SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1
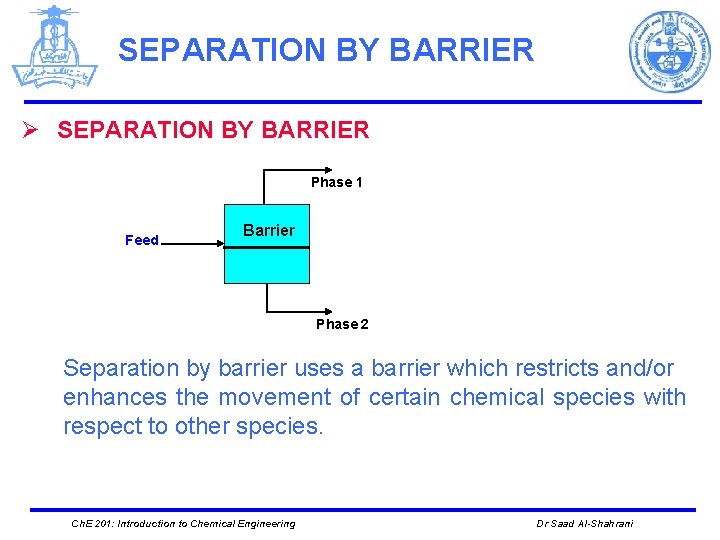
SEPARATION BY BARRIER Ø SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other species. Ch. E 201: Introduction to Chemical Engineering Dr Saad Al-Shahrani
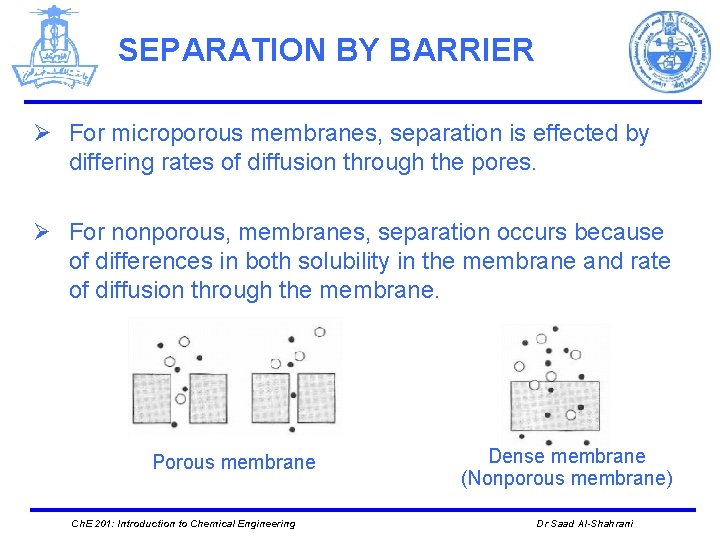
SEPARATION BY BARRIER Ø For microporous membranes, separation is effected by differing rates of diffusion through the pores. Ø For nonporous, membranes, separation occurs because of differences in both solubility in the membrane and rate of diffusion through the membrane. Porous membrane Ch. E 201: Introduction to Chemical Engineering Dense membrane (Nonporous membrane) Dr Saad Al-Shahrani
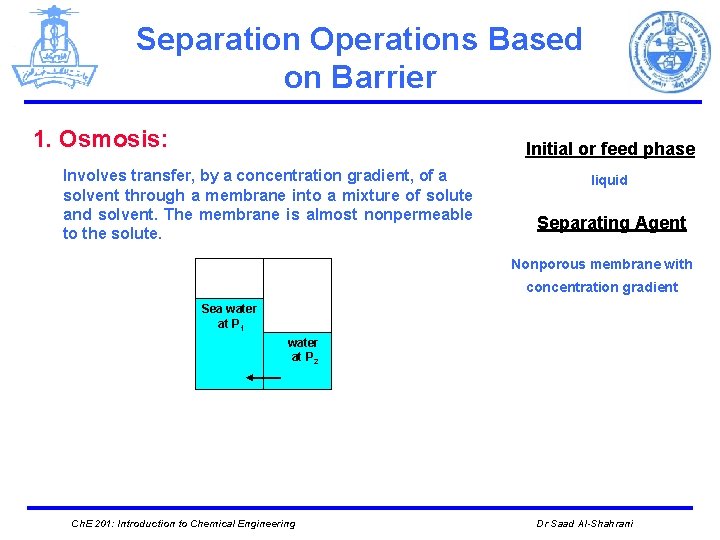
Separation Operations Based on Barrier 1. Osmosis: Initial or feed phase Involves transfer, by a concentration gradient, of a solvent through a membrane into a mixture of solute and solvent. The membrane is almost nonpermeable to the solute. liquid Separating Agent Nonporous membrane with concentration gradient Sea water at P 1 water at P 2 Ch. E 201: Introduction to Chemical Engineering Dr Saad Al-Shahrani
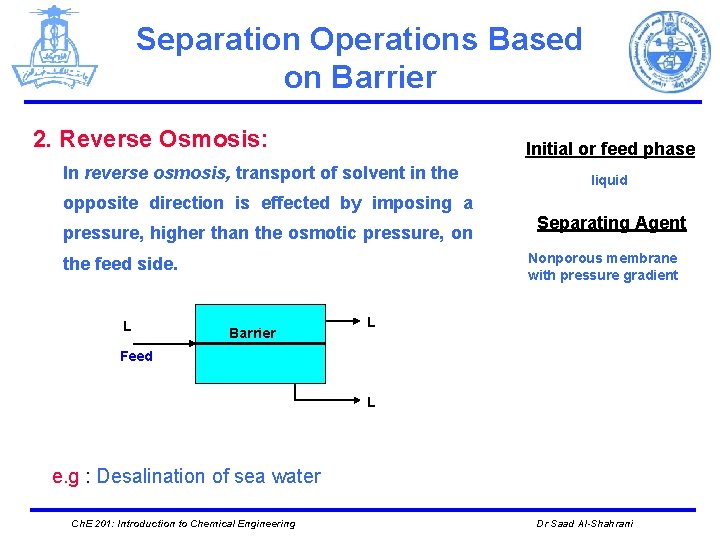
Separation Operations Based on Barrier 2. Reverse Osmosis: Initial or feed phase In reverse osmosis, transport of solvent in the opposite direction is effected by imposing a pressure, higher than the osmotic pressure, on Separating Agent Nonporous membrane with pressure gradient the feed side. L liquid Barrier L Feed L e. g : Desalination of sea water Ch. E 201: Introduction to Chemical Engineering Dr Saad Al-Shahrani
