Selfassembly of nanoparticles Selfassembly of nanoparticles Spontaneous selfassembly

Self-assembly of nanoparticles
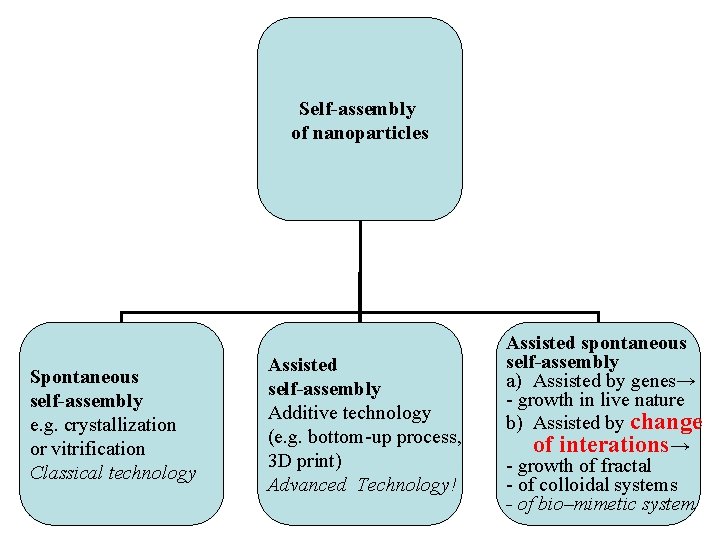
Self-assembly of nanoparticles Spontaneous self-assembly e. g. crystallization or vitrification Classical technology Assisted self-assembly Additive technology (e. g. bottom-up process, 3 D print) Advanced Technology! Assisted spontaneous self-assembly a) Assisted by genes→ - growth in live nature b) Assisted by change of interations→ - growth of fractal - of colloidal systems - of bio–mimetic system

Assisted spontaneous self-assembly in nature Growth of fractal (fern) by DLA of clusters of atoms C, O, H, N, …) enabled by 1. their Brownian motion, and 2. concentration gradients in air or water 3. assisted by genes.
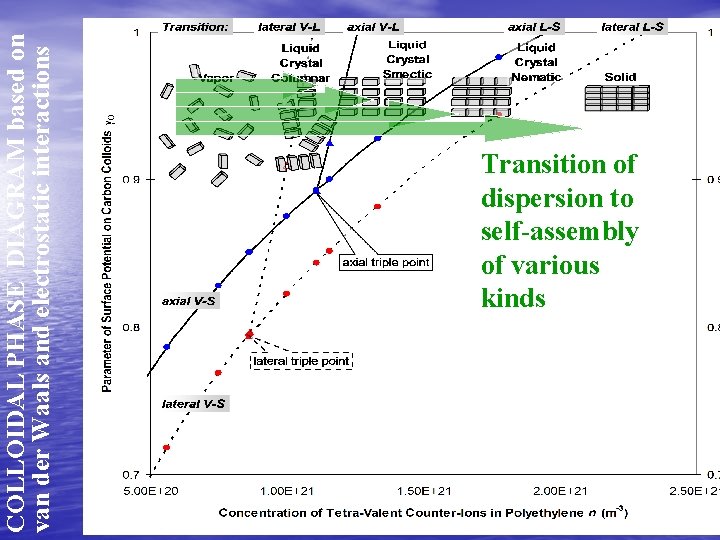
COLLOIDAL PHASE DIAGRAM based on van der Waals and electrostatic interactions Transition of dispersion to self-assembly of various kinds
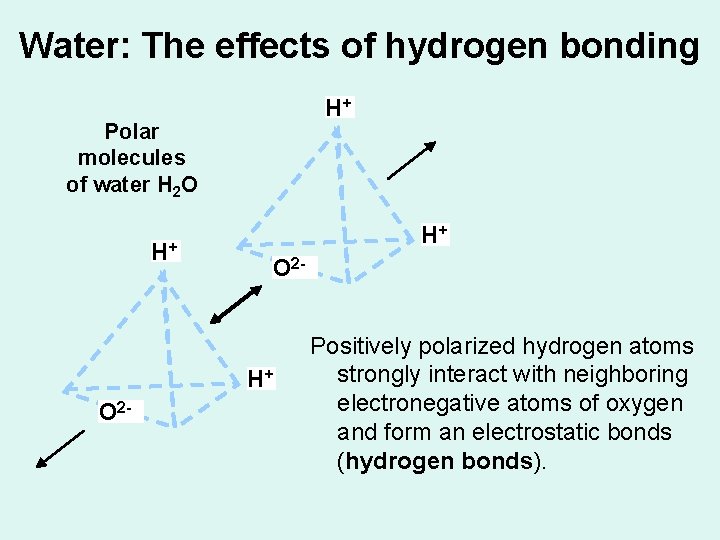
Water: The effects of hydrogen bonding H+ Polar molecules of water H 2 O H+ H+ O 2 - Positively polarized hydrogen atoms strongly interact with neighboring electronegative atoms of oxygen and form an electrostatic bonds (hydrogen bonds).
