SEMEN ANALYSIS What When How Why Dr Sohrab

SEMEN ANALYSIS: What? When? How? Why? Dr Sohrab Salehi

BACKGROUND What is semen, exactly? A mixture of seminal plasma and cells • Seminal plasma contains: – Prostatic fluid (~30% of the volume) – Epididymal plasma (~5% of the volume) – Seminal vesicle fluid (the remainder of the ejaculate) • The cells are: – – – Spermatozoa Germ line cells Leukocytes of various types Bacteria Epithelial cells Occasional red cells © Oozoa Biomedical Inc, April 2005
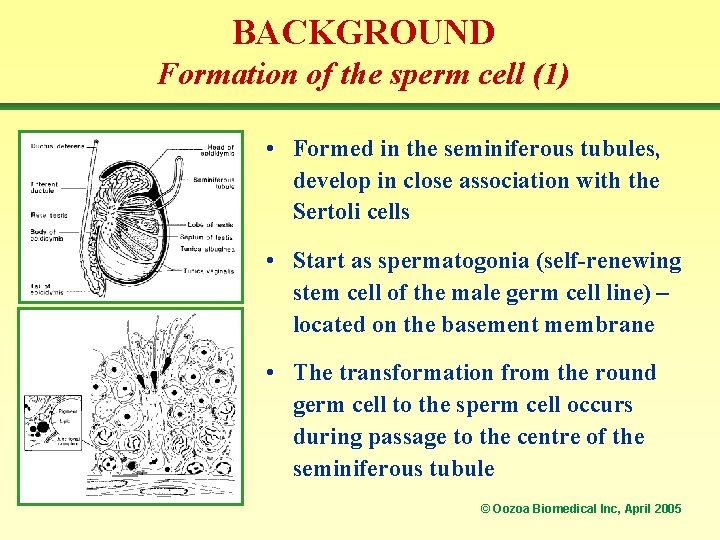
BACKGROUND Formation of the sperm cell (1) • Formed in the seminiferous tubules, develop in close association with the Sertoli cells • Start as spermatogonia (self-renewing stem cell of the male germ cell line) – located on the basement membrane • The transformation from the round germ cell to the sperm cell occurs during passage to the centre of the seminiferous tubule © Oozoa Biomedical Inc, April 2005
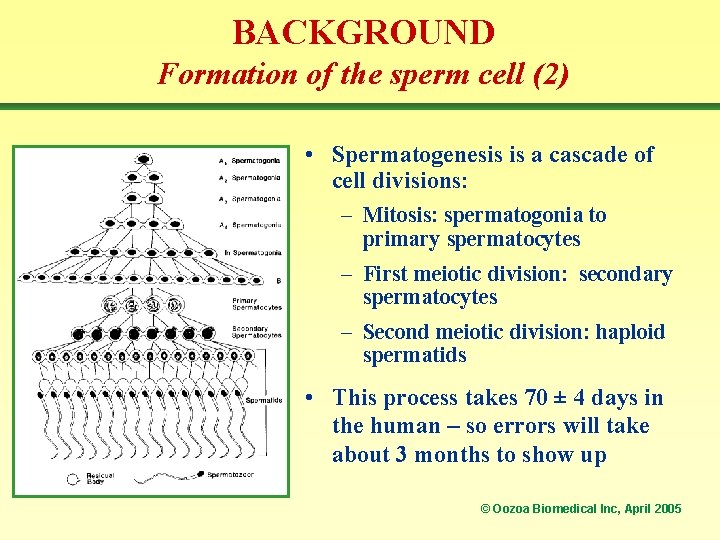
BACKGROUND Formation of the sperm cell (2) • Spermatogenesis is a cascade of cell divisions: – Mitosis: spermatogonia to primary spermatocytes – First meiotic division: secondary spermatocytes – Second meiotic division: haploid spermatids • This process takes 70 ± 4 days in the human – so errors will take about 3 months to show up © Oozoa Biomedical Inc, April 2005
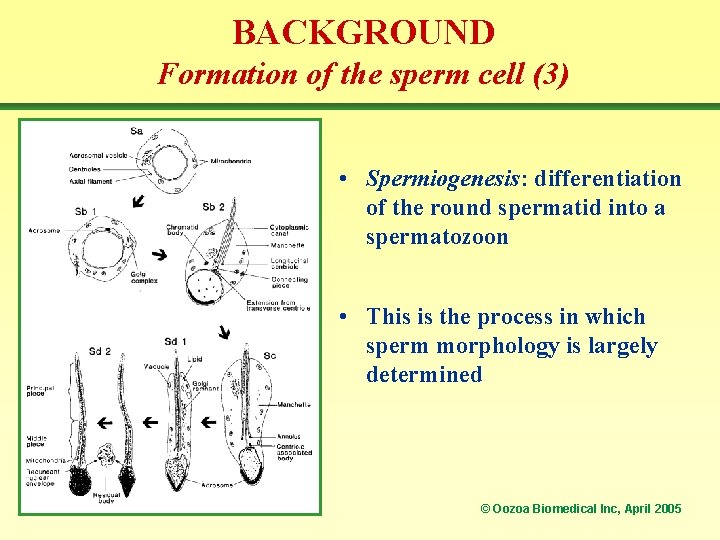
BACKGROUND Formation of the sperm cell (3) • Spermiogenesis: differentiation of the round spermatid into a spermatozoon • This is the process in which sperm morphology is largely determined © Oozoa Biomedical Inc, April 2005

