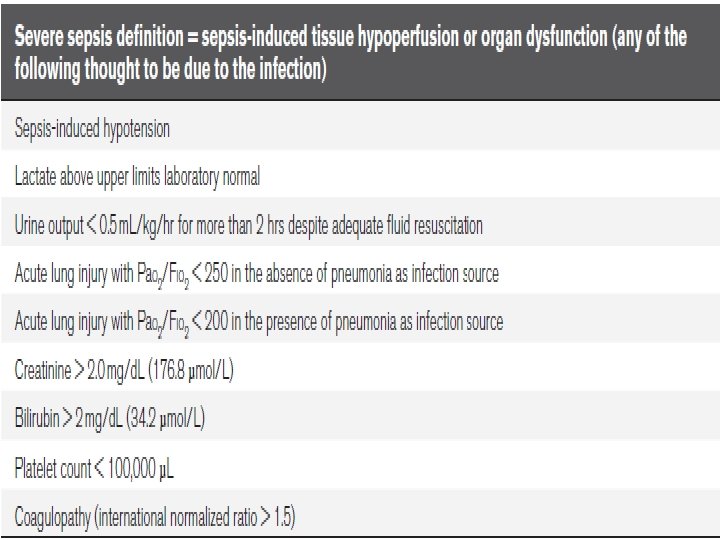

Septic shock The definition of septic shock As children often will maintain their blood pressure until they are severely ill, there is no requirement for systemic hypotension to make the diagnosis of septic shock as there is in adults. Shock may occur long before hypotension occurs in children.

Carcillo et al. defined septic shock in pediatric patients as tachycardia (which may be absent in the hypothermic patient) with signs of decreased perfusion including; decreased peripheral pulses compared with central pulses, altered alertness, flash capillary refill or capillary refill > 2 secs, mottled or cool extremities, or decreased urine output. Hypotension is a sign of late and decompensated shock in children and, although not needed for the definition, is confirmatory of shock state if present in a child with suspected or proven infection. (52) 52. Carcillo JA, Fields AI, Task Force Committee Members: Clinical practice variables for hemodynamic support of pediatric and neonatal patients in septic shock. Crit Care Med 2002; 30: 1365– 1378

Organ dysfunction criteria Cardiovascular dysfunction: Despite administration of isotonic intravenous fluid bolus ≥ 40 m. L/kg in 1 hr, Decrease in BP (hypotension) < 5 th percentile for age or systolic BP < 2 SD below normal for age OR Need for vasoactive drug to maintain BP in normal range (dopamine > 5 g/kg/min or dobutamine, epinephrine, or norepinephrine at any dose) OR Two of the following - Unexplained metabolic acidosis: base deficit > 5. 0 m. Eq/L - Increased arterial lactate > 2 times upper limit of normal - Oliguria: urine output < 0. 5 m. L/kg/hr - Prolonged capillary refill > 5 secs - Core to peripheral temperature gap > 3°C

Respiratory: Pa. O 2/FIO 2 < 300 in absence of cyanotic heart disease or preexisting lung disease OR Pa. CO 2 > 65 torr or 20 mm Hg over baseline Pa. CO 2 OR Proven need or > 50% FIO 2 to maintain saturation ≥ 92% OR Need for nonelective invasive or noninvasive mechanical ventilation

Neurologic: Glasgow Coma Score ≤ 11 OR Acute change in mental status with a decrease in Glasgow Coma Score ≥ 3 points from abnormal baseline

Hematologic: Platelet count 80, 000/mm 3 or a decline of 50% in platelet count from highest value recorded over the past 3 days (for chronic hematology/oncology patients) OR International normalized ratio > 2

Renal: Serum creatinine ≥ 2 times upper limit of normal for age or 2 -fold increase in baseline creatinine Hepatic: Total bilirubin ≥ 4 mg/d. L (not applicable for newborn) OR ● ALT 2 times upper limit of normal for age ●


FUTURE DIRECTIONS The definition of sepsis in children needs further refinement and requires a series of evidence-based consensus conferences in the future. We hope that these definitions will provide a uniform basis for clinicians and researchers to study and diagnose severe sepsis in children.

Etiology - Newborns: Group B streptococci, E. coli, Listeria monocytogenes, enteroviruses, Herpes Simplex, Enterobacter, Stafilococcus aureus. Children: - Streptococcus pneumoniae, - Neisseria meningitidis, - Staf. aureus, - Haemophilus influenzae (not immunized) Children with immune deficiency: - Staf. aureus, - Pseudomonas sp. - Candida albicans

