Separation of polar betalain pigments from cacti fruits

Separation of polar betalain pigments from cacti fruits of Hylocereus polyrhizus by ion-pair HSCCC Sławomir Wybranieca, ∗, Paweł Stalica a, Gerold Jerz b, Bettina Kloseb, Nadine Gebersb, Peter Winterhalterb, Aneta Spornaa, Maciej Szaleniecc, Yosef Mizrahi a Faculty of Analytical Chemistry, Institute C-1, Department of Chemical Engineering and Technology, Cracow University of Technology, ul. Warszawska 24, 31 -155 Cracow, Poland Journal of Chromatography A Accepted 13 August 2009 Presenter : Min. Seok Kang ( 2010 1 30 )
Contents Ion-pair method Introduction Experiments /Result Further study

Ion-pair method on Chromatography (Focus on RP - HPLC)
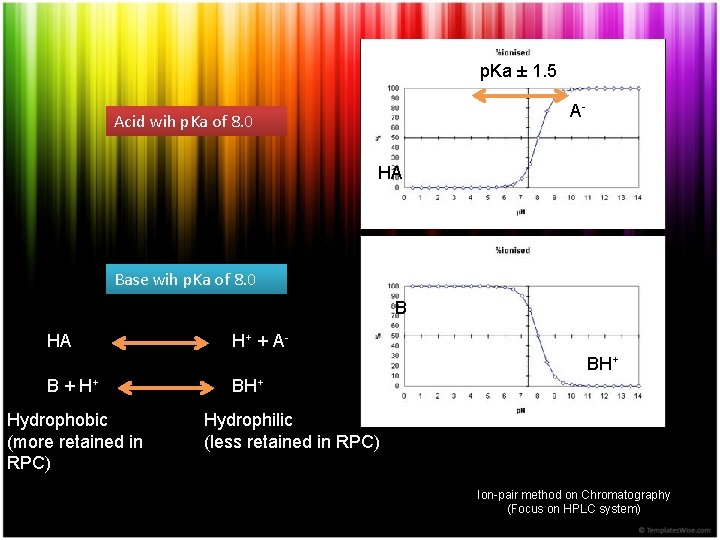
