Selfassembled mesoporous metal oxide thin films Purdue University

Self-assembled mesoporous metal oxide thin films Purdue University MSE REU August 5, 2004 Heidi Springer Advisor: Dr. Hugh Hillhouse Vikrant Urade

What are mesoporous materials? n n Porous materials whose pores are between 2 and 50 nm in diameter. The mesoporous materials investigated here have uniformly ordered pores. Silica based materials first synthesized by scientists at the Mobil Corporation in 1992. Non-silica mesoporous materials first reported at UC Santa Barbara in 1994.

Applications Molecular sieves n High surface area catalysts n Gas sensors n Dye sensitized photovoltaic solar cells n

Mesoporous materials are templated by amphiphilic molecules. n n n Courtesy of Brian Eggiman An amphiphillic molecule has a hydrophillic (water loving) head and a hydrophobic (water loathing) tail. A micelle is an association of amphiphillic molecules. Mesoporous structure is created by the ordered packing of micelles.

Why do amphiphiles form micelles? n Gibb’s Free Energy (ΔG) is a thermodynamic quantity which predicts the spontaneity of a reaction. ¨ n n n A decrease in Gibb’s Free Energy indicates a spontaneous reaction. Water molecules create structure by forming hydrogen bonds with one another. The hydrophobic tails of individual amphiphiles placed in solution force water molecules to associate in a particular way. This decreases the entropy (ΔS) of the system. When the hydrophobic tails associate to each other (form micelles) in order to minimize their interaction with water molecules they increase the entropy of the system. ΔG = ΔH - TΔS n An increase in entropy, decreases Gibb’s Free Energy of the system therefore the reaction will occur spontaneously.
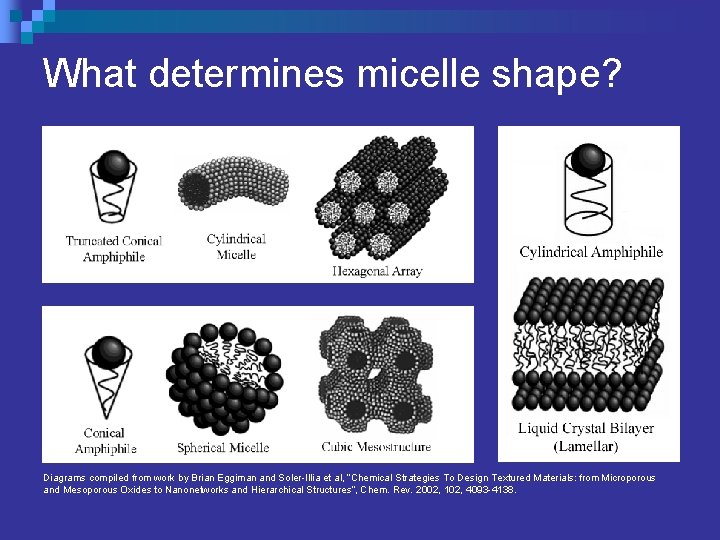
What determines micelle shape? Diagrams compiled from work by Brian Eggiman and Soler-Illia et al, “Chemical Strategies To Design Textured Materials: from Microporous and Mesoporous Oxides to Nanonetworks and Hierarchical Structures”, Chem. Rev. 2002, 102, 4093 -4138.

How do we use micelles to form mesoporous metal oxides? Courtesy of Brian Eggiman

Pore Structure Characterization n X-Ray Diffraction ¨ Small n angle XRD 0. 6 to 3. 0 degrees 2θ ¨ Peaks in XRD pattern show d-spacing between micelles (pores) in parallel planes. n Transmission Electron Microscopy (TEM)
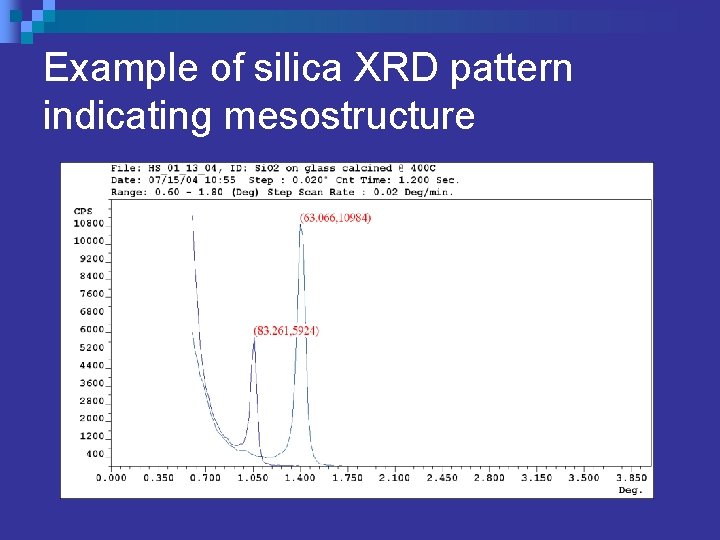
Example of silica XRD pattern indicating mesostructure

Research Goal n Synthesize mesoporous tin oxide thin films with ordered cubic structure. ¨ Explore impact of surfactant concentration on mesostructure formation using amphiphilic triblock copolymers (PEO-PPO-PEO).

