SEPARATION COLUMNS DISTILLATION AND ABSORPTION Dr Kh Nasrifar

SEPARATION COLUMNS (DISTILLATION AND ABSORPTION) Dr. Kh. Nasrifar Department of Chemical and Petrochemical Engineering

Batch Distillation
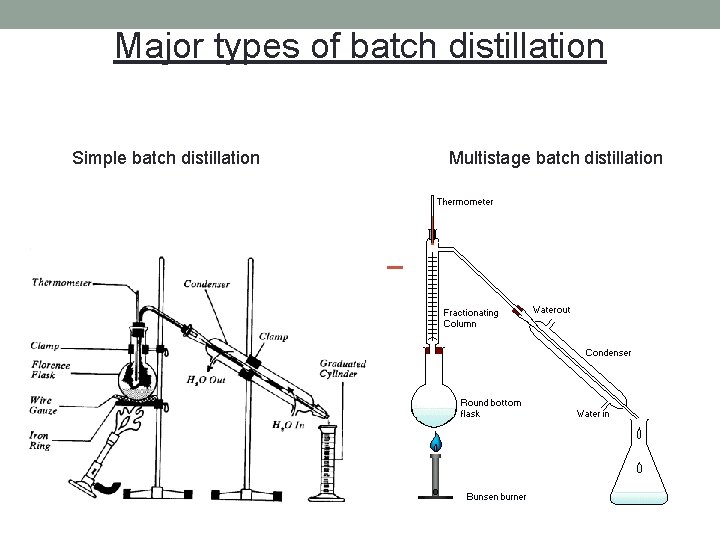
Major types of batch distillation Simple batch distillation Multistage batch distillation

Reasons to use batch distillation 1. 2. 3. 4. 5. 6. Small capacity (e. g. , specialty chemicals) Intermittent need Test run for a new product Up-stream operations are batch (e. g. , alcoholic spirits) Feed inappropriate for continuous distillation (suspended solids) Feed varies widely in composition

Simple batch distillation no rectification ( = no column) Characteristics: - no column; a single equilibrium stage - V, y D, x. D time 0: F, x. F time t: W, x. W - still pot with heater - (= the still pot) single charge (F) to still pot at time = 0 vapor is withdrawn continuously composition of liquid in still pot (x. W) changes continuously composition of liquid distillate (x. D) changes continuously

Rayleigh equation TMB: F = Wfinal + Dtotal CMB: Fx. F = Wfinalx. W, final + Dtotalx. D, avg V, y D, x. D time 0: F, x. F time t: W, x. W Specify F, x. F and x. W, final or x. D, avg Leaves 3 unknowns: Wfinal, Dtotal and x. W, final or x. D, avg Need one more equation: y = x. D still pot, with heater d. CMB: - x. Dd. W = - d(Wx. W) (vapor withdrawn) = (change in still pot composition) WAIT! K is not constant; K = K(T) chain rule: Rayleigh equation - x. Dd. W = - Wdx. W - x. Wd. W where x. D = f(x. W)
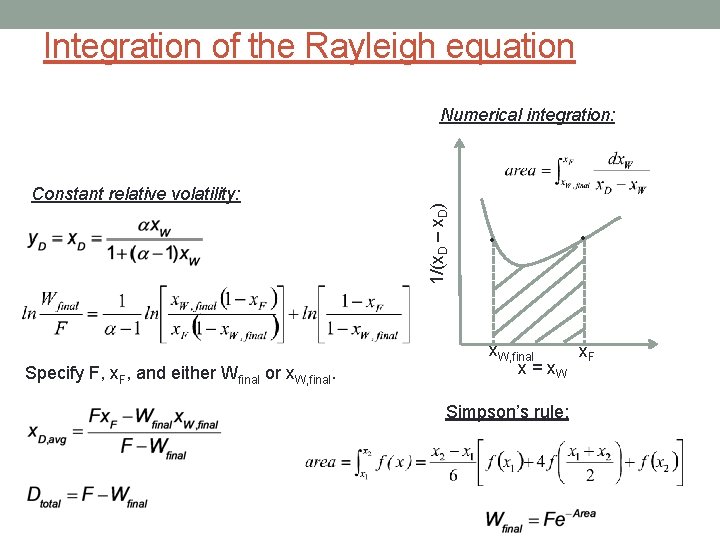
Integration of the Rayleigh equation Constant relative volatility: Specify F, x. F, and either Wfinal or x. W, final. 1/(x. D – x. D) Numerical integration: • • x. W, final x. F x = x. W Simpson’s rule:

Batch steam distillation Used for thermally fragile organics (e. g. , essential oils in perfume industry), and for slurries/sludges containing organics. If W, D are immiscible with water, we have a heterogeneous azeotrope. D. o. F. = 2 components – 3 phases + 2 = 1 Fix Ptotal, then T cannot vary! constant T < Tbp(H 2 O) Both H 2 O and organic vaporize well below their single-component boiling points. Also, constant vapor composition. Raoult’s Law: Ptotal = P*Wx. W + P*H 2 O V, y still pot, no heater A single charge (F) added to still pot at time = 0. Steam is added continuously. D, x. D = 1 W, x. W H 2 O(l) (to waste) steam How much steam is required? Steam also needed to heat and vaporize the material in the still pot.

Batch distillation with rectification y 1 = x 0 = x. D TMB: Vj+1 = Lj + D CMB: Vj+1 yj+1 = Ljxj + Dx. D • both are time-dependent • either D or x. D (or both) change over the course of the distillation y 1 ≠ K / x. W CMO: Vj+1 = Vj and V 1, y 1 stage j Vj+1, yj+1 Lj, xj stage N VN+1, x. N+1 LN , x N time 0: F, x. F time t: W, x. W still pot, with heater stage N+1 L 0 , x 0 D, x. D Lj = Lj-1 operating line equation: y = (L/V) x + (1 - (L/V)) x. D y = x. D slope = L/V • actually a family of operating lines, since L/V or x. D (or both) change over the course of the distillation • therefore the operating line moves on the M-T diagram
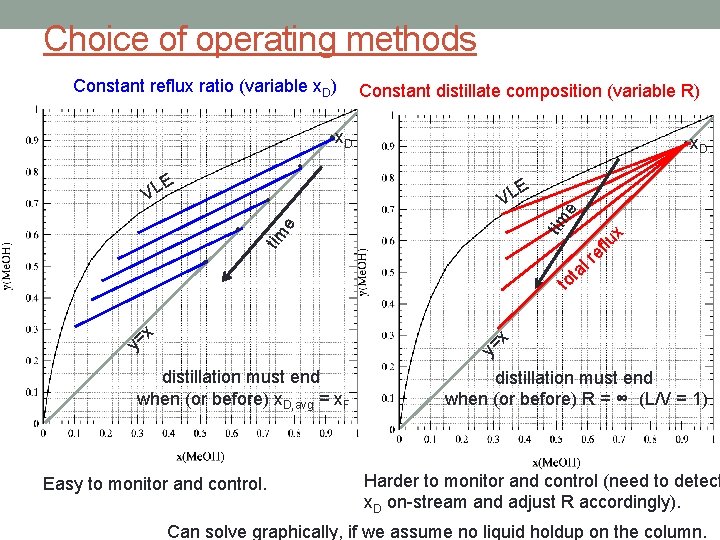
Choice of operating methods Constant reflux ratio (variable x. D) Constant distillate composition (variable R) • x. D • e tim • x flu e • x = y e E VL • tim E VL t to r l a x = y distillation must end when (or before) x. D, avg = x. F Easy to monitor and control. distillation must end when (or before) R = ∞ (L/V = 1) Harder to monitor and control (need to detect x. D on-stream and adjust R accordingly). Can solve graphically, if we assume no liquid holdup on the column.
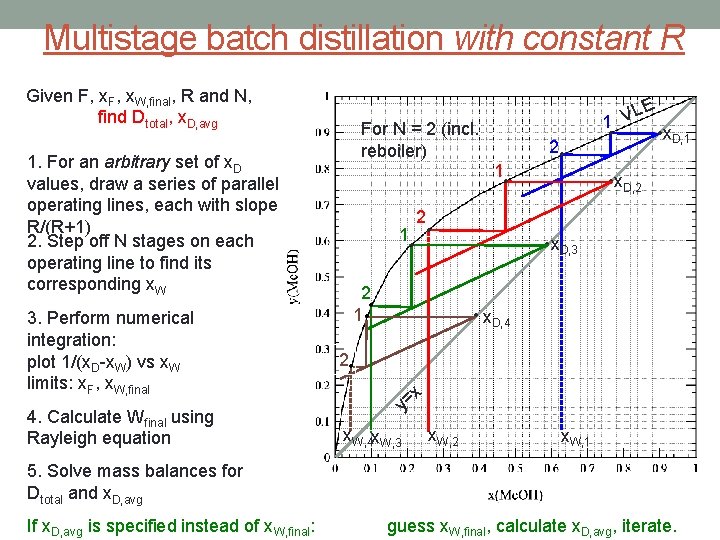
Multistage batch distillation with constant R Given F, x. W, final, R and N, find Dtotal, x. D, avg 1. For an arbitrary set of x. D values, draw a series of parallel operating lines, each with slope R/(R+1) 2. Step off N stages on each operating line to find its corresponding x. W 3. Perform numerical integration: plot 1/(x. D-x. W) vs x. W limits: x. F, x. W, final 4. Calculate Wfinal using Rayleigh equation For N = 2 (incl. reboiler) 1 • 2 1 • • 2 • 1 • 2 • LE 1 V • • x. D, 1 • x. D, 2 • x. D, 3 • x. D, 4 2 • x = y x. W, 4 x. W, 3 x. W, 2 x. W, 1 5. Solve mass balances for Dtotal and x. D, avg If x. D, avg is specified instead of x. W, final: guess x. W, final, calculate x. D, avg, iterate.

