Selective removal of oxygen by carbon monoxide instead

Selective removal of oxygen by carbon monoxide instead of hydrogen Maria Eugenia Sad Post-doctoral fellow Enrique Iglesia Principal Researcher Department of Chemical Engineering University of California at Berkeley Financial Support: BP January 14 th 2009
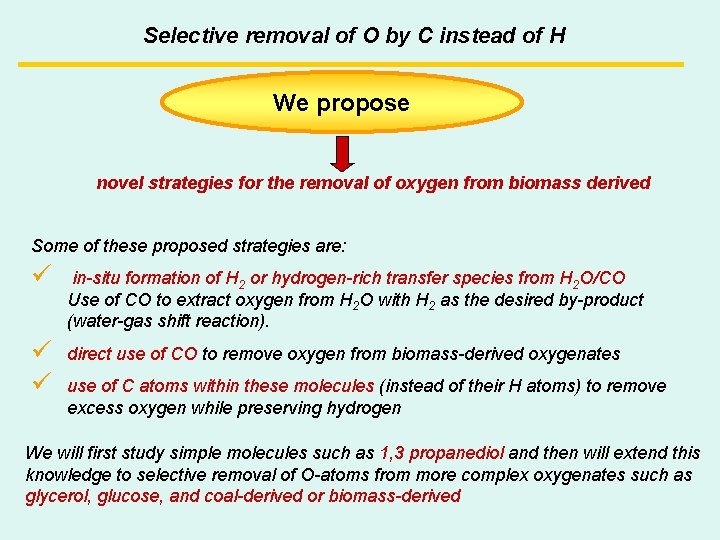
Selective removal of O by C instead of H We propose novel strategies for the removal of oxygen from biomass derived Some of these proposed strategies are: ü in-situ formation of H 2 or hydrogen-rich transfer species from H 2 O/CO Use of CO to extract oxygen from H 2 O with H 2 as the desired by-product (water-gas shift reaction). ü ü direct use of CO to remove oxygen from biomass-derived oxygenates use of C atoms within these molecules (instead of their H atoms) to remove excess oxygen while preserving hydrogen We will first study simple molecules such as 1, 3 propanediol and then will extend this knowledge to selective removal of O-atoms from more complex oxygenates such as glycerol, glucose, and coal-derived or biomass-derived

Our catalysts Cu based catalysts Cu. O/Zn. O/Al 2 O 3 catalysts (Cu. O 52. 5 %, Zn. O 30. 2%, Al 2 O 3 17. 0%) • Cu is a well-know catalyst used for WGS reaction at low temperatures. Zinc oxide is used as structural stabilizers and promoters. Aluminum oxide, although inactive for the WGS reaction, is added to improve the catalyst dispersion Fe based catalysts Fe-Zn oxides prepared by coprecipitation – Zn/Fe atomic ratio=0. 1 • Fe is useful to carry out WGS reaction at high temperatures. Chromium oxide or zinc oxide are generally used as structural stabilizers and promoters Pt based catalysts • 1% wt Pt/Si. O 2 prepared by incipient wetness impregnation • 1. 5% wt Pt/Al 2 O 3 prepared by incipient wetness impregnation • Precious metal-based catalysts are also used for WGS and they catalyze hydrogenation reaction too.