p. Ka ± 1. 5 A- Acid wih p. Ka of 8. 0 HA Base wih p. Ka of 8. 0 B HA H+ + A BH+ B + H+ Hydrophobic (more retained in RPC) BH+ Hydrophilic (less retained in RPC) Ion-pair method on Chromatography (Focus on HPLC system)
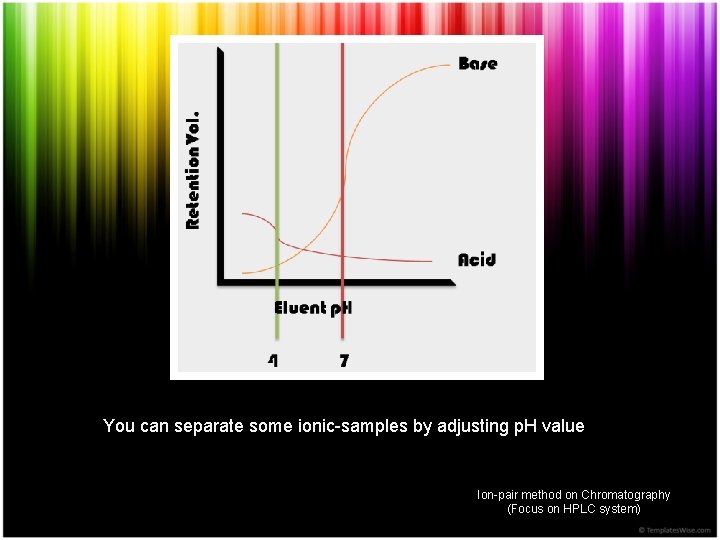
You can separate some ionic-samples by adjusting p. H value Ion-pair method on Chromatography (Focus on HPLC system)
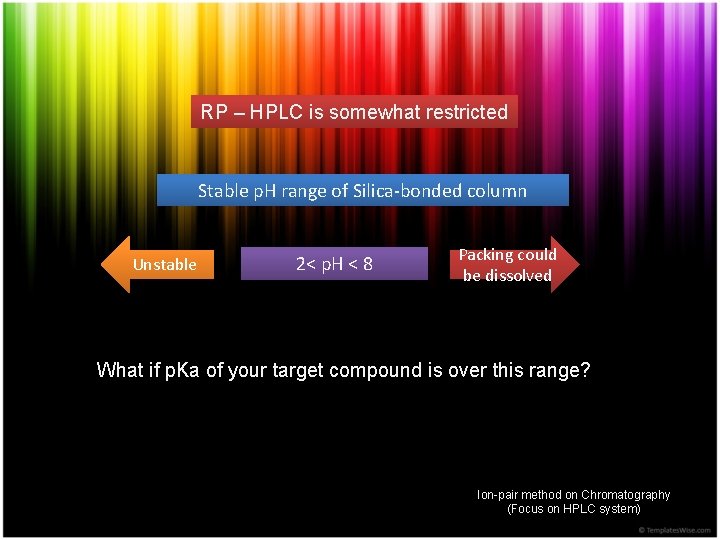
RP – HPLC is somewhat restricted Stable p. H range of Silica-bonded column Unstable 2< p. H < 8 Packing could be dissolved What if p. Ka of your target compound is over this range? Ion-pair method on Chromatography (Focus on HPLC system)
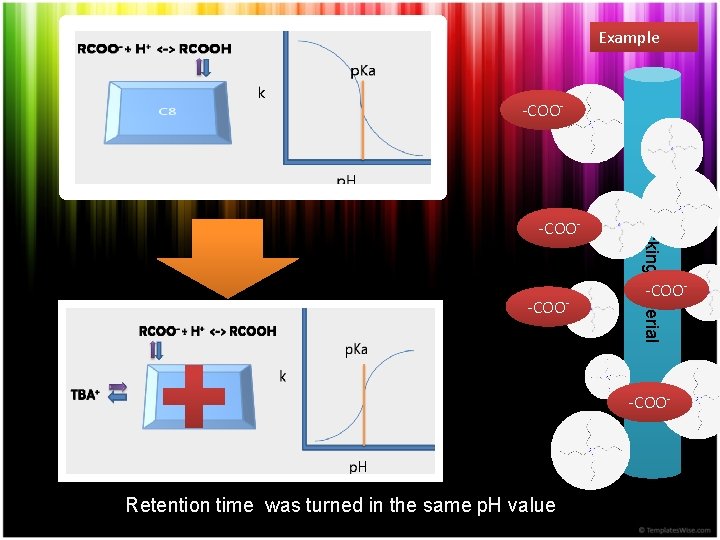
Example -COO- Packing Material -COO- Retention time was turned in the same p. H value