BACKGROUND Sperm transport and seminal plasma • “Testicular sperm” need to undergo more maturation steps before they are ready to fertilize • Transported from the testes to the epididymis, where they mature, and acquire the ability to swim • Then moved to the vas deferens, for storage • At ejaculation, the sperm are transported out of the vas and mix with accessory gland secretions: – prostatic fluid (p. H slightly acidic to neutral; contains citric acid and zinc) – seminal vesicle fluid (p. H strongly alkaline; contains fructose) © Oozoa Biomedical Inc, April 2005

BACKGROUND Sperm function • The ejaculated sperm pass through the cervix, then the uterus, and enter the oviduct • The fertilizing sperm swims through the layers of cells around the egg (cumulus and corona), and reaches the zona pellucida • The sperm then loses the front membranes of its head (the “acrosome reaction”), binds to the zona, then forces its way through the zona to the egg membrane • When the sperm head binds to the egg membrane, its tail stops beating, and the egg incorporates the whole sperm cell • The egg unpacks the sperm, then the male and female pronuclei form. © Oozoa Biomedical Inc, April 2005

WHAT IS A SEMEN ANALYSIS? • An evaluation of spermatogenesis and spermiogenesis. • Traditional descriptive analysis: • interpretation based on population distributions of characteristics, • therefore prone to misinterpretation at the individual level. • Modern approach is to interpret with regard to: • diagnosis of specific lesions; and • indicators of dysfunctional and/or functional potential. • Requires understanding of the relevance of sperm patho-physiology. • In any case, the results must be accurate and reliable. © Oozoa Biomedical Inc, April 2005

WHY PERFORM SEMEN ANALYSIS? • Diagnosis of sterility • Diagnosis of infertility • Prognosis for fertility • Identify treatment options: • surgical treatment • medical treatment • assisted conception treatment Therefore = a screening test to help direct management. © Oozoa Biomedical Inc, April 2005