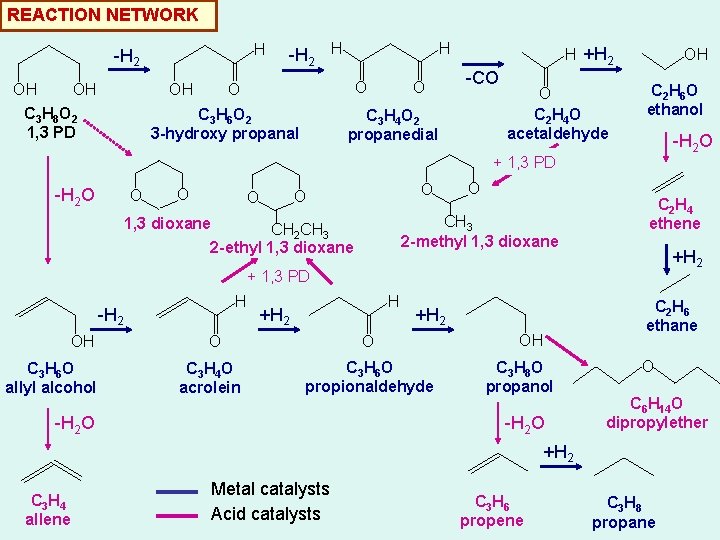
REACTION NETWORK H -H 2 OH OH OH C 3 H 8 O 2 1, 3 PD -H 2 H H O O C 3 H 6 O 2 3 -hydroxy propanal O H +H 2 -CO C 3 H 4 O 2 propanedial O C 2 H 4 O acetaldehyde OH C 2 H 6 O ethanol -H 2 O + 1, 3 PD -H 2 O O O O CH 3 2 -methyl 1, 3 dioxane CH 2 CH 3 2 -ethyl 1, 3 dioxane C 2 H 4 ethene +H 2 + 1, 3 PD H -H 2 OH C 3 H 6 O allyl alcohol H +H 2 O C 3 H 4 O acrolein +H 2 O C 3 H 6 O propionaldehyde -H 2 O OH C 3 H 8 O propanol -H 2 O C 2 H 6 ethane O C 6 H 14 O dipropylether +H 2 C 3 H 4 allene Metal catalysts Acid catalysts C 3 H 6 propene C 3 H 8 propane
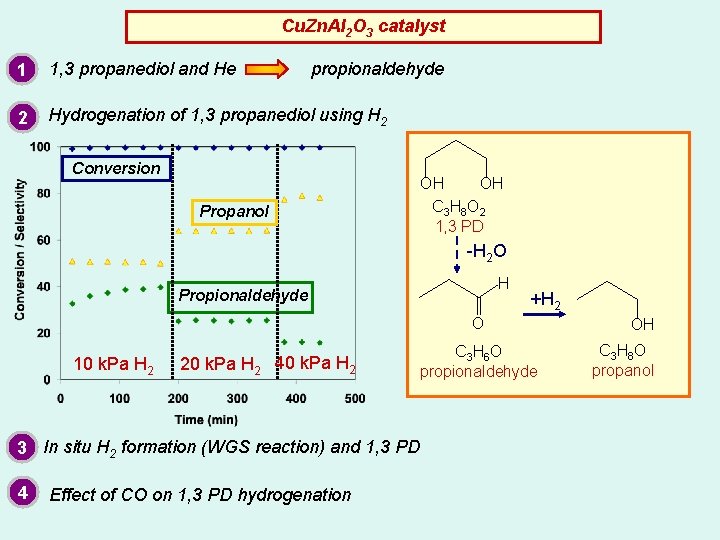
Cu. Zn. Al 2 O 3 catalyst 1 1, 3 propanediol and He propionaldehyde 2 Hydrogenation of 1, 3 propanediol using H 2 Conversion Propanol OH OH C 3 H 8 O 2 1, 3 PD -H 2 O H Propionaldehyde +H 2 O 10 k. Pa H 2 20 k. Pa H 2 40 k. Pa H 2 3 In situ H 2 formation (WGS reaction) and 1, 3 PD 4 Effect of CO on 1, 3 PD hydrogenation C 3 H 6 O propionaldehyde OH C 3 H 8 O propanol
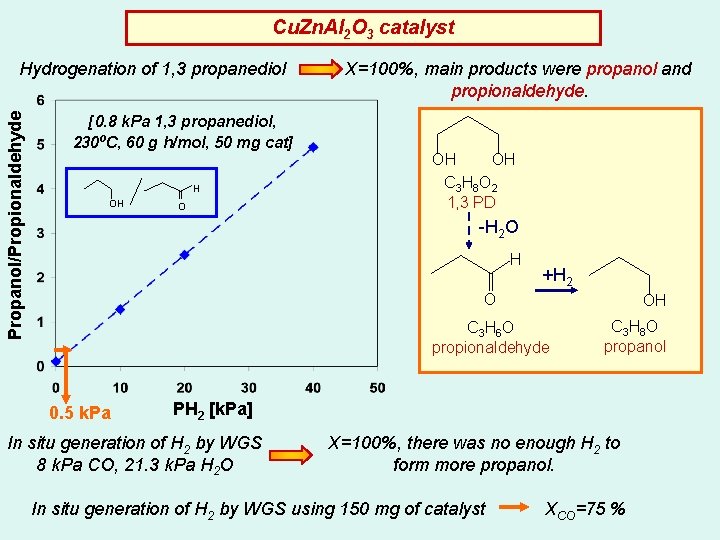
Cu. Zn. Al 2 O 3 catalyst Propanol/Propionaldehyde Hydrogenation of 1, 3 propanediol [0. 8 k. Pa 1, 3 propanediol, 230ºC, 60 g h/mol, 50 mg cat] H OH O X=100%, main products were propanol and propionaldehyde. OH OH C 3 H 8 O 2 1, 3 PD -H 2 O H +H 2 O OH C 3 H 6 O propionaldehyde 0. 