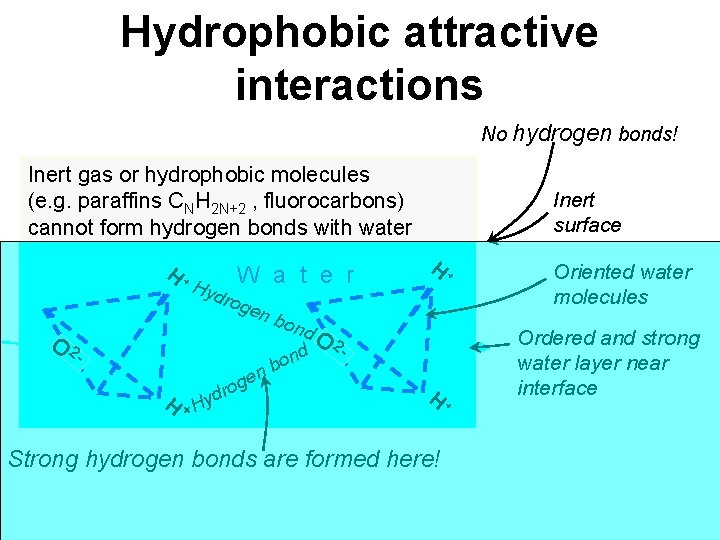
Hydrophobic attractive interactions No hydrogen bonds! Inert gas or hydrophobic molecules (e. g. paraffins CNH 2 N+2 , fluorocarbons) cannot form hydrogen bonds with water H+ Hyd W a t e r rog en O 2 - bon d o nb e g r H + Hyd O 2 Inert surface H+ - H+ Strong hydrogen bonds are formed here! Oriented water molecules Ordered and strong water layer near interface

Hydrophilic Effect Hydrophilic molecules prefer to be in contact with water→they are water soluble and repeal each other in water. Examples of hydrophilic molecules: Ø Ions and charged molecules Ø Polar molecules and groups Ø Molecules capable of hydrogen bonds e. g. electronegative atoms such as N, F, and Cl.