What is the “standard” approach to semen evaluation? International minimum standards are, by consensus, the World Health Organization’s Lab Manual. • Focus is on standardization with expanded section on quality control. • Methods amenable for use in any (“third world”) country. • Basic infertility work-up. © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Sample Collection For a meaningful result, semen samples must always be collected under standardized conditions: – the container has to be sterile and known NOT to be spermotoxic (i. e. provided by the lab) – the man must have had 3 – 5 days of abstinence – the man must have washed his hands before collection (particularly if microbiological analysis is requested) – the man must NOT have used lubricants (except for Pre~Seed or His~Seed, the only “sperm-friendly” ones) – the sample must be kept at 37°C until analysis, which begins ideally within 30 min, but absolutely within 60 min, of ejaculation © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Sample Handling • The semen sample should be mixed gently during the liquefaction period to promote liquefaction • The sample should NEVER be vortexed (the only exception is for the fixed prep for concentration assessment) • The sample should NEVER be “needled” – if it is too viscous to work with, a known volume of sperm buffer (not PBS) should be added and the sample mixed gently. The added volume must be included in the sperm concentration calculation © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Macroscopic Evaluation • There are several macroscopic evaluations which give useful diagnostic information about the sample: – Appearance – Odour – Liquefaction – Volume – Viscosity – p. H © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Macroscopic Evaluation - Odour • Different people have different abilities to smell semen, so this cannot be standardized • However, when the lid is taken off the collection jar, it should be noted if there is a strong smell of urine or of putrefaction • Samples collected after a prolonged abstinence period (i. e. several weeks) are likely to have a stronger odour © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Macroscopic Evaluation – Liquefaction & Viscosity • Liquefaction is the breakdown of the gel portion of the seminal plasma – the enzymes for this are in the prostatic fluid – A sample with incomplete liquefaction has a gelatinous material in a liquid base – this can be seen when the sample is swirled for the appearance assessment • Viscosity is related to the fluid nature of the whole sample – This is rated subjectively according to the length of the thread of semen produced when the sample is allowed to run back out of the volumetric pipette used to measure the ejaculate volume © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Macroscopic Evaluation - Volume • The volume of the sample should be measured to allow an accurate determination of the sperm number • This is most easily assessed using a warmed disposable volumetric pipette (which is sterile and known NOT to be spermotoxic) • After the sample is measured, allow it to run back into the collection jar, noting its viscosity (a normal sample will have some viscosity – i. e. not watery, but it will flow easily) © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Macroscopic Evaluation - p. H • p. H is important because sperm die at p. H < 6. 9 • The p. H of liquefied semen is normally determined using test strips (we use EM Science Colorp. Hast type, p. H 6. 5– 10. 0) • We usually measure p. H after volume and viscosity – by touching the “emptied” volumetric pipette to the test strip • The normal p. H range is 7. 2– 8. 4 • Inflammatory disorders of the accessory glands can take the p. H outside of this range © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS The wet preparation – set-up • Place 10µl of thoroughly mixed, liquefied semen on a microscope slide and cover with a 22 x 22 mm No 1½ coverslip • There are several important points to keep in mind: – The quality of sperm motility is affected by temperature – the lower the temperature, the poorer the motility, and then cold shock starts to occur at around 15°C. So great care must be taken to ensure that the slides and coverslips, as well as the pipette tips are kept at 37°C – The assessment must start as soon as the flow stops – if this is >1 minute, then a new wet prep must be made – Microscope: phase contrast optics and a heated stage © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS The wet preparation – assessments • The characteristics assessed are: – Motility (to be discussed later) – Sperm aggregation (random clumping) – “some” is normal, but large clumps (each with hundreds of sperm) is abnormal – Spermagglutination (between specific sites) – could suggest the presence of antisperm antibodies. – Round cells: should be <1 per 40× field (~ 1 million/ml). If more abundant, a leukocyte test should be run – Epithelial cells: usually present in small numbers – Erythrocytes: should not be present – Debris: particles smaller than sperm head, may be plentiful – Bacteria and protozoa: presence indicates infection © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Motility assessment – interpretation • The WHO’s Reference values for motility • Total motility(np+pr) 40(38 -42) • Progresivemotility(pr %) 32(31 -34) • MARtest(motilespermatozoawithbound particles, %) <50 • Immunobead test(motilespermatozoawith bound beads, %) <50 © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Sperm concentration – interpretation • The WHO Reference values for: – Sperm concentration is > 15 × 106 sperm/ml(12 -16) – Sperm count is > 39 × 106 sperm per ejaculate(38 -42) • A persistently low sperm concentration is associated with impaired fertility • If a man has a sperm concentration < 5 × 106 sperm/ml, the WHO recommends assessment for numerical and structural abnormalities of sex chromosomes • Azoospermia can indicate a failure of spermatogenesis or blockage(s) in the male tract © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Sperm morphology Normal morphology% 4(3 -4)% © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Semen biochemistry • Zinc: marker for prostatic function – colorimetric assay (WHO)>2. 4 • Fructose: marker for seminal vesicle function, and is a substrate for sperm metabolism – spectrophotometric assay (WHO)>13 • -Glucosidase: secreted exclusively by the epididymis and so is a marker for epididymal function – spectrophotometric assay (WHO)<20 • Peroxidase-positive leukocytes<1 © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Sperm Vitality vitality(live spermatozoa, %)58(55 -63) © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Leukocyte tests • Cytochemical stain for peroxidase activity – benzidinecyanosine is the easiest to use (and it is available in a kit). Only stains those cells with peroxidase activity so does not detect activated polymorphs or lymphocytes • Immunocytochemistry: double antibody assay (primary antibody is a mouse MAb to CD 45), reaction is with alkaline phosphatase: anti-alkaline phosphatase complex (available in a kit) • The tests are run on whole semen • The normal range is < 1 × 106 / ml • Values above this are considered to be clinically significant © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Retrograde ejaculation • In some men, the semen passes back into the bladder at ejaculation - this is confirmed by examination of a sample of post-ejaculatory urine • The man must take sodium bicarbonate the day before, and the day of, his appointment – to alkalinize his urine • Before collection, he should pass urine and then wait until he feels there is some urine in his bladder before masturbating • He then collects a urine sample for analysis • Assess volume and p. H of the urine • Centrifuge (600 g for 10 min), resuspend pellets to a total of 20 ml with sperm buffer + protein • Re-spin, resuspend pellets and combine – add buffer to a final volume of 1 ml • Perform a standard semen analysis with this suspension © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS Post-vasectomy analysis To determine the sperm concentration when few/none expected: – centrifuge as much of the sample as possible (1000 g for 15 min) in a disposable conical centrifuge tube – note the exact volume used – Remove most of the supernatant - note volume removed – Make at least 5 wet preps (10µl of resuspended pellet under a 22 × 22 mm coverslip) – Search through at least 5 wet preps – If anything that looks like a sperm is seen – the report is “not clear” – Final decision is the responsibility of the Pathologist © Oozoa Biomedical Inc, April 2005