5 k. Pa C 3 H 8 O propanol PH 2 [k. Pa] In situ generation of H 2 by WGS 8 k. Pa CO, 21. 3 k. Pa H 2 O X=100%, there was no enough H 2 to form more propanol. In situ generation of H 2 by WGS using 150 mg of catalyst XCO=75 %
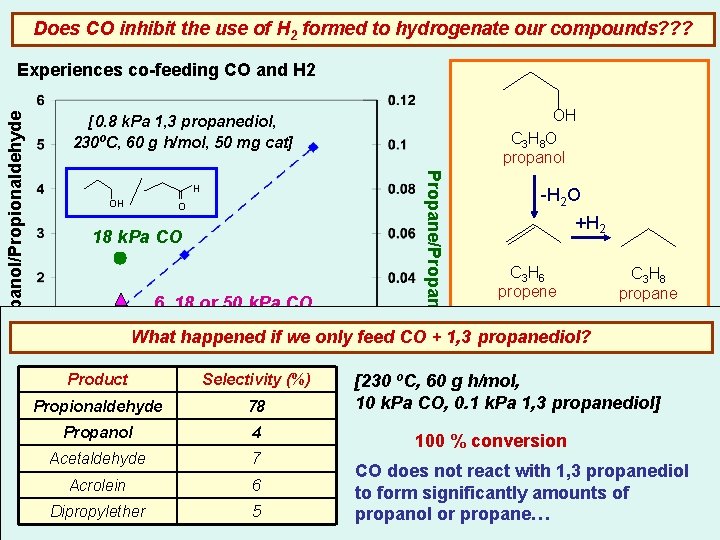
Does CO inhibit the use of H 2 formed to hydrogenate our compounds? ? ? OH C 3 H 8 O propanol [0. 8 k. Pa 1, 3 propanediol, 230ºC, 60 g h/mol, 50 mg cat] H OH O 18 k. Pa CO 6, 18 or 50 k. Pa CO Propane/Propanol/Propionaldehyde Experiences co-feeding CO and H 2 -H 2 O +H 2 C 3 H 6 propene C 3 H 8 propane 6 k. Pa CO What happened if we only feed CO + 1, 3 propanediol? OH 90 k. Pa CO Moderate amounts of CO do not inhibit the use of H 2 to Product Selectivity (%) [230 ºC, 60 g h/mol, propanol and promote 10 k. Pa CO, form 0. 1 k. Pa 1, 3 propanediol] Propionaldehyde PH [k. Pa]78 2 propane formation Propanol 4 100 % conversion Acetaldehyde 7 Acrolein 6 Dipropylether 5 CO does not react with 1, 3 propanediol to form significantly amounts of propanol or propane…
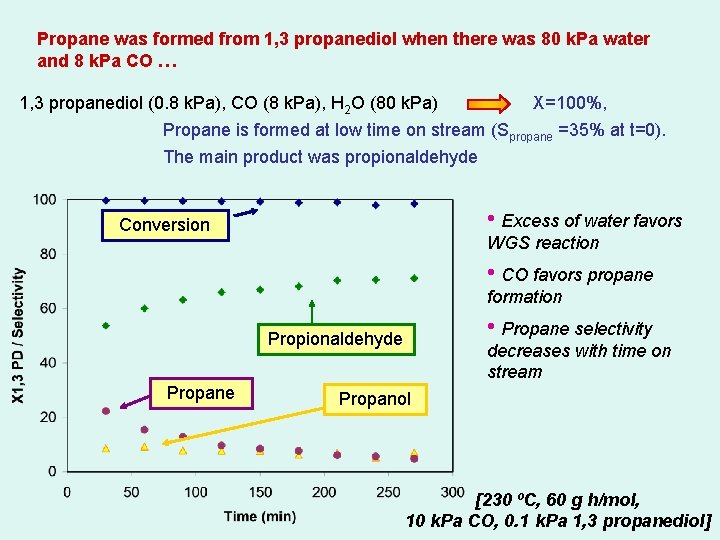
Propane was formed from 1, 3 propanediol when there was 80 k. Pa water and 8 k. Pa CO … 1, 3 propanediol (0. 