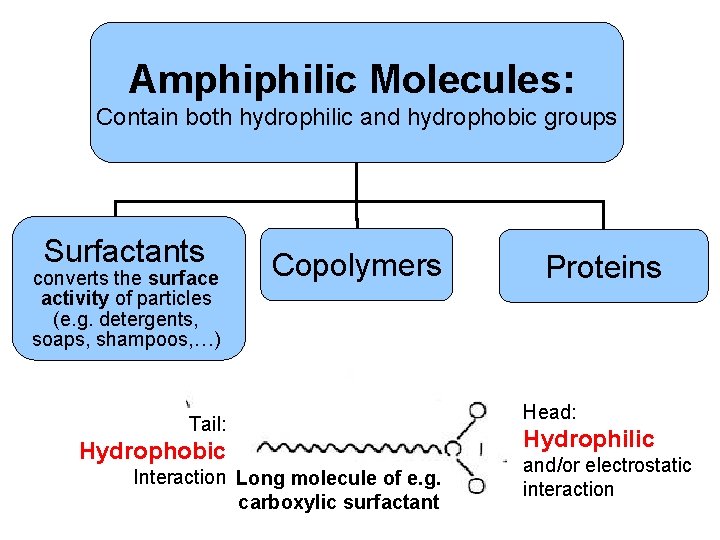
Amphiphilic Molecules: Contain both hydrophilic and hydrophobic groups Surfactants converts the surface activity of particles (e. g. detergents, soaps, shampoos, …) Copolymers x x x Tail: x Hydrophobic x Interaction Long molecule of e. g. x carboxylic surfactant Proteins Head: Hydrophilic and/or electrostatic interaction
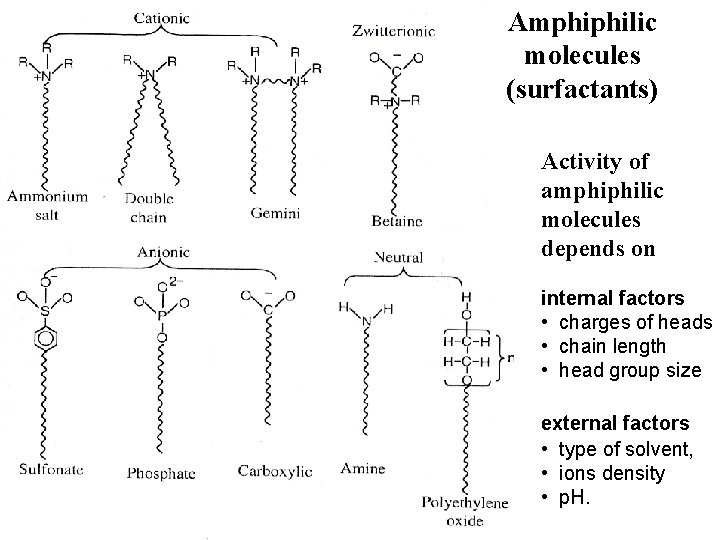
Amphiphilic molecules (surfactants) Activity of amphiphilic molecules depends on internal factors • charges of heads • chain length • head group size external factors • type of solvent, • ions density • p. H.

Amphiphlic molecules inside water
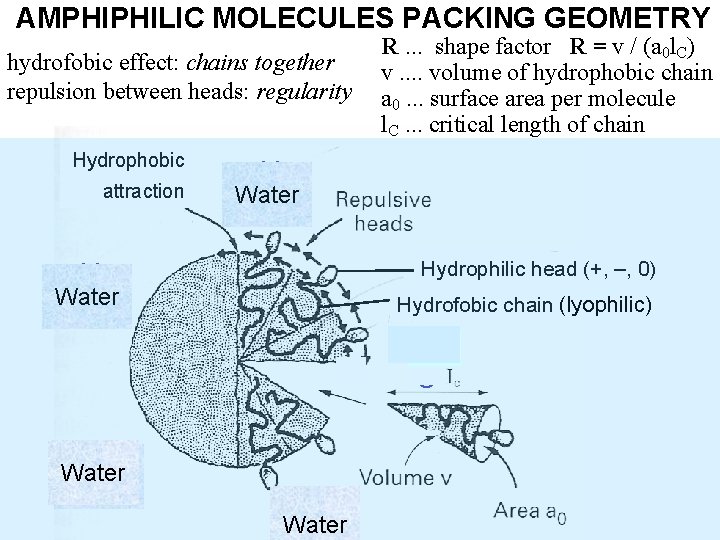
AMPHIPHILIC MOLECULES PACKING GEOMETRY hydrofobic effect: chains together repulsion between heads: regularity Hydrophobic attraction H 2 Water O Hydrophilic head (+, –, 0) H 2 Water O R. . . shape factor R = v / (a 0 l. C) v. . volume of hydrophobic chain a 0. . . surface area per molecule l. C. . . critical length of chain Hydrofobic chain (lyophilic) H 2 O H 2 Water