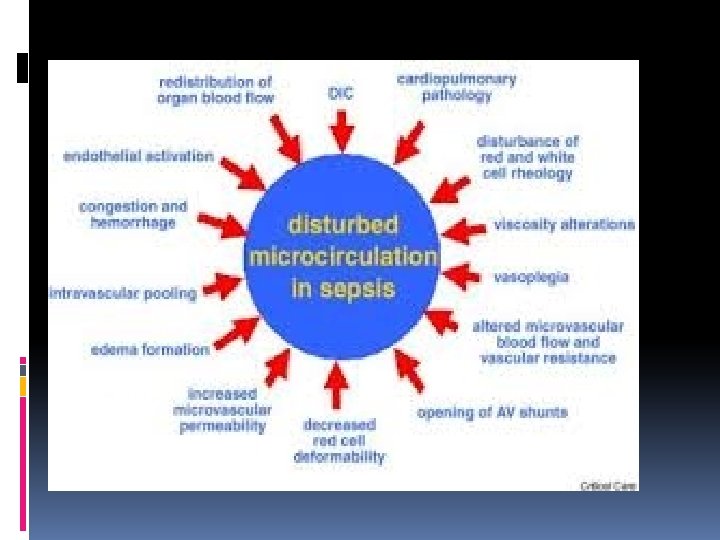
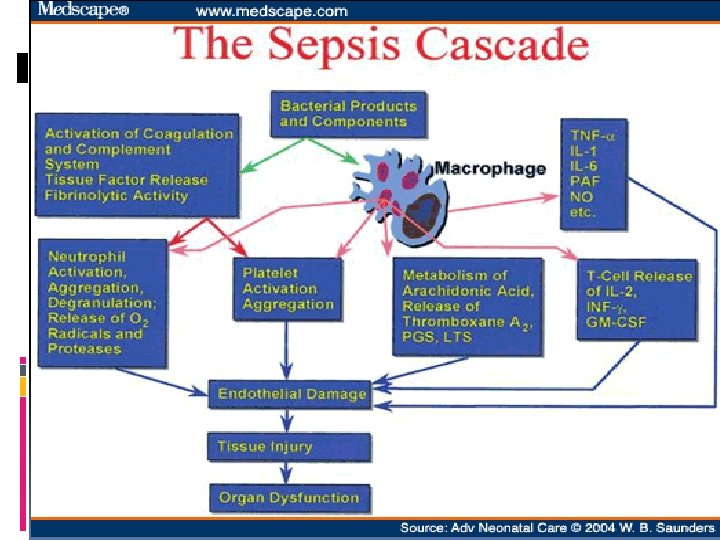
Pathogenesis Tissue injury or organ failure from bacteria or bacterial products, Excessive inflammatory response of the host, Inadequate anti-inflammatory response of the host.

Diagnosis Leukocyte, neutrophil, CRP and procalcitonin Biochemistry (ALT, AST, BUN, creatinin) Coagulatin (PT, a. PTT, INR, fibrinogen, D-Dimer) Blood culture Urine analysis, and culture Stool culture Viral culture, ELISA, immunochromatography, PCR

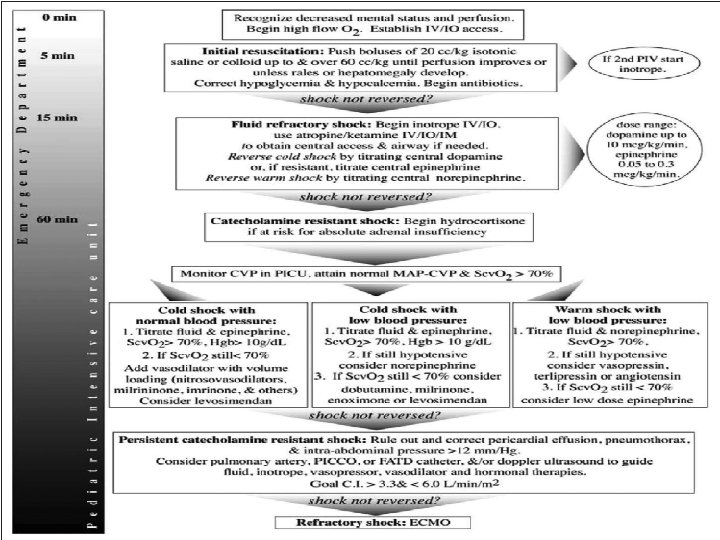
Treatment Supportive treatment - Oxygenation and respiratory support methods (correction of tissue hypoxia) - Bolus and maintenance fluid-electrolyte - Vasoactive drugs - Anti-inflammatory therapy - Coagulation therapy Antibiotherapy Antiviral or antifungal drugs


Meningococcal diseases - meningococcemia Neisseria meningitidis (Meningococcus) – Gram-Negative Bacteria - There are 13 chemically and serologically distinct meningococcal capsular groups, of which five, designated A, B, C, W-135 and Y, are responsible for almost all cases of human disease. - One of the most dangerous gram-negative bacterial infections - N. meningitidis also is unique for its ability to cause epidemic bacterial meningitis and sepsis. - The organism remains an important cause of serious endemic disease in the country and of epidemic disease throughout the world. - Despite advances in critical care medicine, previously healthy children and adolescents continue to succumb to fulminant meningococcal disease. It lives as a commensal in the nasopharynx of humans and is typically carried by 10% or more of the population at any one time. Relatively rarely the organism enters the bloodstream and may cause devastating disease. Why invasive meningococcal disease develops in a small proportion of exposed individuals is still largely not understood.