8 k. Pa), CO (8 k. Pa), H 2 O (80 k. Pa) X=100%, Propane is formed at low time on stream (Spropane =35% at t=0). The main product was propionaldehyde • Excess of water favors Conversion WGS reaction • CO favors propane formation • Propane selectivity Propionaldehyde Propane decreases with time on stream Propanol [230 ºC, 60 g h/mol, 10 k. Pa CO, 0. 1 k. Pa 1, 3 propanediol]
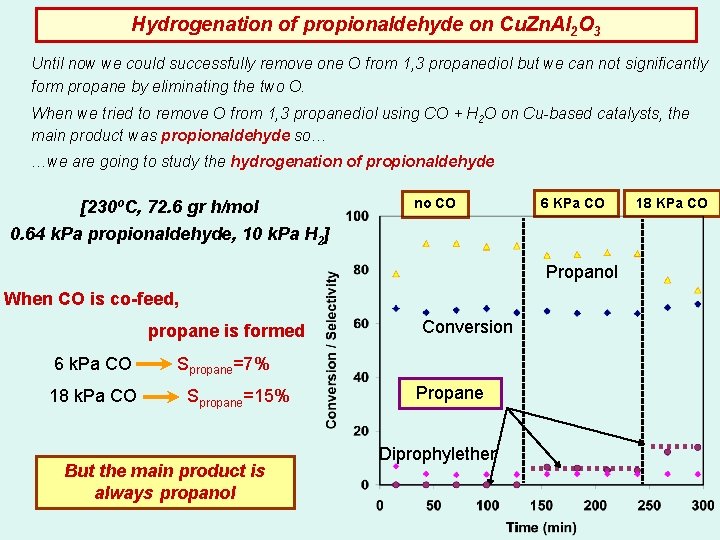
Hydrogenation of propionaldehyde on Cu. Zn. Al 2 O 3 Until now we could successfully remove one O from 1, 3 propanediol but we can not significantly form propane by eliminating the two O. When we tried to remove O from 1, 3 propanediol using CO + H 2 O on Cu-based catalysts, the main product was propionaldehyde so… …we are going to study the hydrogenation of propionaldehyde [230ºC, 72. 6 gr h/mol no CO 6 KPa CO 0. 64 k. Pa propionaldehyde, 10 k. Pa H 2] Propanol When CO is co-feed, propane is formed 6 k. Pa CO 18 k. Pa CO Conversion Spropane=7% Spropane=15% But the main product is always propanol Propane Diprophylether 18 KPa CO
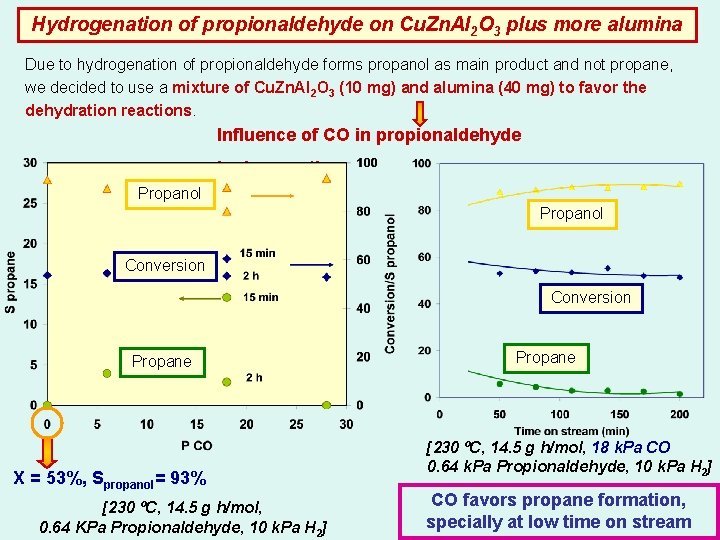
Hydrogenation of propionaldehyde on Cu. Zn. Al 2 O 3 plus more alumina Due to hydrogenation of propionaldehyde forms propanol as main product and not propane, we decided to use a mixture of Cu. Zn. Al 2 O 3 (10 mg) and alumina (40 mg) to favor the dehydration reactions. Influence of CO in propionaldehyde hydrogenation Propanol Conversion Propane X = 53%, Spropanol= 93% [230 ºC, 14. 