SEMEN ANALYSIS What tests should always be done? Always: • Semen volume • Sperm concentration • Differential motility • Morphology If there is an indication: • White cells • Vitality © Oozoa Biomedical Inc, April 2005

ABNORMAL SEMEN ANALYSIS Treatments to correct abnormalities • There is some evidence accumulating that there are some treatments which can improve semen analysis results, including: – antioxidants – carnitine – reducing scrotal temperature • However, there is still no good evidence that these changes in semen analysis results are related to improved fertility • Therefore, two abnormal semen analyses will often lead to a couple being recommended fertility treatments © Oozoa Biomedical Inc, April 2005

FERTILITY WORK-UP • Fertility is a “couple” problem, and so both partners are assessed • For the woman, the investigations may include hormone assessments (such as a day 3 FSH), a hysterosalpingogram, and/or a laparoscopy • Both partners also undergo physical examinations • For these functional assessments, the investigation is based on finding where the problem(s) lie – i. e. predicting failure • The treatment chosen is the one most likely to circumvent the problem(s) – i. e. increasing the likelihood of success © Oozoa Biomedical Inc, April 2005
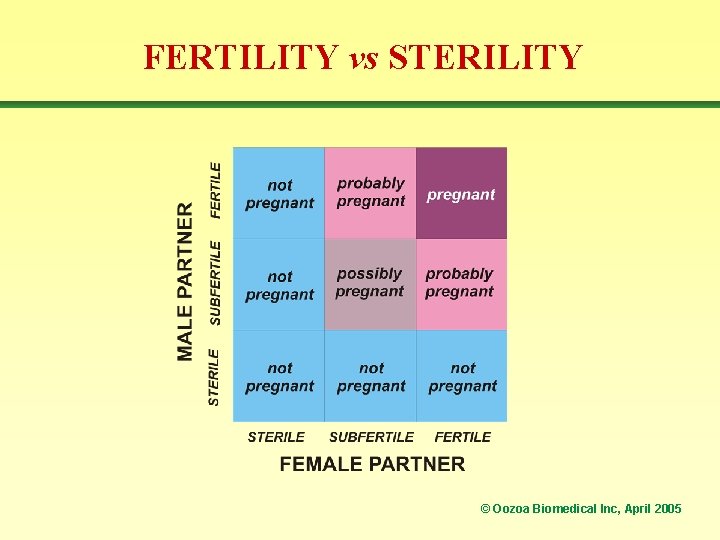
FERTILITY vs STERILITY © Oozoa Biomedical Inc, April 2005

FERTILITY TREATMENT OPTIONS • If a partner is sterile (i. e. no gametes), then the couple would need donor gametes to achieve a pregnancy • If one or both partners are sub-fertile, then the treatment options are: – no treatment, or ovulation induction – intra-uterine insemination (+ ovulation induction) – in vitro fertilization (includes ICSI) © Oozoa Biomedical Inc, April 2005

INTRA-UTERINE INSEMINATION (IUI) • IUI is the least invasive of the ARTs - involves the selective washing of semen to isolate the motile spermatozoa (can’t put whole semen into the uterus) • Up to 15 million motile spermatozoa are inseminated • Advantages: – – relatively inexpensive – simple procedures minimal use of FSH can be used in consecutive cycles can usually start treatment virtually immediately • Disadvantages: – lower success rate per cycle than other ARTs © Oozoa Biomedical Inc, April 2005