Epidemiology Meningococci are transmitted by aerosol droplets or through contact with respiratory secretions, such as through kissing or sharing a drinking glass. The organism is not thought to survive for long periods in the environment, and transmission is decreased during periods of high ambient ultraviolet B radiation. Viral respiratory infections (influenza), exposure to tobacco smoke, marijuana use, bar patronage, binge drinking, attendance at nightclubs, and freshmen college students living in dormitories are all associated with increased rates of meningococcal carriage or disease. Respiratory viruses and/or exposure to smoke may alter the mucosal surface and enhance bacterial binding and/or decrease clearance of the organism from the nasopharynx.

Pathogenesis After exposure to meningococci, attachment of an organism to nasopharyngeal mucosal cells is mediated by specific bacterial adhesins. The most important virulence determinant is the presence of a capsular polysaccharide, which enhances resistance of the organism to killing by normal human serum and helps resist opsonophagocytic killing. Although carriage can persist for weeks to months, onset of invasive meningococcal disease usually occurs within a few days to a week after acquisition of the organism. Development of disease depends on the virulence of the organism, innate susceptibility of the host, and presence or absence of serum antibodies capable of activating complement-mediated bacteriolysis and/or opsonophagocytosis.

The severity of meningococcal disease is related to the circulating level of endotoxin in the bloodstream. During bacterial growth, outer membrane blebs, which are rich in endotoxin, are released. Meningococcal endotoxin is composed of lipopolysaccharide—also referred to as lipooligosaccharide (LOS) because of the presence of repeating short saccharides instead of long-chain saccharides characteristic of endotoxins of many other gram-negative bacteria.

Stimulation of Toll-like receptors (TLRs) activates genes via pathways related to nuclear factor-κB (NF-κB), which leads to production of multiple proinflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and IL-8. Subsequently both the extrinsic (by way of induction of tissue factor expression on endothelial cells and monocytes) and intrinsic pathways of coagulation are activated. Progression of capillary leak and disseminated intravascular coagulopathy (DIC) can lead to multiple organ system failure, septic shock, and death. Following initiation of antibiotic therapy, circulating LOS and TNF-α levels can increase transiently as a result of rapid bacterial lysis, which then decreases with clearance of viable microbes. Activation of the complement and clotting cascades can continue well beyond this point, especially in fulminant cases.

Diffuse vasculitis and DIC are common with meningococcemia. Leukocyte-rich fibrin clots are seen in small vessels, including arterioles and capillaries. The resulting focal hemorrhage and necrosis that initially manifest as purpura in the skin may occur in any organ. The heart, central nervous system, skin, mucous and serous membranes, and adrenal glands are affected in most fatal cases, and microbes are often present in these lesions. Myocarditis is present in >50% of patients who die of meningococcal disease. Diffuse adrenal hemorrhage without vasculitis, the Waterhouse-Friderichsen syndrome, is common during fulminant meningococcemia. Meningitis is characterized by acute inflammatory cells in the leptomeninges and perivascular spaces. Focal cerebritis is uncommon.

Immunity & Host Factors A large number of host immunologic and genetic factors appear to affect the risk and/or severity of meningococcal disease. Persons with inherited deficiencies of properdin, factor D, or terminal complement components have up to a 1000 -fold higher risk for development of meningococcal disease than complement-sufficient persons. The risk of meningococcal disease is also increased in patients with acquired complement deficiencies associated with diseases such as nephrotic syndrome, systemic lupus erythematosus, and hepatic failure. Among persons with complement deficiencies, meningococcal disease is more prevalent during late childhood and adolescence, when carriage rates are higher than in children <10 yr; meningococcal infections may be recurrent. The presence of factor V Leiden, which is known to increase the risk of thrombosis, also may exacerbate meningococcal purpura fulminans. Plasminogen activator converts plasminogen into its active form, plasmin, which elicits fibrinolysis. Functional polymorphisms in the promoter region of the gene for plasminogen-activator-inhibitor-1, which result in higher inhibitor levels and decreased fibrinolysis, have been associated with increased severity of meningococcal disease.
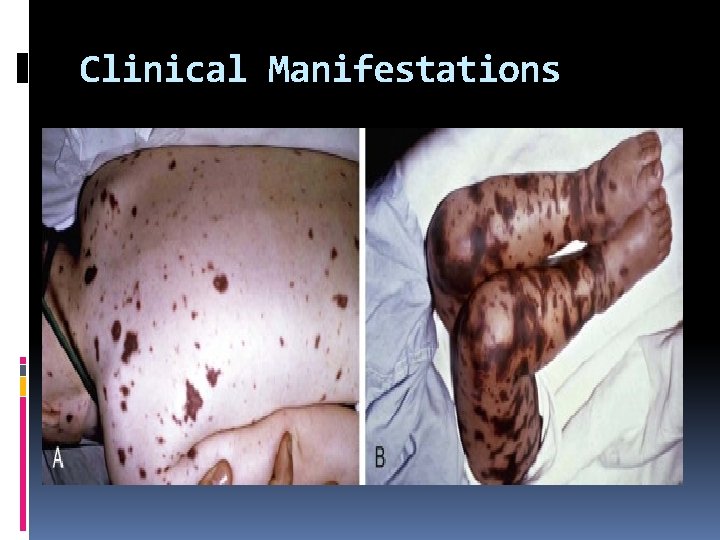
Clinical Manifestations