5 g h/mol, 0. 64 KPa Propionaldehyde, 10 k. Pa H 2] Propane [230 ºC, 14. 5 g h/mol, 18 k. Pa CO 0. 64 k. Pa Propionaldehyde, 10 k. Pa H 2] CO favors propane formation, specially at low time on stream
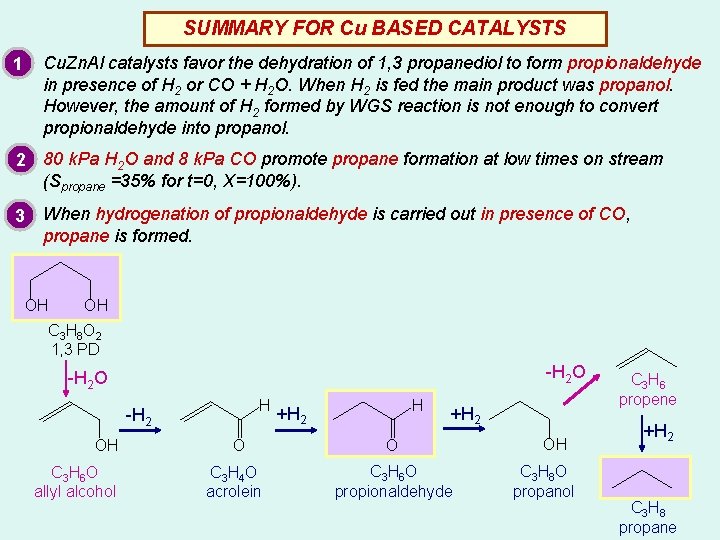
SUMMARY FOR Cu BASED CATALYSTS Cu. Zn. Al catalysts favor the dehydration of 1, 3 propanediol to form propionaldehyde in presence of H 2 or CO + H 2 O. When H 2 is fed the main product was propanol. However, the amount of H 2 formed by WGS reaction is not enough to convert propionaldehyde into propanol. 1 2 80 k. Pa H 2 O and 8 k. Pa CO promote propane formation at low times on stream (Spropane =35% for t=0, X=100%). 3 When hydrogenation of propionaldehyde is carried out in presence of CO, propane is formed. OH OH C 3 H 8 O 2 1, 3 PD -H 2 O H -H 2 OH C 3 H 6 O allyl alcohol O C 3 H 4 O acrolein H +H 2 O C 3 H 6 O propionaldehyde OH C 3 H 8 O propanol C 3 H 6 propene +H 2 C 3 H 8 propane
![Fe-Zn oxide catalyst 0. 8 k. Pa 1, 3 propanediol [300ºC, 30 g h/mol] Fe-Zn oxide catalyst 0. 8 k. Pa 1, 3 propanediol [300ºC, 30 g h/mol]](http://slidetodoc.com/presentation_image_h2/5eafea1b8e52e5eb119c388a2c67597e/image-12.jpg)