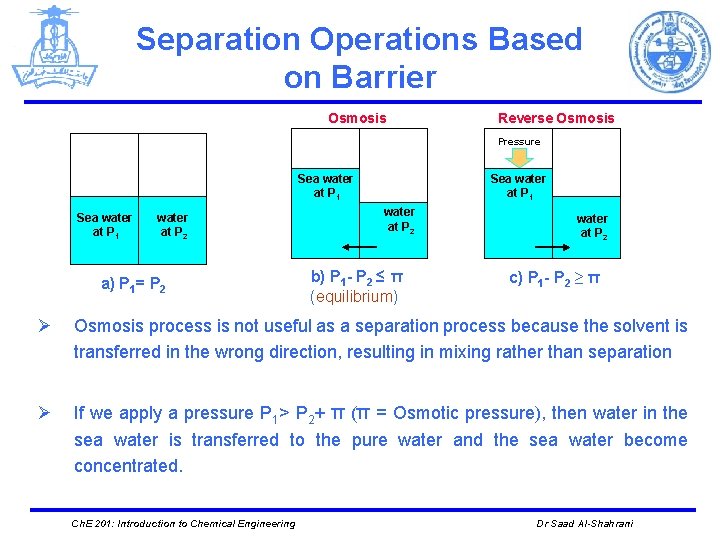
Separation Operations Based on Barrier Osmosis Reverse Osmosis Pressure Sea water at P 1 water at P 2 a) P 1= P 2 Sea water at P 1 water at P 2 b) P 1 - P 2 ≤ π (equilibrium) water at P 2 c) P 1 - P 2 ≥ π Ø Osmosis process is not useful as a separation process because the solvent is transferred in the wrong direction, resulting in mixing rather than separation Ø If we apply a pressure P 1> P 2+ π (π = Osmotic pressure), then water in the sea water is transferred to the pure water and the sea water become concentrated. Ch. E 201: Introduction to Chemical Engineering Dr Saad Al-Shahrani
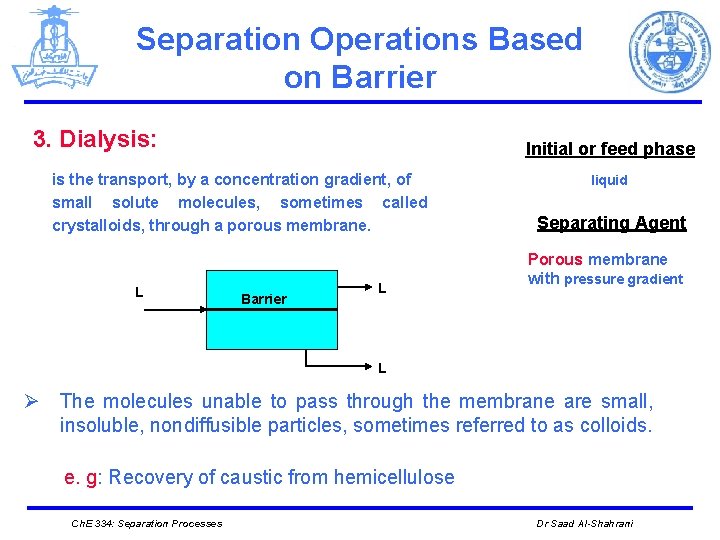
Separation Operations Based on Barrier 3. Dialysis: Initial or feed phase is the transport, by a concentration gradient, of small solute molecules, sometimes called crystalloids, through a porous membrane. L Barrier L liquid Separating Agent Porous membrane with pressure gradient L Ø The molecules unable to pass through the membrane are small, insoluble, nondiffusible particles, sometimes referred to as colloids. e. g: Recovery of caustic from hemicellulose Ch. E 334: Separation Processes Dr Saad Al-Shahrani
Separation Operations Based on Barrier dialysis Ch. E 334: Separation Processes Dr Saad Al-Shahrani
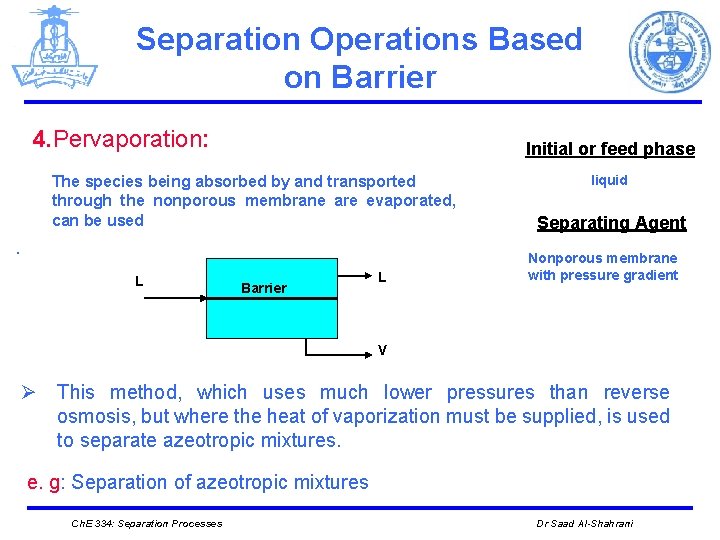