IN VITRO FERTILIZATION (IVF) Overview • There are many types of IVF • For virtually all types, the woman is treated with “fertility drugs” to stimulate the development of a group of eggs (the average is around 10 – but the range can be enormous) • Just prior to ovulation, the oocytes are retrieved • That afternoon, they are inseminated with prepared sperm • Inseminated eggs checked the next day for fertilization • The fertilized eggs are kept in culture for up to 5 -6 days • Embryo transfer / possibly cryopreservation © Oozoa Biomedical Inc, April 2005
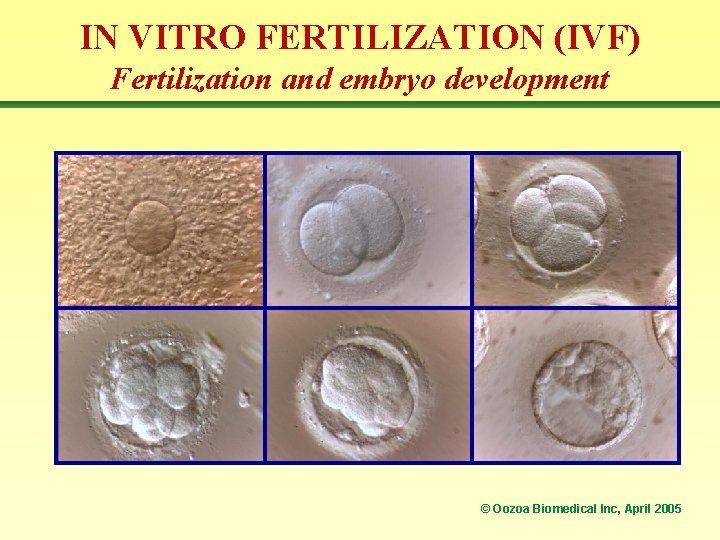
IN VITRO FERTILIZATION (IVF) Fertilization and embryo development © Oozoa Biomedical Inc, April 2005
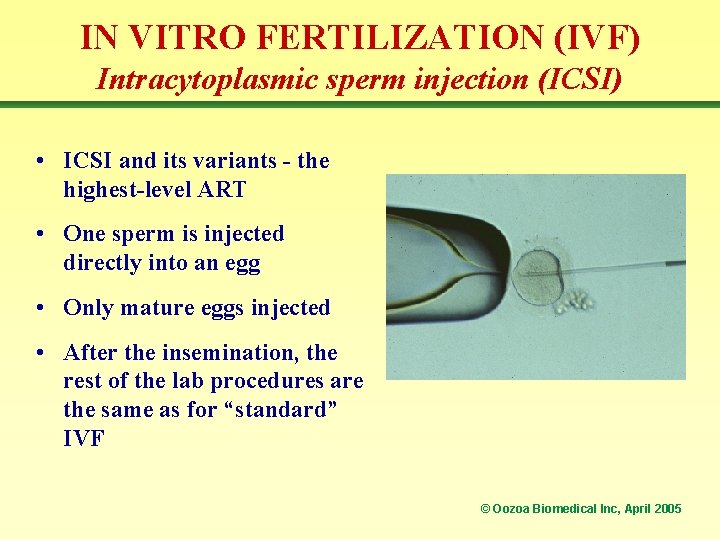
IN VITRO FERTILIZATION (IVF) Intracytoplasmic sperm injection (ICSI) • ICSI and its variants - the highest-level ART • One sperm is injected directly into an egg • Only mature eggs injected • After the insemination, the rest of the lab procedures are the same as for “standard” IVF © Oozoa Biomedical Inc, April 2005

IN VITRO FERTILIZATION (IVF) Risks and rewards • IVF gives the highest chance of pregnancy per cycle – although this is still controlled by the woman’s age • Traditionally there was a high risk of multiple pregnancy with IVF – as several embryos were transferred at once • The new culture systems give embryos with a much higher chance of implanting – so fewer are transferred • The advent of ICSI has allowed men with very poor fertility prognoses (including azoospermia) to become biological fathers • This can bring the requirement for genetic counselling prior to treatment (Y-chromosome deletions, etc. ) © Oozoa Biomedical Inc, April 2005

ART SUCCESS AND FEES Pregnancy rates (women < 37 years old): • No treatment / fertile couple: around 15 -20% per cycle • IUI + COH: 20% per cycle • IVF (includes ICSI): 50 -60% per cycle (was about 10%) Cost in BC (excluding drugs, which can be $2000–$4000): • IUI + COH: $300 – $400 • IVF: $4500 – $4700 • ICSI: $5750 – $6250 • ICSI with aspirated sperm: $6250 – $6500 (+ urologist fees) © Oozoa Biomedical Inc, April 2005