The spectrum of meningococcal disease varies widely, and recognized patterns include bacteremia without sepsis, meningococcemia without meningitis, meningitis with or without meningococcemia, and chronic infection. At least 80% of cases have overt clinical signs. Occult meningococcal bacteremia often manifests as fever with or without associated symptoms that suggest minor viral infections. Resolution of bacteremia may occur without antibiotics, but sustained bacteremia leads to meningitis in ≈60% of cases and to distant infection of other tissues. N. meningitidis is isolated from blood in about 65% of patients with meningococcal infections, from cerebrospinal fluid (CSF) in about 50% of patients, and from joint fluid in 1% of patients.

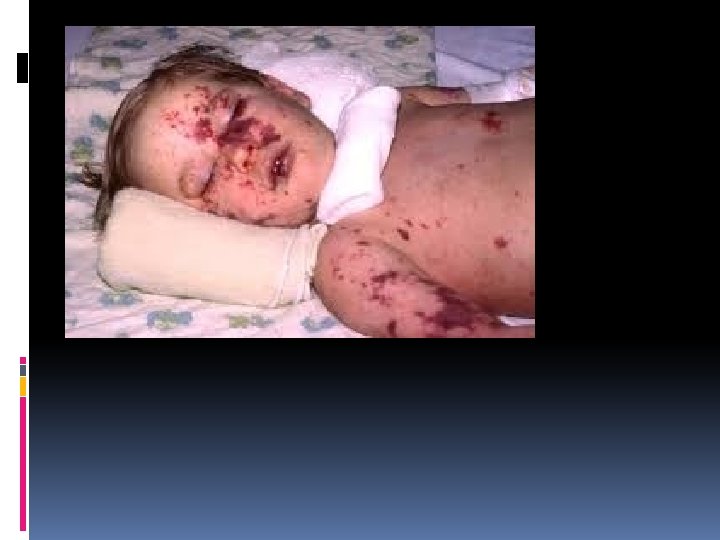
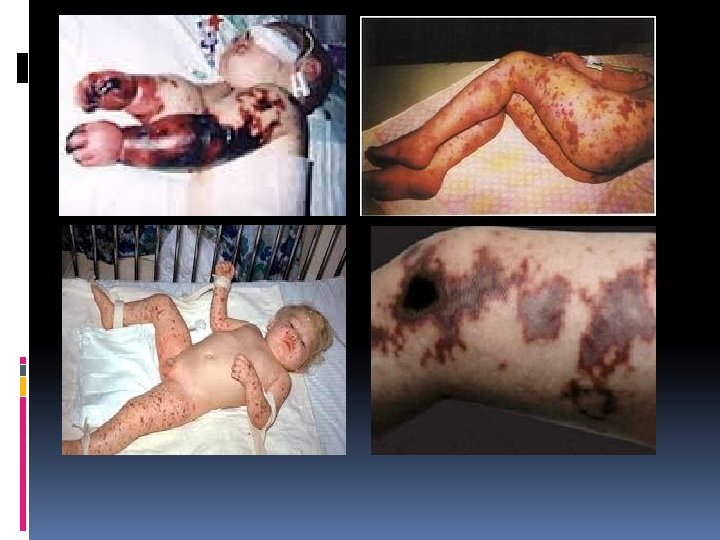
Acute meningococcemia: Acute meningococcemia may initially mimic illnesses caused by viruses or other bacteria, causing pharyngitis, fever, myalgias, weakness, vomiting, diarrhea, and/or headache. A fine maculopapular rash is evident in about 7% of cases, with onset typically early in the course of infection. Limb pain, myalgias, or refusal to walk occurs often and is the primary complaint in 7% of otherwise clinically unsuspected cases. Cold hands or feet and abnormal skin color are also early signs. In fulminant meningococcemia, the disease progresses rapidly over several hours from fever without other signs to septic shock characterized by prominent petechiae and purpura (purpura fulminans), hypotension, DIC, acidosis, adrenal hemorrhage, renal failure, myocardial failure, and coma. Meningitis may or may not be present.
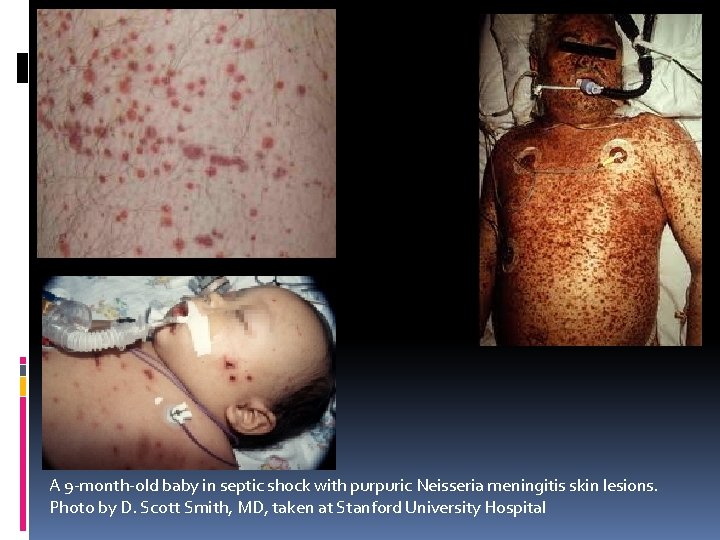
A 9 -month-old baby in septic shock with purpuric Neisseria meningitis skin lesions. Photo by D. Scott Smith, MD, taken at Stanford University Hospital