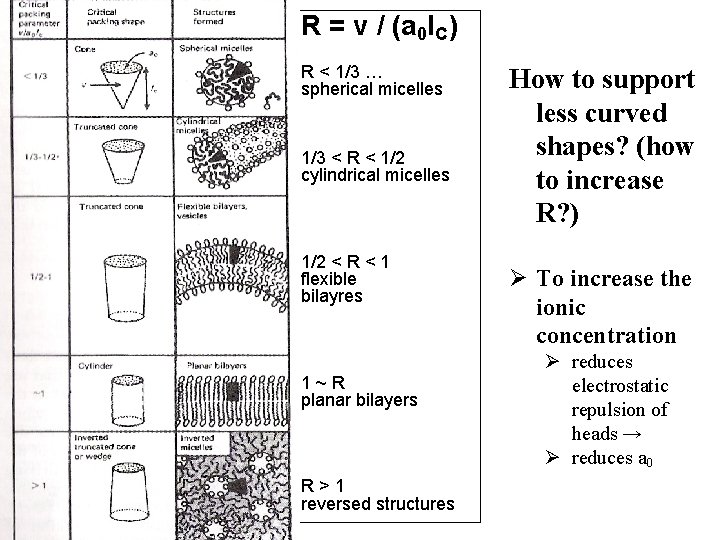
R = v / (a 0 l. C) R < 1/3 … spherical micelles 1/3 < R < 1/2 cylindrical micelles 1/2 < R < 1 flexible bilayres 1~R planar bilayers R>1 reversed structures How to support less curved shapes? (how to increase R? ) Ø To increase the ionic concentration Ø reduces electrostatic repulsion of heads → Ø reduces a 0
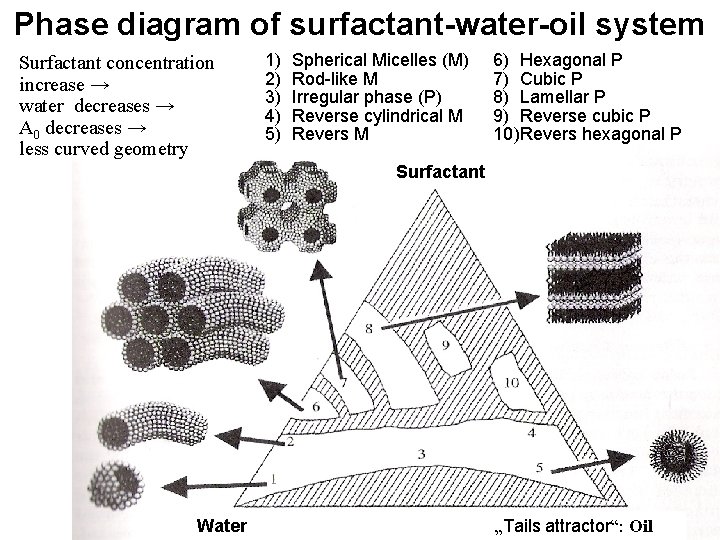
Phase diagram of surfactant-water-oil system Surfactant concentration increase → water decreases → A 0 decreases → less curved geometry 1) 2) 3) 4) 5) Spherical Micelles (M) Rod-like M Irregular phase (P) Reverse cylindrical M Revers M 6) Hexagonal P 7) Cubic P 8) Lamellar P 9) Reverse cubic P 10)Revers hexagonal P Surfactant Water „Tails attractor“: Oil

Amphiphlic molecules on the surface of water