Fe-Zn oxide catalyst 0. 8 k. Pa 1, 3 propanediol [300ºC, 30 g h/mol] 1 Only 1, 3 PD X Acrolein Propanol Propane Others 25 60 30 0 10 0. 8 k. Pa 1, 3 propanediol + 10 to 40 k. Pa H 2 [300ºC, 30 g h/mol] 2 Hydrogenation of 1, 3 PD 3 1, 3 PD + CO + H 2 O 4 1, 3 PD + CO + H 2 O X Acrolein Propanol Propane Others 40 50 40 0 10 0. 8 k. Pa 1, 3 propanediol + 8 k. Pa CO + 21. 3 k. Pa H 2 O [300ºC, 30 g h/mol] X Acrolein Propanol Propane Others 25 67 23 0 10 0. 8 k. Pa 1, 3 propanediol + 8 k. Pa CO + 80 k. Pa H 2 O [300ºC, 60 g h/mol] X Acrolein Propanol Propane Others 35 (20) 35 (55) 18 (14) 35 (15) 12 (16)
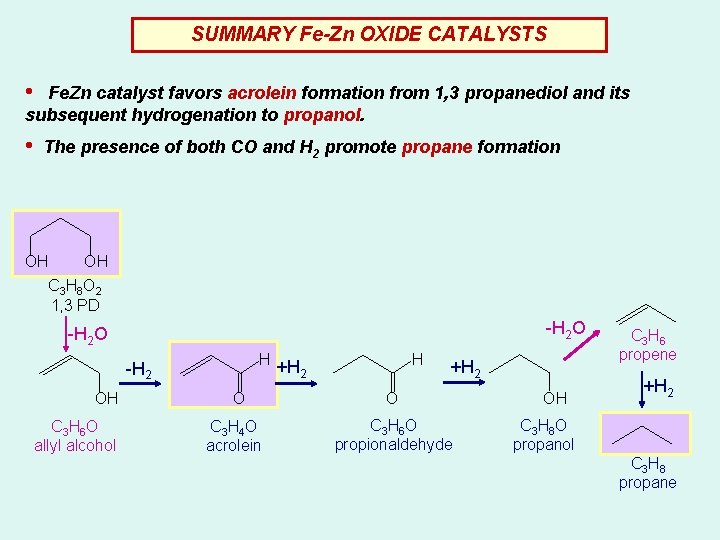
SUMMARY Fe-Zn OXIDE CATALYSTS • Fe. Zn catalyst favors acrolein formation from 1, 3 propanediol and its subsequent hydrogenation to propanol. • The presence of both CO and H 2 promote propane formation OH OH C 3 H 8 O 2 1, 3 PD -H 2 O H -H 2 OH C 3 H 6 O allyl alcohol O C 3 H 4 O acrolein H +H 2 O C 3 H 6 O propionaldehyde OH C 3 H 6 propene +H 2 C 3 H 8 O propanol C 3 H 8 propane
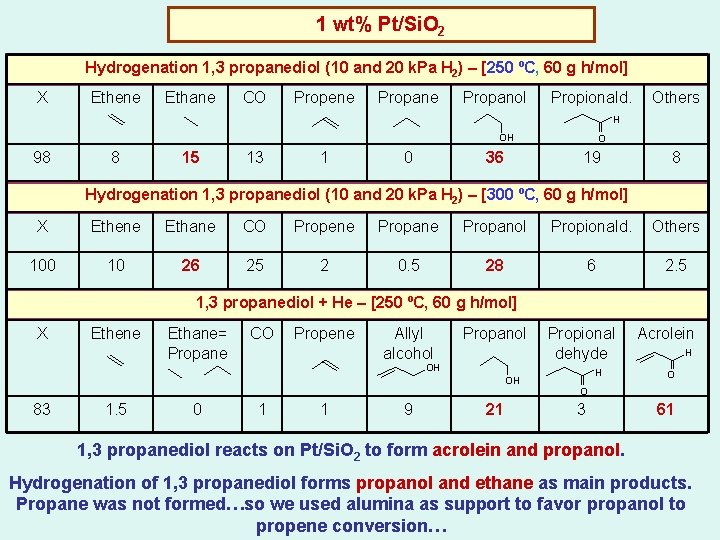
1 wt% Pt/Si. O 2 Hydrogenation 1, 3 propanediol (10 and 20 k. Pa H 2) – [250 ºC, 60 g h/mol] X Ethene Ethane CO Propene Propanol Propionald. Others H OH 98 8 15 13 1 0 36 O 19 8 Hydrogenation 1, 3 propanediol (10 and 20 k. Pa H 2) – [300 ºC, 60 g h/mol] X Ethene Ethane CO Propene Propanol Propionald. Others 100 10 26 25 2 0. 5 28 6 2. 