Ion-pair reagent Anionic ion-pair reagent Sodium decyl sulfate Sodium 1 -tetradecanesulfonate Sodium 1 -hexanesulfonate SDS Perfluorocarboxylic acid (TFA , HFBA…) Cationic ion-pair reagent Tetraethylammonium Tetrahexylammonium Tetrabutylammonium THA-Br Hexadecyltrimethylammonium Ion-pair method on Chromatography (Focus on HPLC system)

So, What happens if IPC-reagent was introduced in HSCCC?

Hylocereus polyrhizus (Cactaceae-Cacoideae) , 火龍果 Pitaya (spanish) Dragon fruit (English) Activity : anti-oxidant Hylocereus undatus Hylocereus megalanthus (Yellow pitaya)
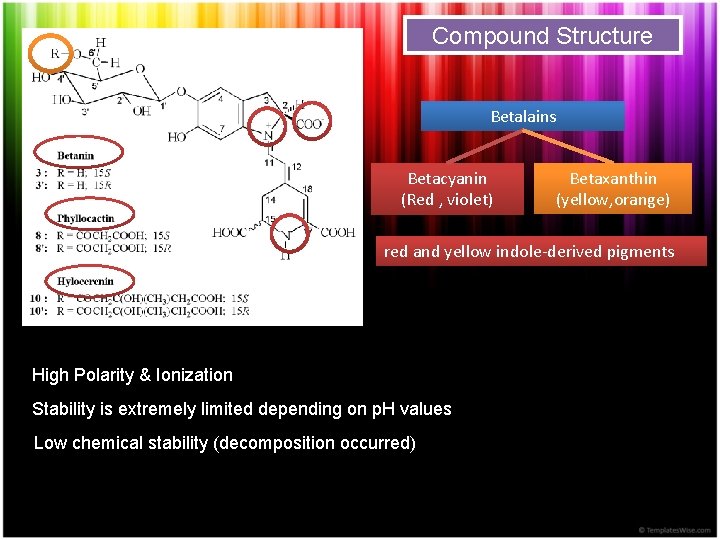
Compound Structure Betalains Betacyanin (Red , violet) Betaxanthin (yellow, orange) red and yellow indole-derived pigments High Polarity & Ionization Stability is extremely limited depending on p. H values Low chemical stability (decomposition occurred)
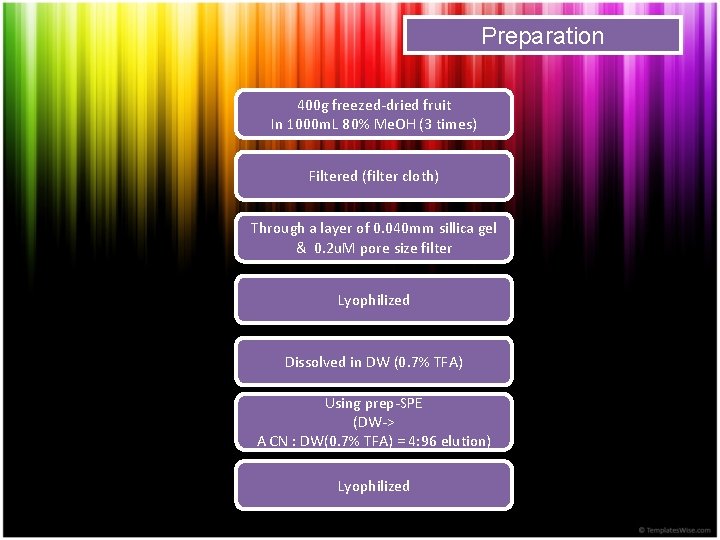
Preparation 400 g freezed-dried fruit In 1000 m. L 80% Me. OH (3 times) Filtered (filter cloth) Through a layer of 0. 040 mm sillica gel & 0. 2 u. M pore size filter Lyophilized Dissolved in DW (0. 7% TFA) Using prep-SPE (DW-> A CN : DW(0. 7% TFA) = 4: 96 elution) Lyophilized

HSCCC Solvent system Previous Method to separate some betacyanins from Phytolacca americana. (But, It didn’t work on this experiment !! ) Upper (Stationary phase) Lower (Mobile phase) Shifting Kd value Betalains + Ion-pair agents = Increased hydrophobicity
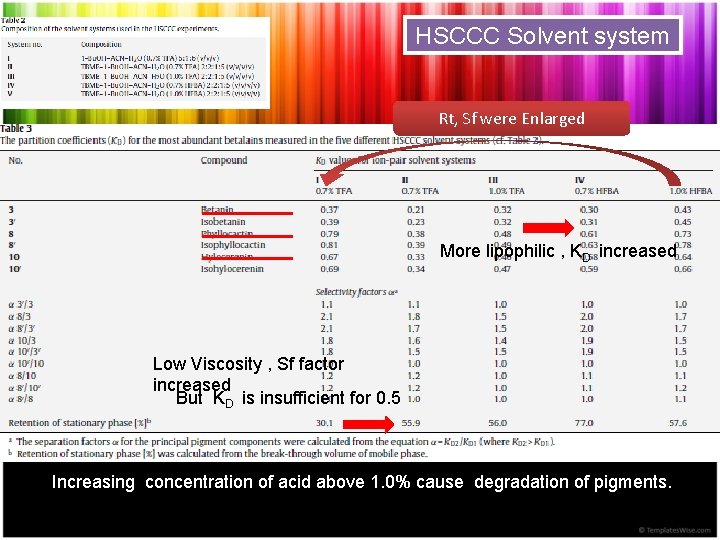
HSCCC Solvent system Rt, Sf were Enlarged More lipophilic , KD increased Low Viscosity , Sf factor increased But KD is insufficient for 0. 5 Increasing concentration of acid above 1. 0% cause degradation of pigments.