Langmuir - Blodgett films Langmuir (born in Boston 1706) studied the effect of oils on water surfaces and proposed the new method of creation of ordered molecular monolayer. Langmuir-Blodgett (France) technology for the multilayer ordered molecular monolayers on the solid substrates
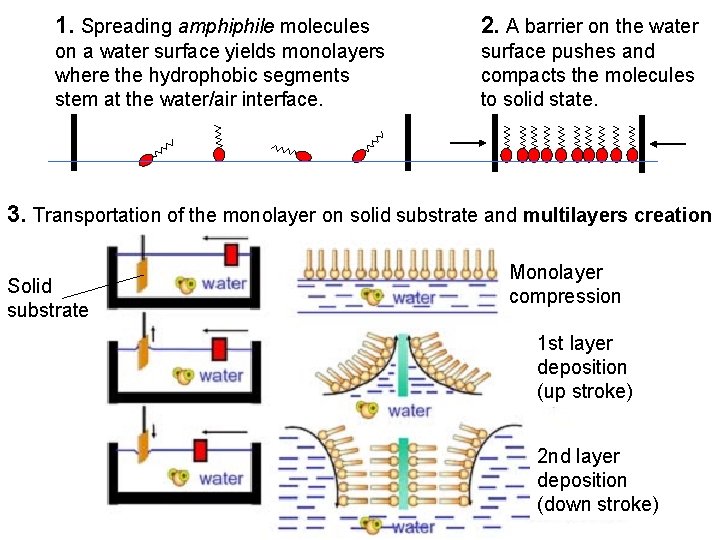
1. Spreading amphiphile molecules 2. A barrier on the water on a water surface yields monolayers where the hydrophobic segments stem at the water/air interface. surface pushes and compacts the molecules to solid state. 3. Transportation of the monolayer on solid substrate and multilayers creation Solid substrate Monolayer compression 1 st layer deposition (up stroke) 2 nd layer deposition (down stroke)
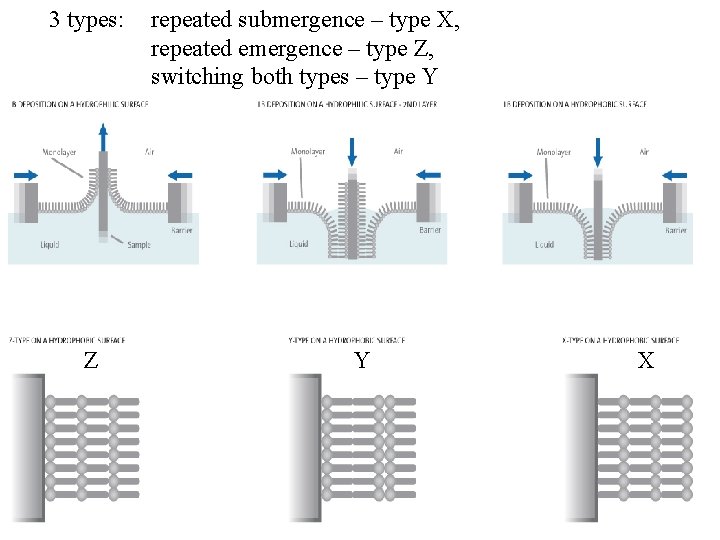
3 types: Z repeated submergence – type X, repeated emergence – type Z, switching both types – type Y Y X

SELF-ASSEMBLED BLOCK COPOLYMER NANOSTRUCTURES • Block copolymers are composed of distinctively different polymer chain segments of amphiphilic molecules.