Meningococcal meningitis: Meningococcal meningitis is indistinguishable from meningitis due to other bacteria. Headache, photophobia, lethargy, vomiting, nuchal rigidity, and other signs of meningeal irritation are typically present. Seizures and focal neurologic signs occur less frequently than in patients with meningitis due to Streptococcus pneumoniae or Haemophilus influenzae type b. A meningoencephalitis-like picture can occur that may be associated with rapidly progressive cerebral edema, which may be more common with capsular group A infection.
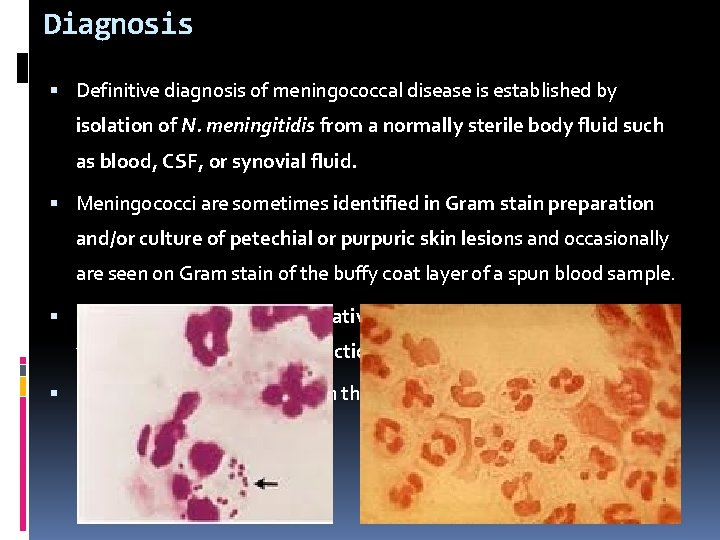
Diagnosis Definitive diagnosis of meningococcal disease is established by isolation of N. meningitidis from a normally sterile body fluid such as blood, CSF, or synovial fluid. Meningococci are sometimes identified in Gram stain preparation and/or culture of petechial or purpuric skin lesions and occasionally are seen on Gram stain of the buffy coat layer of a spun blood sample. Culture results often are negative if the patient has been treated with antibiotics prior to collection of the culture specimen. Isolation of the organism from the nasopharynx is not diagnostic for invasive disease.

In patients with meningococcal meningitis, the cellular and chemical characteristics of the CSF are those of acute bacterial meningitis, showing gram-negative diplococci on Gram stain in 75% of cases. CSF culture results may be positive in patients with meningococcemia in the absence of CSF pleocytosis or clinical evidence of meningitis; conversely, positive CSF specimens that are positive for Gram stain are sometimes culture negative. Over-decolorized pneumococci in Gram stain preparations can be mistaken for meningococci, and, therefore, empirical therapy should not be narrowed to N. meningitidis infection on the basis of Gram stain findings alone.

Detection of capsular polysaccharide antigens by rapid latex agglutination tests in CSF can support the diagnosis in cases clinically consistent with meningococcal disease and is found in 53 -90% of cases. Because false-positive results are reported and results do not significantly affect clinical practice, latex agglutination tests are not routinely recommended. These tests are most useful when their results are positive in the setting of partially treated infections in which Gram stain and culture results are negative. On the other hand, latex agglutination tests of serum or urine are not clinically useful. Capsular antigen tests are not reliable for group B strains because of cross reactions with other bacterial species (Escherichia coli K 1 antigen). PCR-based assays for detection of meningococci in blood and CSF have been developed, and multiplex PCR assays that detect several bacterial species associated with meningitis, including the meningococcus, are under development.

Other laboratory findings include leukocytopenia or leukocytosis, often with increased percentages of neutrophils and band forms, thrombocytopenia, proteinuria, and hematuria. Elevations of erythrocyte sedimentation rate (ESR) and C-reactive protein, hypoalbuminemia, hypocalcemia, and metabolic acidosis, often with increased lactate levels, are common. Patients with DIC have decreased serum concentrations of prothrombin and fibrinogen and prolonged coagulation times.

Differential Diagnosis Meningococcal disease can appear similar to sepsis or meningitis caused by many other gram-negative bacteria, S. pneumoniae, Staphylococcus aureus, or group A streptococcus; to Rocky Mountain spotted fever, ehrlichiosis, or epidemic typhus; and to bacterial endocarditis. Viral and other infectious etiologies of meningoencephalitis should be considered in some cases. Autoimmune vasculitides (especially Henoch-Schönlein purpura), serum sickness, hemolytic-uremic syndrome, Kawasaki disease, idiopathic thrombocytopenic purpura, drug eruptions, and ingestion of various poisons can have features that overlap with those of meningococcal infection.