Operating time at constant R (D) depends on vapor flow rate (V), which depends on boilup rate shut down, cleaning and recharging still pot, restart • if the boilup rate is constant, then V is constant, and D will be constant condenser TMB: • V = Vmax when vapor velocity u = uflood • uflood depends on column diameter • typically, operate at D = 0. 75 Dmax

Calculating column diameter We want to use the smallest diameter that will not cause the column to flood. where σ is surface tension, ρ L and ρV are liquid and vapor densities, respectively. Csb, flood is the capacity factor, depends on flow parameter FP and tray spacing; obtain from graphical correlation. where η is the fraction of the column cross-sectional area available for vapor flow (i. e. , column cross-sectional area minus downcomer area).
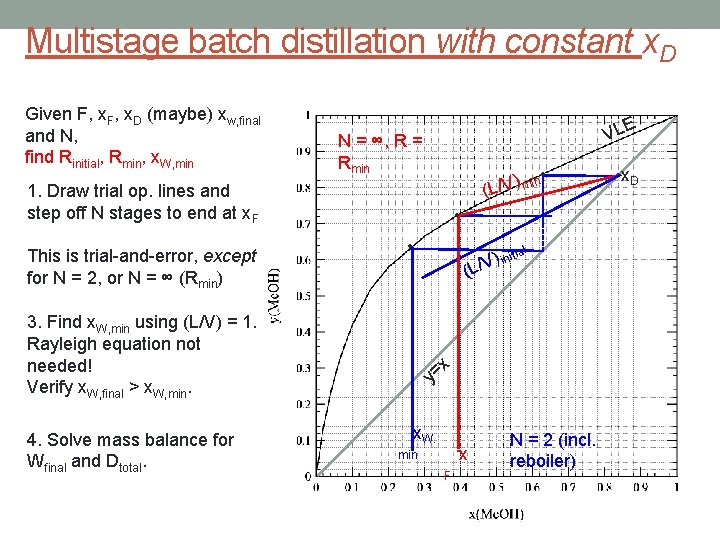
Multistage batch distillation with constant x. D Given F, x. D (maybe) xw, final and N, find Rinitial, Rmin, x. W, min VLE N = ∞, R = Rmin • n i ) m V (L/ 1. Draw trial op. lines and step off N stages to end at x. F This is trial-and-error, except for N = 2, or N = ∞ (Rmin) • • ( 3. Find x. W, min using (L/V) = 1. Rayleigh equation not needed! Verify x. W, final > x. W, min. 4. Solve mass balance for Wfinal and Dtotal. l ) initia V / L x = y x. W. x min F N = 2 (incl. reboiler) • x. D

Operating time with constant x. D mass balance: Numerical integration: • • x. W, final x. F x = x. W 1. Draw a series of arbitrary operating lines, each with a different slope L/V 2. Step off N stages on each operating line to find its corresponding x. W 3. Perform numerical integration (plot graph, use Simpson’s rule) 4. Calculate toperating

Optimal control • use optimal, time-dependent reflux ratio (not constant R, not constant x. D) • more energy-efficient • useful for difficult separations Effect of liquid holdup on the column • usually, we can assume vapor holdup is negligible • liquid holdup causes xw to be lower than it would be in the absence of holdup • causes the degree of separation to decrease To assess the effect on batch distillation: • measure the amount of holdup at total reflux • perform computational simulation
- Slides: 16