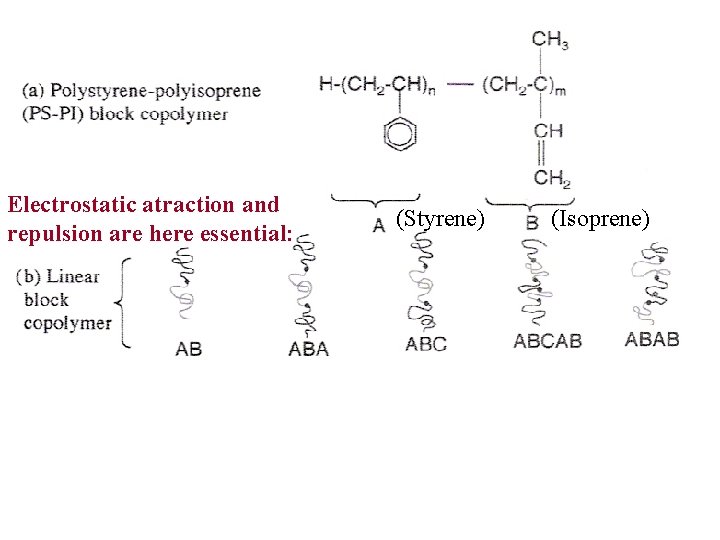
Electrostatic atraction and repulsion are here essential: (Styrene) (Isoprene)
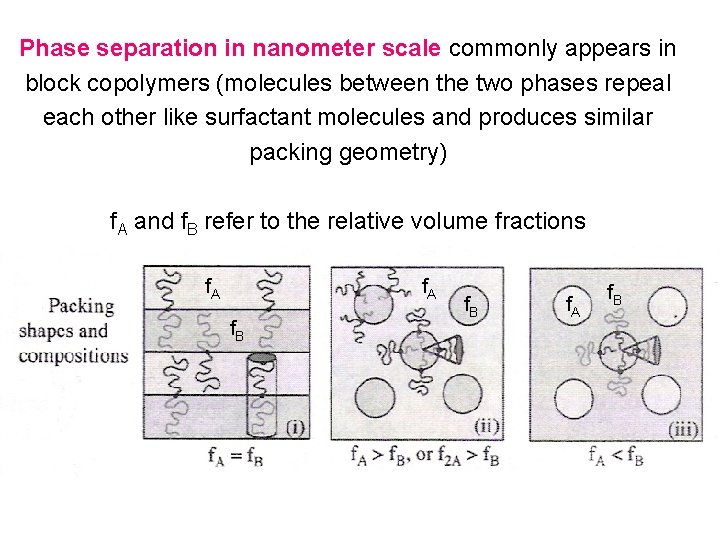
Phase separation in nanometer scale commonly appears in block copolymers (molecules between the two phases repeal each other like surfactant molecules and produces similar packing geometry) f. A and f. B refer to the relative volume fractions f. A f. B
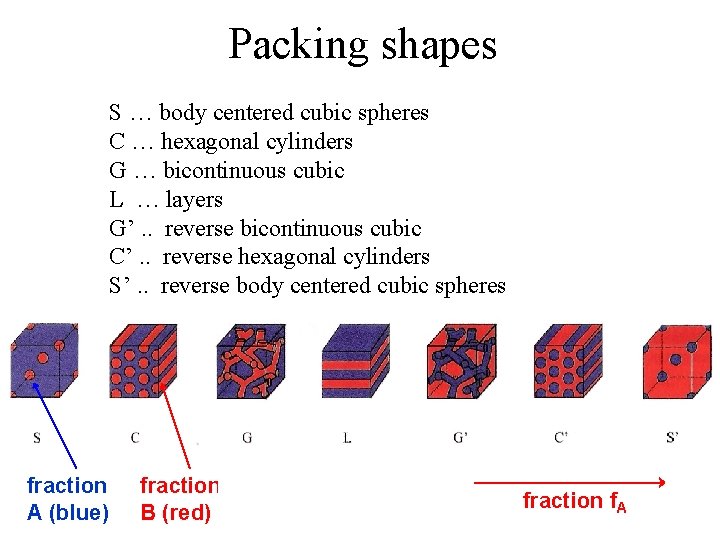
Packing shapes S … body centered cubic spheres C … hexagonal cylinders G … bicontinuous cubic L … layers G’. . reverse bicontinuous cubic C’. . reverse hexagonal cylinders S’. . reverse body centered cubic spheres fraction A (blue) fraction B (red) fraction f. A
Deoxyribonucleic Acid (DNA)
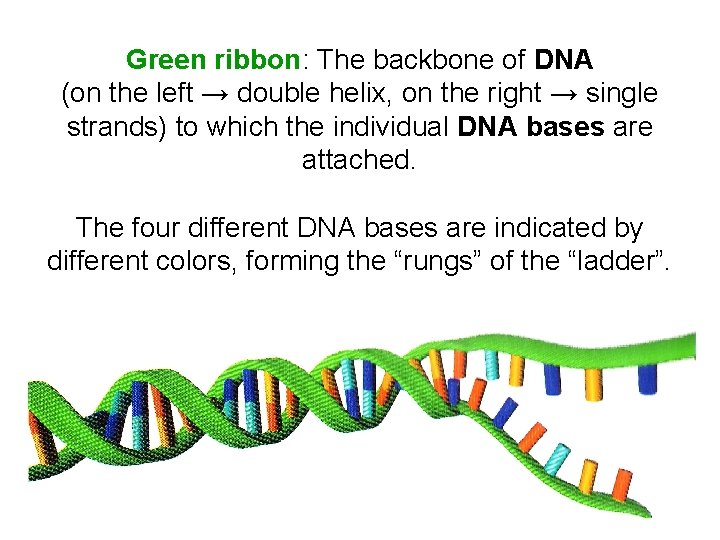