Benign petechial rashes are common in viral and group A streptococcal infections. The nonpetechial, blanching maculopapular rash observed in some cases of meningococcal disease may initially be confused with a viral exanthem.

Treatment Empirical therapy should be initiated immediately for possible invasive meningococcal infections. β-Lactam antibiotics are the drugs of choice. Because of concerns about penicillin- or cephalosporin-resistant S. pneumoniae, intravenous (IV) vancomycin (60 mg/kg/day, divided in four doses, each dose given every 6 hr) should be added empirically as a second drug as part of initial empiric regimens for bacterial meningitis of unknown cause. More specific therapy for meningococcal disease may be initiated when culture and antibiotic susceptibility results become available. Although ciprofloxacin may be an alternative to cephalosporins for treatment of meningococcal infection, ciprofloxacin-resistant meningococci have been identified. Therapy in children is generally continued for 5 -7 days.
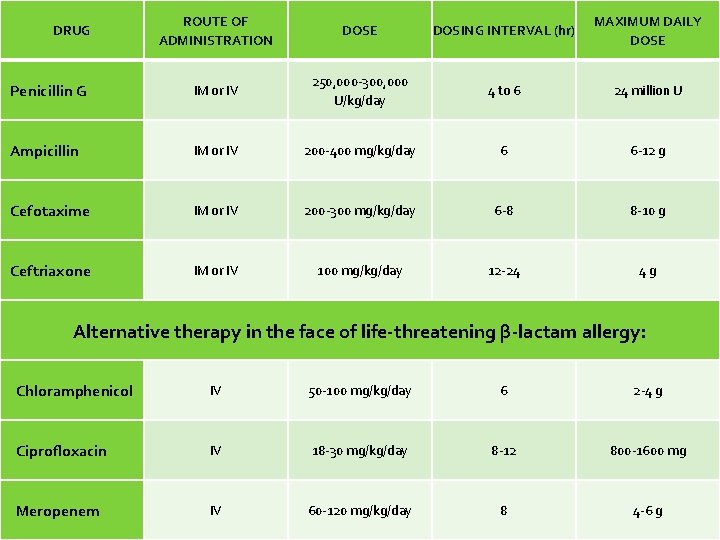
ROUTE OF ADMINISTRATION DOSE DOSING INTERVAL (hr) MAXIMUM DAILY DOSE Penicillin G IM or IV 250, 000 -300, 000 U/kg/day 4 to 6 24 million U Ampicillin IM or IV 200 -400 mg/kg/day 6 6 -12 g Cefotaxime IM or IV 200 -300 mg/kg/day 6 -8 8 -10 g Ceftriaxone IM or IV 100 mg/kg/day 12 -24 4 g DRUG Alternative therapy in the face of life-threatening β-lactam allergy: Chloramphenicol IV 50 -100 mg/kg/day 6 2 -4 g Ciprofloxacin IV 18 -30 mg/kg/day 8 -12 800 -1600 mg Meropenem IV 60 -120 mg/kg/day 8 4 -6 g

Early treatment of meningococcal infections may prevent serious sequelae, but timely early diagnosis is often difficult in the absence of petechial or purpuric skin findings. High fever and leukocytosis with increased neutrophil and band counts are common in older children and adolescents with otherwise unsuspected meningococcal infection. Empiric outpatient treatment with careful follow-up of selected patients during meningococcal outbreaks and of nontoxic children with petechial rashes can be considered after blood culture specimens are obtained. Most of the latter do not have meningococcal infection.

Supportive care Optimal supportive care is essential. Many adjunctive therapies have been attempted, but to date, none has shown clear benefit in children. Dexamethasone therapy for 2 to 4 days, with the first dose given before or during the initiation of antibiotic therapy, has decreased mortality in adults with S. pneumoniae meningitis; the benefit in patients with meningococcal meningitis has not been firmly established. Anticoagulant or fibrinolytic agents and vasodilators have been used with variable success in anecdotal reports. Activated protein C therapy is not recommended for infants with severe sepsis and purpura fulminans because of the increased risk of intracranial hemorrhage associated with its use.

Most children with meningococcal disease who do not require intubation or vasopressor support show ready response to antibiotics plus supportive care and demonstrate clinical improvement within 24 -72 hr. Those requiring mechanical ventilation and other critical care interventions often have prolonged and complicated courses that may require hospitalization for weeks. Children with severe disease who show poor response to aggressive fluid and inotropic therapies may have adrenal insufficiency and may benefit from hydrocortisone supplementation. Extracorporeal membrane oxygenation, plasmapheresis, and hyperbaric oxygen have been described anecdotally as having limited success.