Experimental Method Ø Surfactant (amphiphile source) solution is mixed with metal oxide precursor. Ø Combined solution is deposited onto glass slides using dip coating. Ø As slides are withdrawn from solution, solvent evaporates leaving hybrid surfactant/metal oxide thin film.

Experimental Method Ø Samples are subjected to humidity or thermal treatments to aid structure condensation. Ø Samples are calcined to remove organic template leaving a uniformly structured mesoporous material.

Surfactant Concentration P 123 n Pluronic P 123 (EO 20 PO 70 EO 20) ¨ Maintained consistent solution of: 5. 4 grams tin (IV) chloride n 5. 4 grams water n 41 grams ethyl alcohol. n ¨ Varied P 123 content from 1. 2 g – 1. 8 g ¨ (EO): Sn molar ratio of 0. 4 – 0. 6.
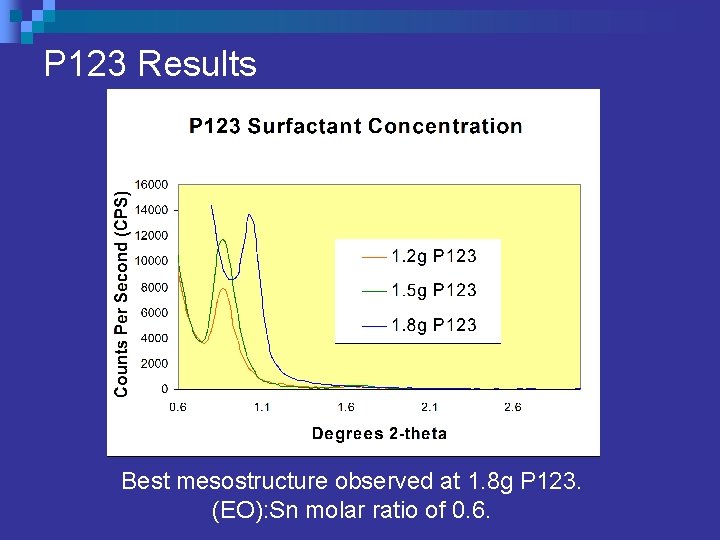
P 123 Results Best mesostructure observed at 1. 8 g P 123. (EO): Sn molar ratio of 0. 6.