Separation Operations Based on Barrier 4. Pervaporation: Initial or feed phase The species being absorbed by and transported through the nonporous membrane are evaporated, can be used. L Barrier L liquid Separating Agent Nonporous membrane with pressure gradient V Ø This method, which uses much lower pressures than reverse osmosis, but where the heat of vaporization must be supplied, is used to separate azeotropic mixtures. e. g: Separation of azeotropic mixtures Ch. E 334: Separation Processes Dr Saad Al-Shahrani
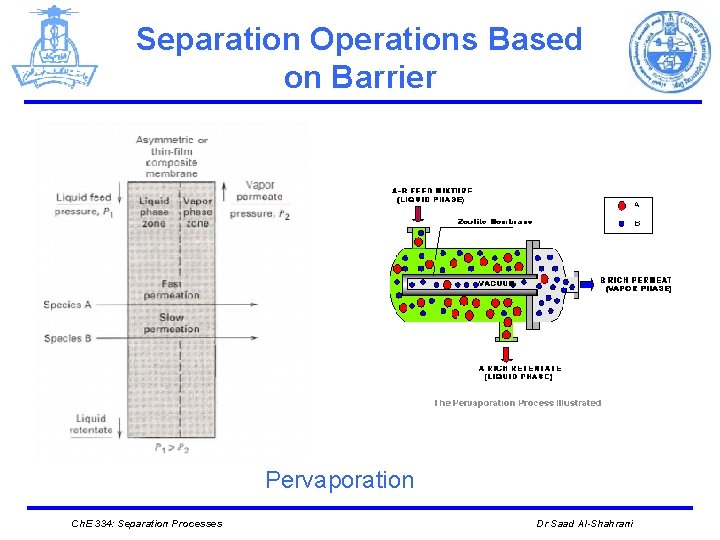
Separation Operations Based on Barrier Pervaporation Ch. E 334: Separation Processes Dr Saad Al-Shahrani
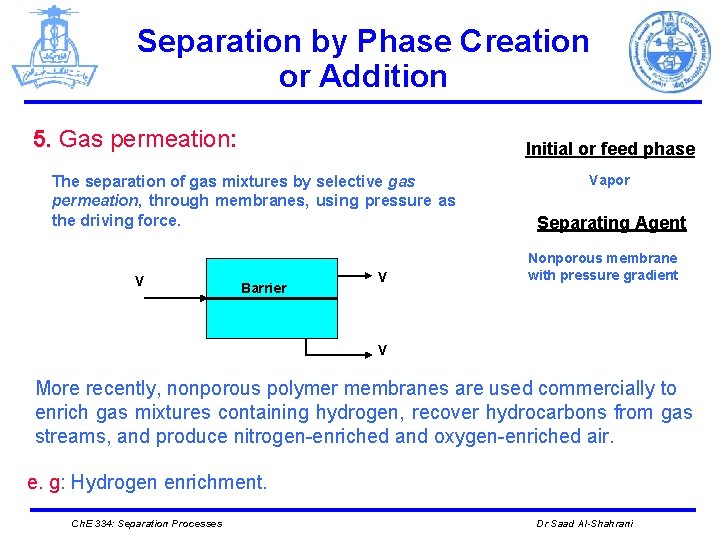
Separation by Phase Creation or Addition 5. Gas permeation: Initial or feed phase The separation of gas mixtures by selective gas permeation, through membranes, using pressure as the driving force. V Barrier V Vapor Separating Agent Nonporous membrane with pressure gradient V More recently, nonporous polymer membranes are used commercially to enrich gas mixtures containing hydrogen, recover hydrocarbons from gas streams, and produce nitrogen-enriched and oxygen-enriched air. e. g: Hydrogen enrichment. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
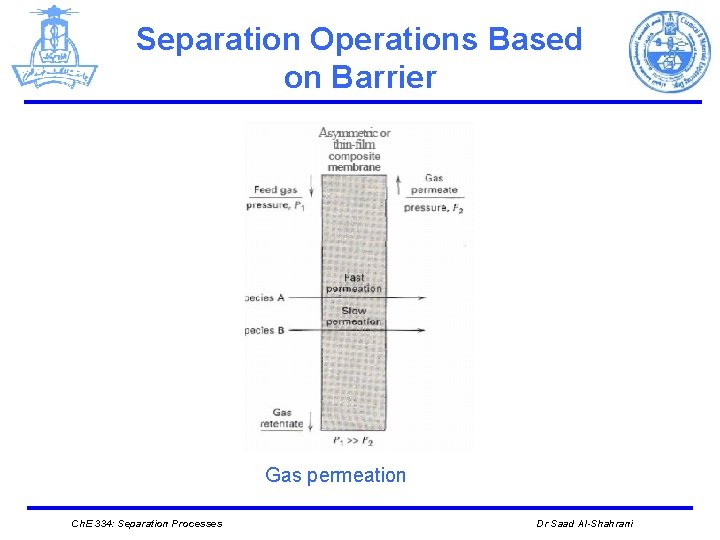
Separation Operations Based on Barrier Gas permeation Ch. E 334: Separation Processes Dr Saad Al-Shahrani