Green ribbon: The backbone of DNA (on the left → double helix, on the right → single strands) to which the individual DNA bases are attached. The four different DNA bases are indicated by different colors, forming the “rungs” of the “ladder”.
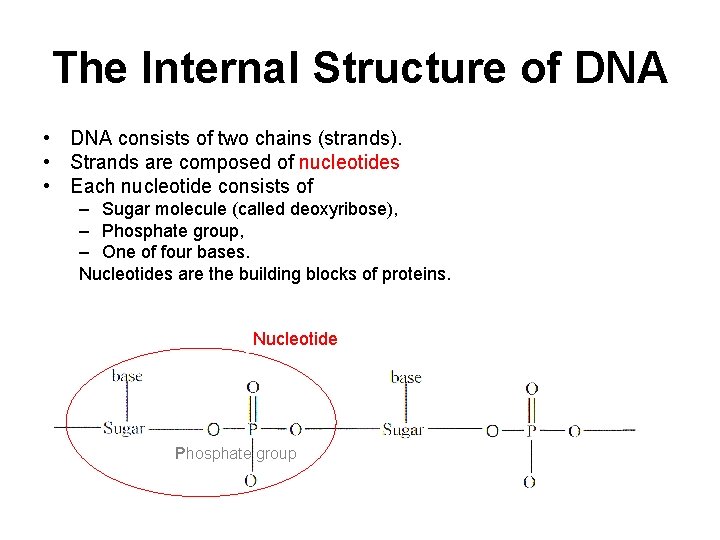
The Internal Structure of DNA • DNA consists of two chains (strands). • Strands are composed of nucleotides • Each nucleotide consists of – Sugar molecule (called deoxyribose), – Phosphate group, – One of four bases. Nucleotides are the building blocks of proteins. Nucleotide Phosphate group
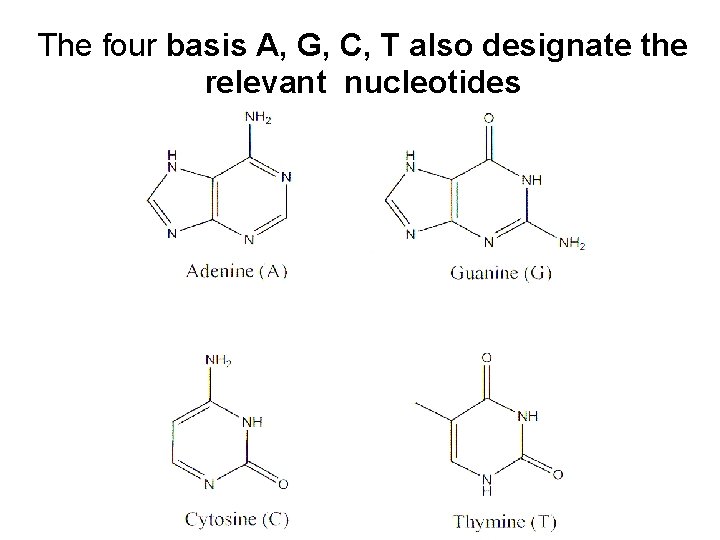
The four basis A, G, C, T also designate the relevant nucleotides The sugar molecule (Deoxribosa)
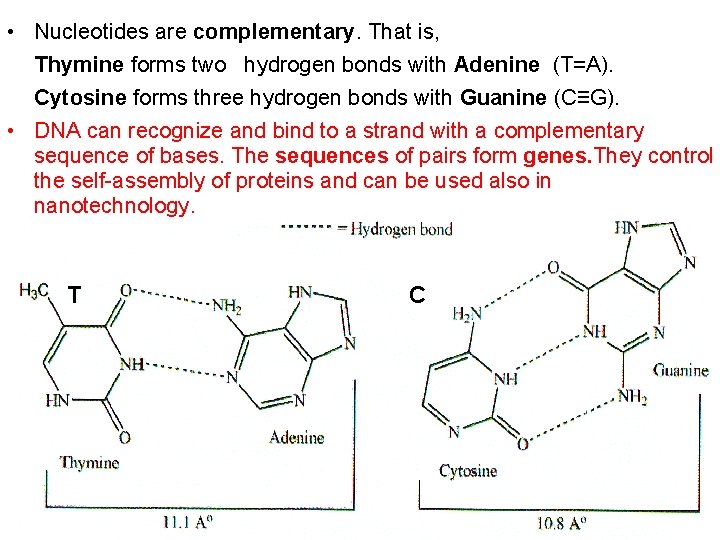
• Nucleotides are complementary. That is, Thymine forms two hydrogen bonds with Adenine (T=A). Cytosine forms three hydrogen bonds with Guanine (C≡G). • DNA can