Complications Acute complications of severe meningococcal disease are related to the vasculitis, DIC, and hypotension. Focal skin infarctions usually heal but can become secondarily infected, resulting in significant scarring and requiring skin grafting. The dry gangrene of extremities often seen with purpura fulminans may necessitate amputations. Adrenal hemorrhage, endophthalmitis, arthritis, endocarditis, pericarditis, myocarditis, pneumonia, lung abscess, peritonitis, and renal infarcts can occur during acute infection. Avascular necrosis of epiphyses and epiphyseal-metaphyseal defects can result from the generalized DIC and may lead to growth disturbances and late skeletal deformities. Deafness is the most frequent neurologic sequela of meningitis, occurring in 5 -10% of children. Cerebral arterial or venous thrombosis with resultant cerebral infarction can occur in severe cases. Meningococcal meningitis is rarely complicated by subdural effusion or empyema or by brain abscess. Other rare neurologic sequelae include ataxia, seizures, blindness, cranial nerve palsies, hemiparesis or quadriparesis, and obstructive hydrocephalus. The last often manifests 3 -4 wk after onset of illness.
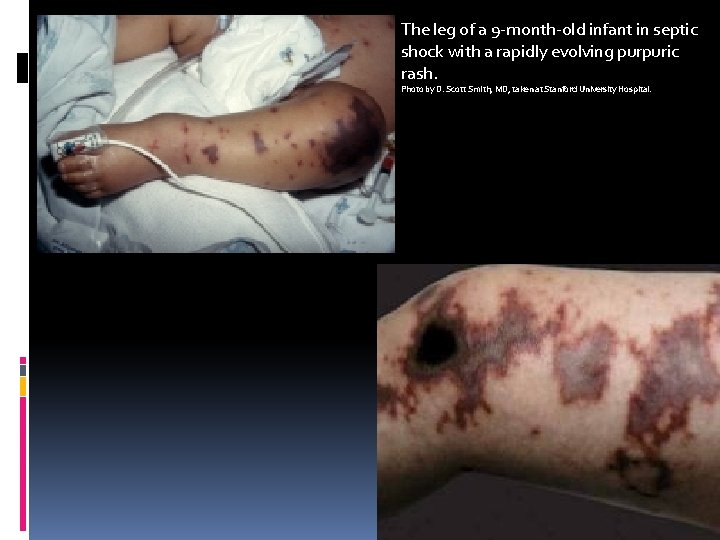
The leg of a 9 -month-old infant in septic shock with a rapidly evolving purpuric rash. Photo by D. Scott Smith, MD, taken at Stanford University Hospital.

Nonsuppurative complications of meningococcal disease appear to be immune complex mediated and become apparent 4 -9 days after the onset of illness. Arthritis and cutaneous vasculitis (erythema nodosum) are most common. The arthritis usually is monoarticular or oligoarticular, involves large joints, and is associated with sterile effusions that respond to nonsteroidal antiinflammatory agents. Long-term sequelae are uncommon. Because most patients with meningococcal meningitis become afebrile by the 7 th hospital day, persistence or recrudescence of fever after 5 days of antibiotics warrants evaluation for immune complex–mediated complications. Reactivation of latent herpes simplex virus infections (primarily herpes labialis) is common during meningococcal infection.

Prognosis The mortality rate for invasive meningococcal disease remains about 10% in the USA despite modern medical interventions. Most deaths occur within 48 hr of hospitalization in children with meningococcemia. Poor prognostic factors on presentation include hypothermia or extreme hyperpyrexia, hypotension or shock, purpura fulminans, seizures, leukopenia, thrombocytopenia (including DIC), acidosis, and high circulating levels of endotoxin and TNF-α. The presence of petechiae for <12 hr before admission, absence of meningitis, and low or normal ESR indicate rapid, fulminant progression and poorer prognosis.

Screening for complement deficiency after resolution of the acute infection should be performed in older children, adolescents, and adults with meningococcal infection and in young children with recurrent infection or with first episodes caused by strains with unusual capsular groups such as W-135 or X.

Prevention Close contacts of patients with meningococcal disease are at increased risk for infection. Antibiotic prophylaxis is indicated for household, daycare, and nursery school contacts and for anyone who has had contact with the patient's oral secretions during the 7 days before onset of illness. Prophylaxis of contacts should be offered as soon as possible, ideally within 24 hr of diagnosis of the patient. Because prophylaxis is not 100% effective, close contacts should be carefully monitored and brought to medical attention if they experience fever. Prophylaxis is not routinely recommended for medical personnel except those with intimate exposure, such as through mouth-to-mouth resuscitation, intubation, or suctioning before antibiotic therapy was begun.

Neither penicillin nor ampicillin treatment eradicates nasopharyngeal carriage; patients with meningococcal infection treated with penicillin or ampicillin should receive prophylaxis before hospital discharge. Droplet precautions should be observed for hospitalized patients for 24 hr after initiation of effective therapy. All confirmed or probable cases of meningococcal infection must be reported to the local public health department.

Vaccination As of October 2010, three quadrivalent meningococcal vaccines containing capsular groups A, C, W-135, and Y are licensed in the USA. MPSV 4 (Menomune, Sanfi Pasteur) contains only purified polysaccharides. The other two are conjugates of either purified polysaccharides coupled with diphtheria toxoid (MCV 4 -DT, Menactra, Sanofi Pasteur) All vaccines are safe and effective. The conjugate vaccines more frequently cause transient fever and local redness, pain, or swelling at the injection site than MPSV 4, which is attributed to the presence of the carrier proteins in the conjugate vaccines. Following a single dose in otherwise healthy adolescents, all vaccines elicit serum bactericidal antibody titers that peak at about 4 -6 wk. The conjugate vaccines also prime for immunologic memory, which results in booster responses to a second injection. Vaccination also has the potential to decrease meningococcal carriage. For all of these reasons, use of either conjugate vaccine is preferred over MPSV 4. > 9 month, two doses 8 weeks apart.