TREATMENT OPTIONS Normal semen analysis The recommended treatment will depend on the woman’s test results, and could be: • No treatment: if her results were normal, and she is young, and they have been trying for only a short time (< 1 year) • IUI: if the couple has already been trying for some years and the woman has normal results • IVF: if the woman is in her mid to late thirties, or if she has blocked tubes © Oozoa Biomedical Inc, April 2005

TREATMENT OPTIONS Low concentration (oligozoospermia) The recommended treatment will depend on the woman’s test results, and could be: • IUI: if enough motile sperm can be recovered from the ejaculate (and if the woman’s results are normal) • IVF: if enough sperm can be recovered from the ejaculate, and if the woman is older, or has blocked tubes. • ICSI: only need to be able to isolate as many motile sperm as there are eggs, so used in cases of extreme oligozoospermia • ICSI with aspirated sperm: in the case of ejaculatory azoospermia (i. e. there are sperm in the testes but not in the ejaculate) © Oozoa Biomedical Inc, April 2005

TREATMENT OPTIONS Low motility (asthenozoospermia) The recommended treatment will depend on the woman’s test results, and could be: • IUI: less likely, and depends on whether enough sperm with good progressive motility can be recovered, and if the woman’s results are normal • IVF: if enough motile sperm can be recovered from the ejaculate, with a final total motility of >90% with good progression (and if hyperactivation is seen) • ICSI: quite likely, as the sperm may not be able to generate enough power to break through the outer layer of the egg (the zona pellucida) © Oozoa Biomedical Inc, April 2005

TREATMENT OPTIONS Poor morphology (teratozoospermia) • If the sperm morphology is very poor, then ICSI is the most likely option • This is due to the high likelihood of failure of fertilization by IUI or IVF, related to functional failure in sperm-egg interactions, including: – Geometric interference in sperm-egg binding (head too round, or too long) – Interference in penetration of the zona (low energy, poor transmission of force due to midpiece or tail defects) © Oozoa Biomedical Inc, April 2005
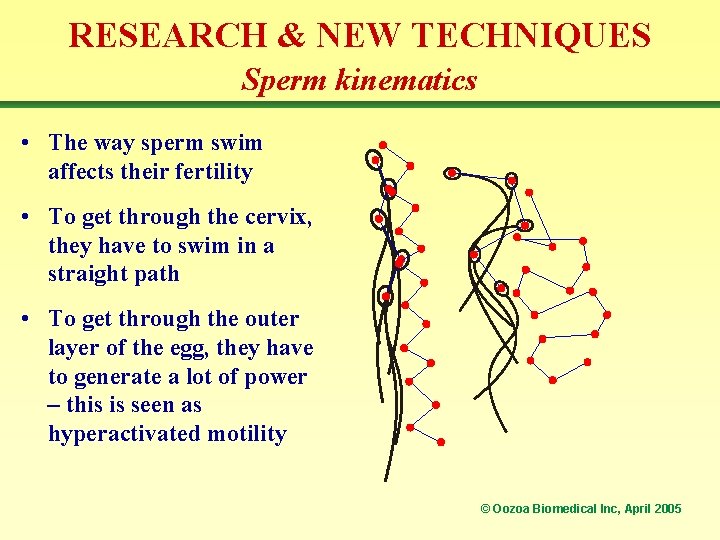
RESEARCH & NEW TECHNIQUES Sperm kinematics • The way sperm swim affects their fertility • To get through the cervix, they have to swim in a straight path • To get through the outer layer of the egg, they have to generate a lot of power – this is seen as hyperactivated motility © Oozoa Biomedical Inc, April 2005

RESEARCH & NEW TECHNIQUES Sperm kinematics • The sperm tracks are analyzed and a number of kinematic parameters are derived, including: – Velocity (VCL, VSL and VAP) – Velocity ratios (expression of the path shape and regularity) – Amplitude of lateral head displacement – Beat/cross frequency • The proportion of sperm in a sample which meet particular kinematic criteria is used to predict (failure) of: – Mucus-penetrating ability – Hyperactivation (a marker of sperm function) © Oozoa Biomedical Inc, April 2005

RESEARCH & NEW TECHNIQUES CASA • Computer-aided sperm analysis • Able to assess the kinematics of hundreds of sperm in a couple of minutes • Means that these tests can be part of the infertility work-up © Oozoa Biomedical Inc, April 2005