SEPARATION BY SOLID AGENT Ø Separation by solid agent use solid as mass-separating agents Ø The solid, usually in the form of a granular material or packing, acts as an inert support for a thin layer of absorbent or enters directly into the separation operation by selective adsorption of, or chemical reaction with, certain species in the feed mixture. Ø Adsorption is confined to the surface of the solid adsorbent, unlike absorption, which occurs throughout the bulk of the absorbent. Ø Such separations are often conducted batchwise or semicontinuously. However, equipment is available to simulate continuous operation. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Separation Operations Based on a Solid Agent 1. Adsorption: is used to remove components present in low concentrations in nonadsorbing solvents or gases and to separate the components in gas or liquid mixtures by selective adsorption on solids, followed by desorption to regenerate the adsorbents, which include activated carbon, activated bentonite, aluminum oxide, silica gel, and synthetic sodium or calcium aluminosilicate zeolite adsorbents Initial or feed phase Vapor or liquid Separating Agent Solid Adsorbent V/L e. g: Removal of heavy metals in H 2 O, Purification of p-xylene Ch. E 334: Separation Processes V/L Dr Saad Al-Shahrani

Separation Operations Based on a Solid Agent Ø A simple adsorption device consists of a cylindrical vessel packed with a bed of solid adsorbent particles through which the gas or liquid flows. Ø Regeneration of the adsorbent is conducted periodically, so two or more vessels are used, one vessel desorbing while the other(s) adsorb(s). Ø Regeneration is accomplished by one of four methods: 1. Vaporizing the adsorbate with a hot purge gas. 2. Reducing the pressure to vaporize the adsorbate. 