Bibliography Allport T, Read L, Nadel S, et al: Critical illness and amputation in meningococcal septicemia: is life worth saving? . Pediatrics 2008; 122: 629 -632. Centers for Disease Control and Prevention : Licensure of a meningococcal conjugate vaccine (Menveo) and guidance for use—Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Morbid Mortal Wkly Rep 2010; 59: 273. Centers for Disease Control and Prevention : Updated recommendation from the Advisory Committee on Immunization Practices (ACIP) for revaccination of persons at prolonged increased risk for meningococcal disease. MMWR Morbid Mortal Wkly Rep 2009; 58: 1042 -1043. Centers for Disease Control and Prevention : Report from the Advisory Committee on Immunization Practices (ACIP): decision not to recommend routine vaccination of all children aged 2 – 10 years with quadrivalent meningococcal conjugate vaccine (MCV 4). MMWR Morbid Mortal Wkly Rep 2008; 57: 462 -465. Centers for Disease Control and Prevention : Revised recommendations of the Advisory Committee on Immunization Practices to vaccinate all persons aged 11– 18 years with meningococcal conjugate vaccine. MMWR Morbid Mortal Wkly Rep 2007; 56: 794 -795. Centers for Disease Control and Prevention : Updated recommendation from the Advisory Committee on Immunization Practices (ACIP) for revaccination of persons at prolonged increased risk for meningococcal disease. MMWR Morb Mortal Wkly Rep 2009; 58: 1042 -1043. Centers for Disease Control and Prevention : Licensure of a meningococcal conjugate vaccine (Menveo) and guidance for use—Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Morb Mortal Wkly Rep 2009; 59: 273. Cohn AC, Mac. Neil JR, Harrison LH, et al: Changes in Neisseria meningitidis disease epidemiology in the United States, 1998– 2007: implications for prevention of meningococcal disease. Clin Infect Dis 2010; 50: 184 -193. Davila S, Wright VJ, Chuen Khor C, et al: Genome-wide association study identifies variants in the CFH region associated with host susceptibility to meningococcal disease. Nat Genet 2010; 42: 772 -776. Gardner P: Clinical practice: prevention of meningococcal disease. N Engl J Med 2006; 355: 1466 -1473. Granoff DM: Relative importance of complement-mediated serum bactericidal and opsonic activity for protection against meningococcal disease. Vaccine 2009; 27(Suppl 2): B 117 -B 125. Granoff DM, Harrison L, Borrow R: Meningococcal vaccines. In: Plotkin SA, Offit P, Orenstein WA, ed. Vaccines, ed 5. Philadelphia: Saunders Elsevier; 2008: 399 -434. Harrison LH, Kreiner CJ, Shutt KA, et al: Risk factors for meningococcal disease in student in grades 9– 12. Pediatr Infect Dis J 2008; 27: 193 -199. Hart CA, Thomson AP: Meningococcal disease and its management in children. BMJ 2006; 333: 685 -690. Heckenberg SGB, de Gans J, Brouwer MC, et al: Clinical features, outcome, and meningococcal genotype in 258 adults with meningococcal meningitis. Medicine 2008; 87: 185 -192. Maiden MC, Ibarz-Pavon AB, Urwin R, et al: Impact of meningococcal serogroup C conjugate vaccines on carriage and herd immunity. J Infect Dis 2008; 197: 737 -743. Nascimento-Carvalho CM, Moreno-Carvalho OA: Changing the diagnostic framework of meningococcal disease. Lancet 2006; 367: 371 -372. Snape MD, Perrett KP, Ford KJ, et al: Immunogenicity of a tetravalent meningococcal glycoconjugate vaccine in infants. JAMA 2008; 299: 173 -184. Thompson MJ, Ninis N, Perera R, et al: Clinical recognition of meningococcal disease in children and adolescents. Lancet 2006; 367: 397 -403. Vu DM, Welsch JA, Zuno-Mitchell P, et al: Antibody persistence 3 years after immunization of adolescents with quadrivalent meningococcal conjugate vaccine. J Infect Dis 2006; 193: 821828. Wu HM, Harcourt BH, Hatcher CP, et al: Emergence of ciprofloxacin-resistant Neisseria meningitidis in North America. N Engl J Med 2009; 360: 886 -892. Wright V, Hibber M, Levin M: Genetic polymorphisms in host response to meningococcal infection: the role of susceptibility and severity genes. Vaccine 2009; 27(Suppl 2): B 90 -B 102. Copyright © 2013 Elsevier Inc. All rights reserved. Read our Terms and Conditions of Use and our Privacy Policy. For problems or suggestions concerning this service, please contact: online. help@elsevier. com
- Slides: 70