RESEARCH & NEW TECHNIQUES ROS and sperm © Oozoa Biomedical Inc, April 2005
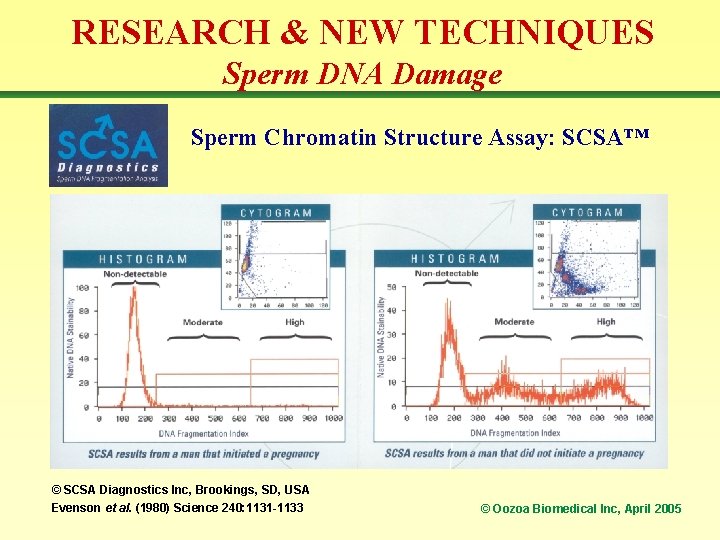
RESEARCH & NEW TECHNIQUES Sperm DNA Damage Sperm Chromatin Structure Assay: SCSA™ © SCSA Diagnostics Inc, Brookings, SD, USA Evenson et al. (1980) Science 240: 1131 -1133 © Oozoa Biomedical Inc, April 2005

RESEARCH & NEW TECHNIQUES Structured management • Use the test results to predict where blocks to fertilization are occurring • A treatment plan to go around these blocks is created for each couple • It is designed to maximize the chance of achieving a successful pregnancy in the most practicable and economical way • The treatment plan is implemented after consultation with the couple © Oozoa Biomedical Inc, April 2005
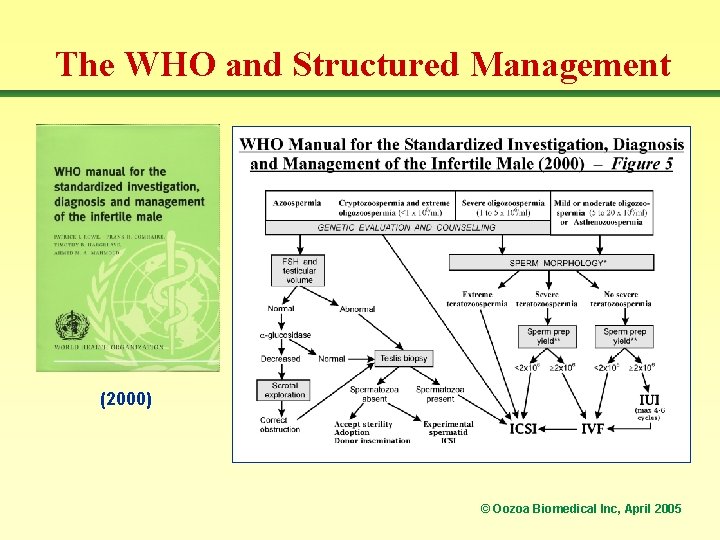
The WHO and Structured Management (2000) © Oozoa Biomedical Inc, April 2005

STANDARDIZATION & QUALITY CONTROL Fundamental requirements of all laboratory tests : • appropriate and robust methodology • careful training • operator experience } accuracy & precision • suitable assay control limits • regular internal comparisons = IQC • regular external comparisons = EQA • cost effectiveness College of Reproductive Biology Inaugural Meeting, Houston, May 1997 © Oozoa Biomedical Inc, April 2005

ISO 15189: 2003 Medical laboratories – Particular requirements for quality and competence. 5. 5. 1 Appropriate procedures that meet the needs of the users of the laboratory’s services shall be used. 5. 5. 2 Only validated procedures shall be used. 5. 5. 3 All procedures must be documented and available: a) b) c) d) e) f) g) h) i) purpose principle performance specifications primary sample(s) sample container equipment & reagents calibration procedural steps QC procedures j) sources of interference k) calculation of results (includes uncertainty of measurement) l) reference intervals m) reportable interval for patients n) alert &/or critical values o) interpretation by laboratory p) safety precautions q) potential sources of variability 5. 5. 4 Performance specifications must related to intended use. 5. 5. 5 Periodic review of biological reference intervals © Oozoa Biomedical Inc, April 2005