3. Inert purge stripping without change in temperature or pressure. 4. Displacement desorption by a fluid containing a more strongly adsorbed species. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Separation Operations Based on a Solid Agent 2. Ion exchange: Ion exchange resembles adsorption in that solid Initial or feed phase particles are used and regeneration is necessary. Vapor or liquid However, a chemical reaction is involved Ion exchangers Separating Agent (ion Resin with ion-active sites exchange resins) are either cation exchangers that L L exchange positively charged ions (cations) or anion exchangers that exchange negatively charged ions (anions). L e. g: Water softening. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
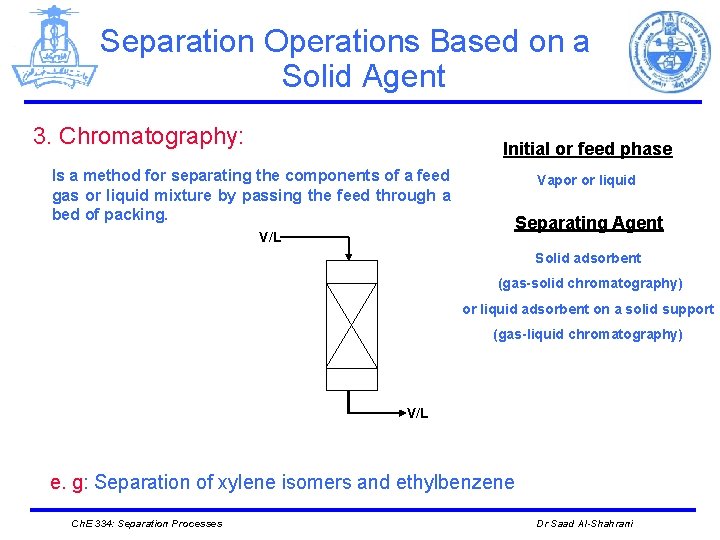
Separation Operations Based on a Solid Agent 3. Chromatography: Initial or feed phase Is a method for separating the components of a feed gas or liquid mixture by passing the feed through a bed of packing. V/L Vapor or liquid Separating Agent Solid adsorbent (gas-solid chromatography) or liquid adsorbent on a solid support (gas-liquid chromatography) V/L e. g: Separation of xylene isomers and ethylbenzene Ch. E 334: Separation Processes Dr Saad Al-Shahrani

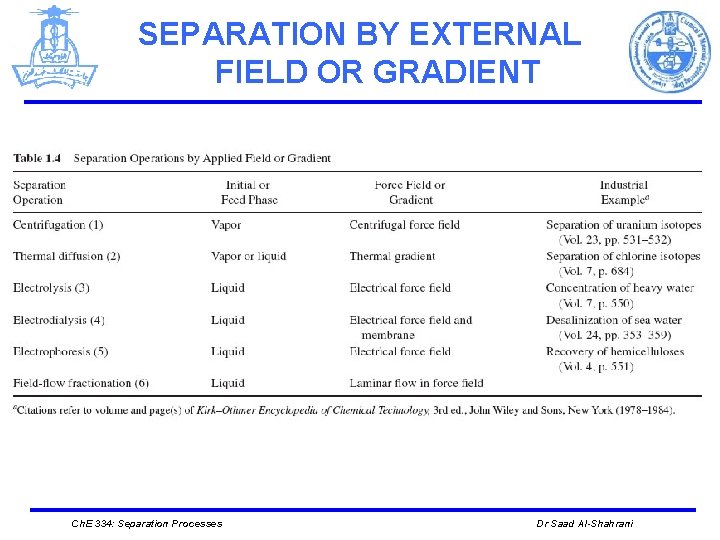