HSCCC procedure Pharma-Tech Research model CCC 1000 Pump Upper organic phase(Stationary) into Coil Rotation speed at 1000 rpm 850 -900 mg sample was dissolved in 10 m. L each of phase Total volume : 850 m. L (20 m. L sample loop) Head to tail mode Upper (Stationary phase) Lower (Mobile phase) Injection before Equlibrium Mobile phase : 3. 0 m. L After eluants had decreased, EECCC started (flow 5. 0 m. L , 800 rpm) Effluent stream was monitored by UV-absorbance at 540 nm
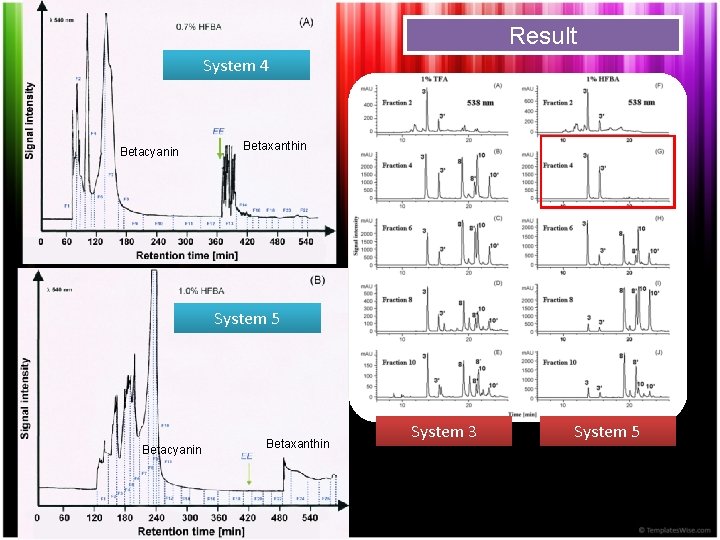
Result System 4 Betacyanin Betaxanthin System 5 Betacyanin Betaxanthin System 3 System 5
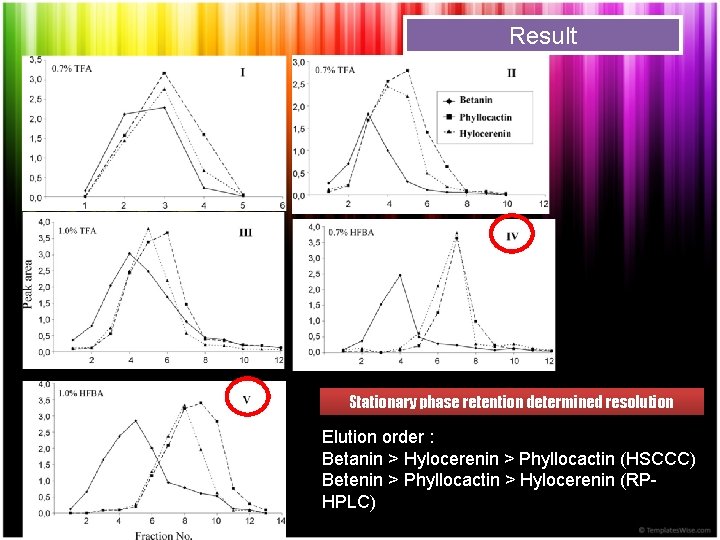
Result Stationary phase retention determined resolution Elution order : Betanin > Hylocerenin > Phyllocactin (HSCCC) Betenin > Phyllocactin > Hylocerenin (RPHPLC)

Conclusion Upper (Stationary phase) Lower (Mobile phase) Shifting Kd value Betalains + Ion-pair agents = Increased hydrophobicity 1. In case of highly water soluble compound , patition will occurred increasing hydrophobicity by using ion-pair reagent 2. Stationary phase retention is important part of resolution 3. EECCC approach is able to recover all pigment compound of H. polyrhizus ( all polarity scale )
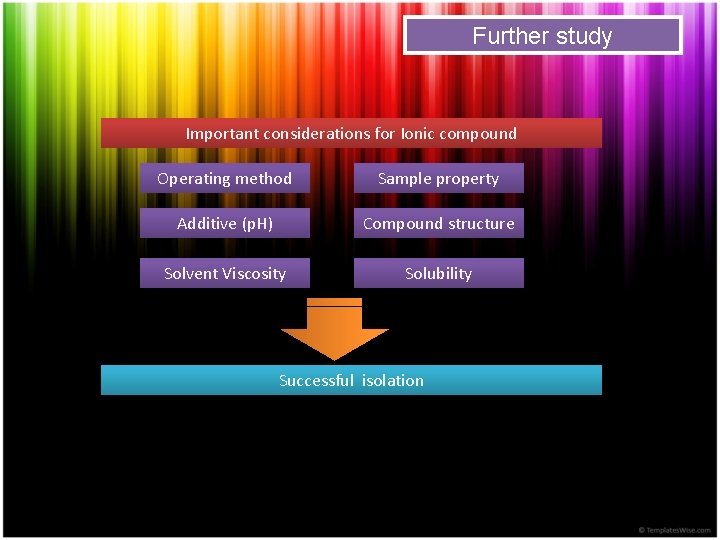
Further study Important considerations for Ionic compound Operating method Sample property Additive (p. H) Compound structure Solvent Viscosity Solubility Successful isolation

Thank you for your attention.
- Slides: 20