QC IN SEMEN ANALYSIS Mean ± SD% differences between 4 trained andrology scientists using WHO manual/visual semen analysis methods on 60 determinations. (Calgary Diagnostic Semen Lab, ca. 1990) Concentration Total motility Prog motility A -2. 3 ± 7. 4 +0. 3 ± 3. 0 +0. 4 ± 2. 6 B -1. 7 ± 4. 9 -0. 8 ± 3. 1 -0. 8 ± 2. 9 C +4. 5 ± 7. 3 -1. 0 ± 3. 3 -0. 6 ± 3. 3 D -0. 5 ± 7. 0 +1. 6 ± 2. 9 +1. 0 ± 2. 8 © Oozoa Biomedical Inc, April 2005
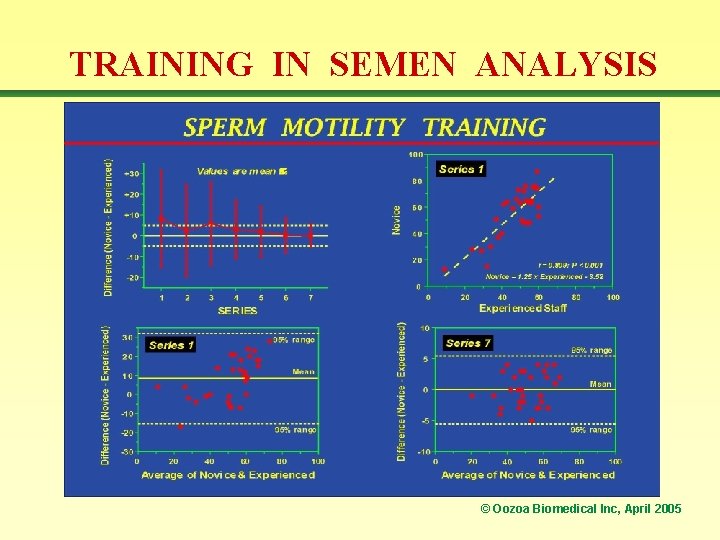
TRAINING IN SEMEN ANALYSIS © Oozoa Biomedical Inc, April 2005

GOAL-ORIENTED TRAINING • Originally elaborated in Calgary during the mid/late 1980 s as a means to facilitate the training of new staff. • Subsequently applied in numerous locations where we were involved in training andrology lab staff: e. g. Sydney (Australia), London (UK), Stockholm (Sweden), Boston (USA), Bangkok (Thailand). • Adopted by the ESHRE Andrology SIG Education Subcommittee as the basis for their Basic Semen Analysis Courses (25 run by the end of 2003). © Oozoa Biomedical Inc, April 2005
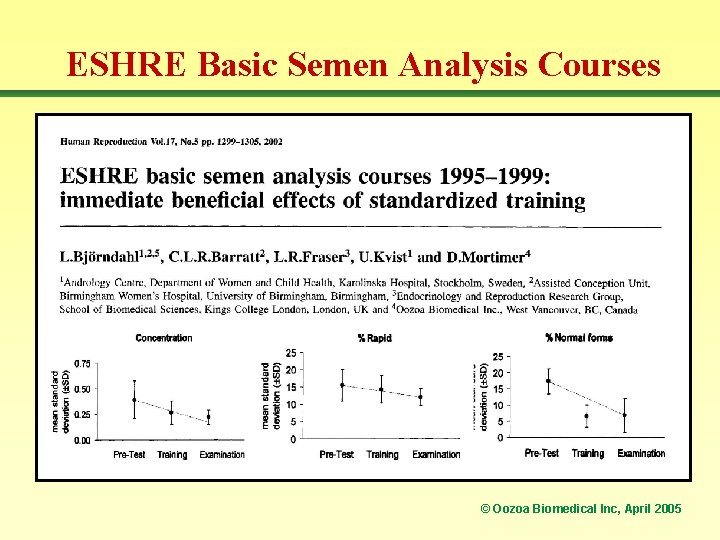
ESHRE Basic Semen Analysis Courses © Oozoa Biomedical Inc, April 2005

CONCLUSION • Semen analysis is an important laboratory test and should be thought of in the same way as any other diagnostic assay • It is used in determining treatment plans for infertility • The results can therefore have a huge impact on the level of intervention, with the associated emotional and financial costs to the couple © Oozoa Biomedical Inc, April 2005
- Slides: 56