SEPARATION BY EXTERNAL FIELD OR GRADIENT Ø In separation, external fields can be used to take advantage of differing degrees of response of molecules and ions to forces and gradients. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

SEPARATION BY EXTERNAL FIELD OR GRADIENT 1. Centrifugation (uses a pressure field that separates fluid mixtures according to molecular weight ) Initial or feed phase Liquid Force field or gradient Centrifugal force field e. g : Separation Ch. E 334: Separation Processes of uranium isotopes ( 235 UF 6 from 238 UF 6). Dr Saad Al-Shahrani

SEPARATION BY EXTERNAL FIELD OR GRADIENT 2. Thermal Diffusion Initial or feed phase (By applying temperature to a homogeneous Vapor or liquid solution, concentration gradients can be Force field or gradient established and thermal diffusion is induced ) Thermal gradient e. g : Separation of chlorine isotopes. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

SEPARATION BY EXTERNAL FIELD OR GRADIENT 3. Electrolysis When electrical current is applied across a pair of electrodes immersed in the electrolyte. Each electrode attracts ions that are of the opposite charge. Positively- Initial or feed phase liquid Force field or gradient Electrical force field charged ions (cations) move towards the electron-providing (negative) cathode, whereas negatively-charged ions (anions) move towards the positive anode. e. g : Removal of heavy metals from aqueous solutions. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
SEPARATION BY EXTERNAL FIELD OR GRADIENT Ch. E 334: Separation Processes Dr Saad Al-Shahrani
- Slides: 21