Surfactant Concentration F 127 n Pluronic F 127 (EO 106 PO 70 EO 106) ¨ Maintained consistent solution of: 5. 4 grams tin (IV) chloride n 5. 4 grams water n 41 grams ethyl alcohol. n ¨ Varied F 127 content from. 75 - 2. 0 grams.
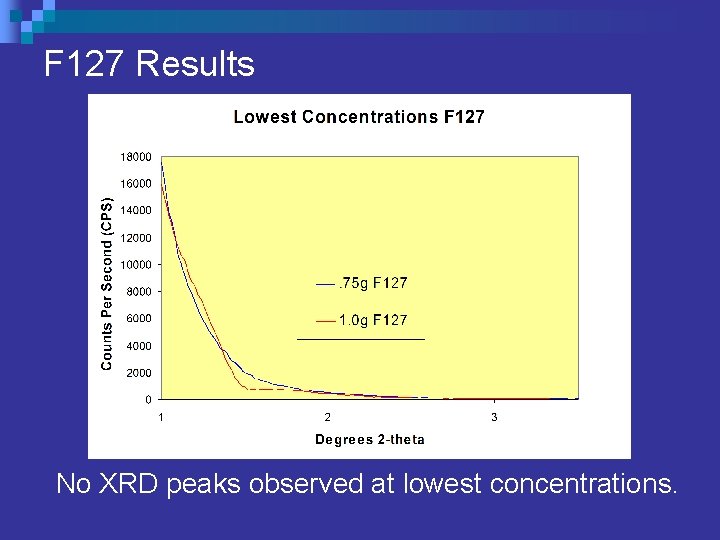
F 127 Results No XRD peaks observed at lowest concentrations.
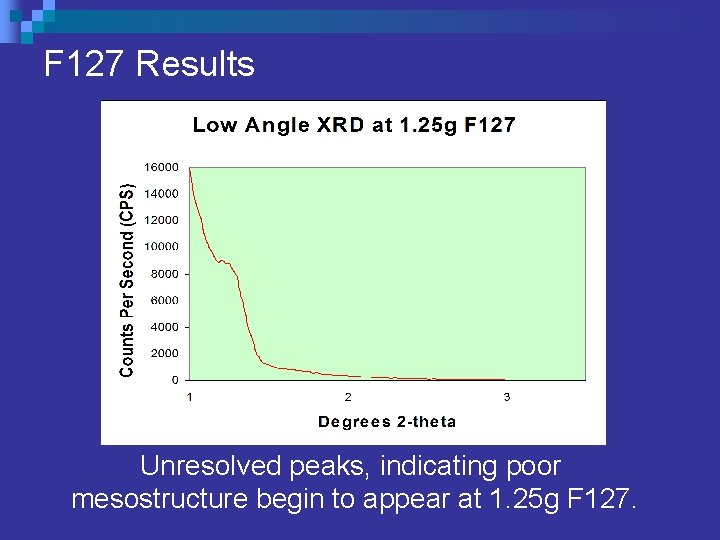
F 127 Results Unresolved peaks, indicating poor mesostructure begin to appear at 1. 25 g F 127.
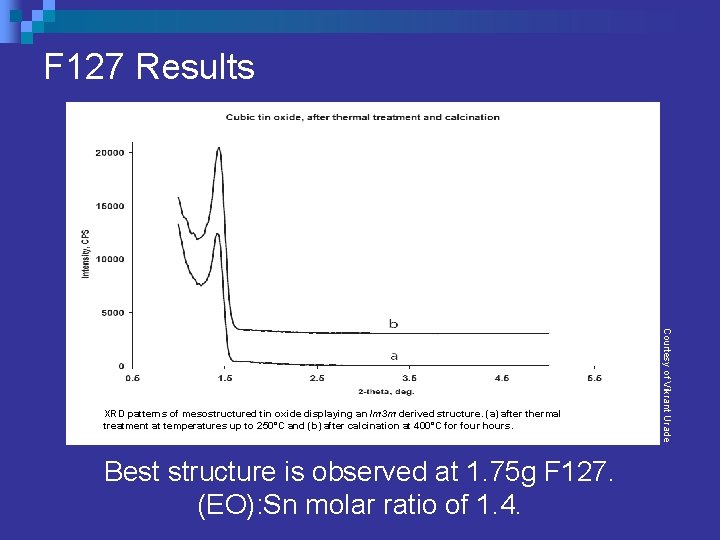
F 127 Results Best structure is observed at 1. 75 g F 127. (EO): Sn molar ratio of 1. 4. Courtesy of Vikrant Urade XRD patterns of mesostructured tin oxide displaying an Im 3 m derived structure. (a) after thermal treatment at temperatures up to 250°C and (b) after calcination at 400°C for four hours.
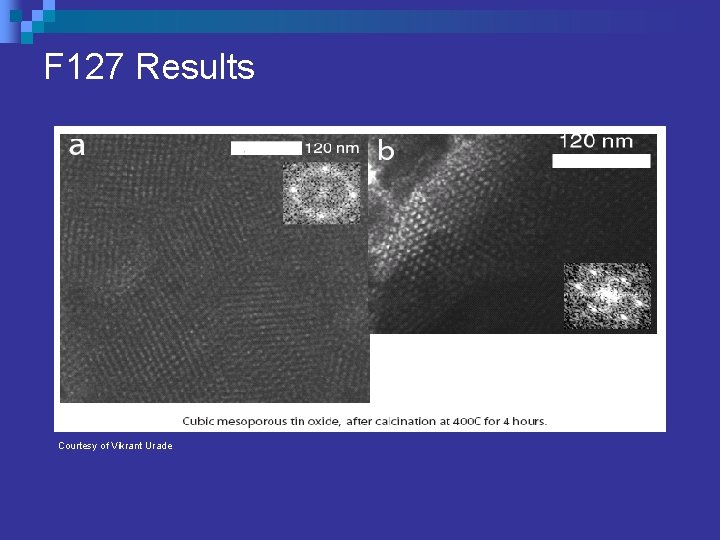
F 127 Results Courtesy of Vikrant Urade

Future Work n Expand synthesis methods to other metal oxides. ¨ Synthesize p-type mesoporous metal oxide for dye-sensitized solar cells.

Acknowledgments Professor Hillhouse n Vikrant Urade and Brian Eggiman n Members of the Hillhouse group n 2004 MSE REU group n

Thank You
- Slides: 22