recognize and bind to a strand with a complementary sequence of bases. The sequences of pairs form genes. They control the self-assembly of proteins and can be used also in nanotechnology. T C
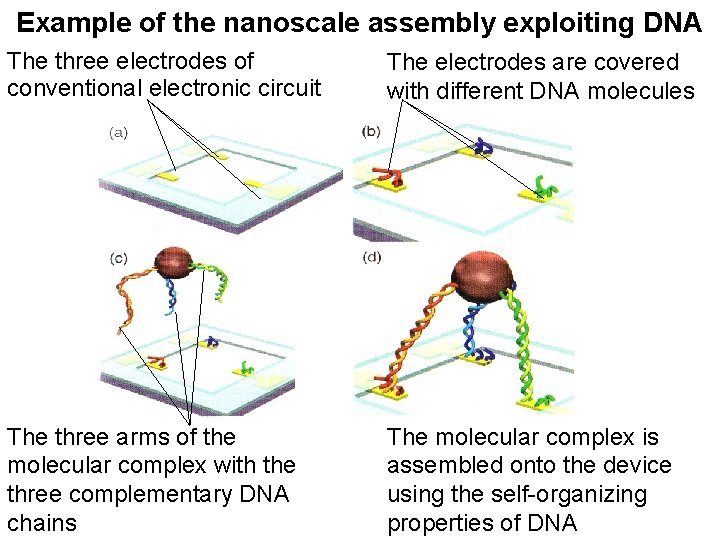
Example of the nanoscale assembly exploiting DNA The three electrodes of conventional electronic circuit The electrodes are covered with different DNA molecules The three arms of the molecular complex with the three complementary DNA chains The molecular complex is assembled onto the device using the self-organizing properties of DNA

Some possible application of DNA in nanotechnology 1) DNA can store information with extremely high density. 2) DNA not only store and transport information but constitutes parts of the machinary of the protein (in biology) or artificial devices (in technology) as well.
- Slides: 28