5 1, 3 propanediol + He – [250 ºC, 60 g h/mol] X Ethene Ethane= Propane CO Propene Allyl alcohol Propanol Propional dehyde OH OH 83 1. 5 0 1 1 9 21 H Acrolein H O O 3 61 1, 3 propanediol reacts on Pt/Si. O 2 to form acrolein and propanol. Hydrogenation of 1, 3 propanediol forms propanol and ethane as main products. Propane was not formed…so we used alumina as support to favor propanol to propene conversion…
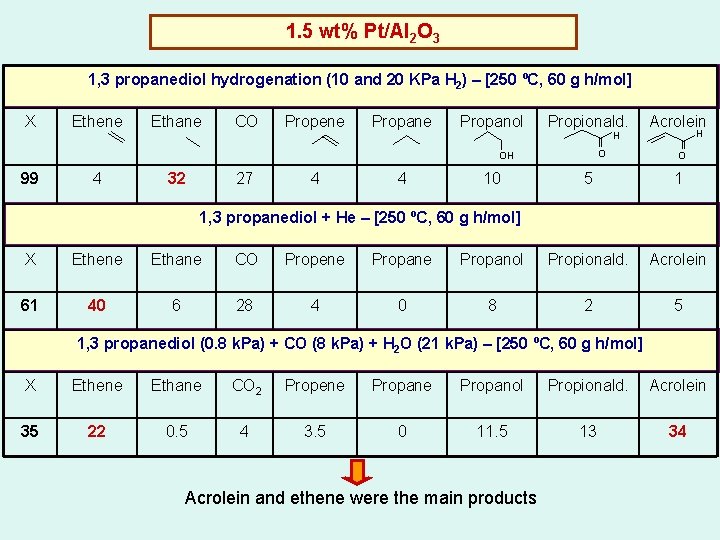
1. 5 wt% Pt/Al 2 O 3 1, 3 propanediol hydrogenation (10 and 20 KPa H 2) – [250 ºC, 60 g h/mol] X Ethene Ethane CO Propene Propanol Propionald. H O OH 99 4 32 27 4 4 10 Acrolein H O 5 1 1, 3 propanediol + He – [250 ºC, 60 g h/mol] X Ethene Ethane CO Propene Propanol Propionald. Acrolein 61 40 6 28 4 0 8 2 5 1, 3 propanediol (0. 8 k. Pa) + CO (8 k. Pa) + H 2 O (21 k. Pa) – [250 ºC, 60 g h/mol] X Ethene Ethane CO 2 Propene Propanol Propionald. Acrolein 35 22 0. 5 4 3. 5 0 11. 5 13 34 Acrolein and ethene were the main products
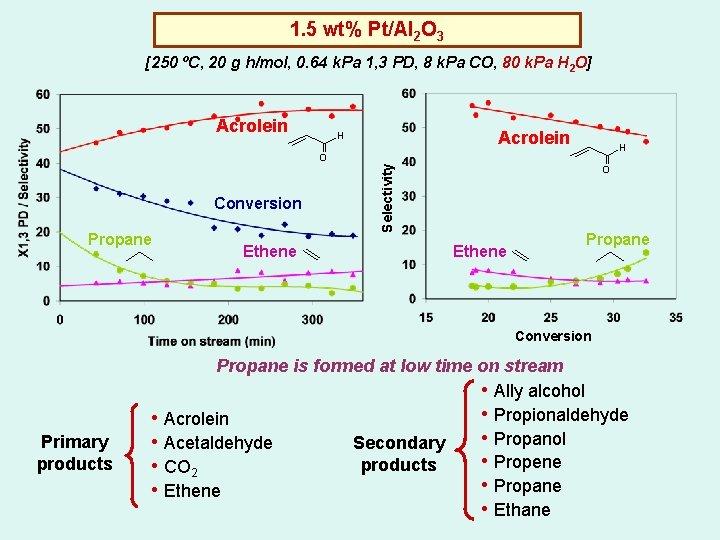
1. 5 wt% Pt/Al 2 O 3 [250 ºC, 20 g h/mol, 0. 64 k. Pa 1, 3 PD, 8 k. Pa CO, 80 k. Pa H 2 O] Acrolein H H Conversion Propane Ethene Selectivity O O Ethene Propane Conversion Primary products Propane is formed at low time on stream • Ally alcohol • Propionaldehyde • Acrolein • Propanol • Acetaldehyde Secondary • Propene products • CO 2 • Propane • Ethene • Ethane
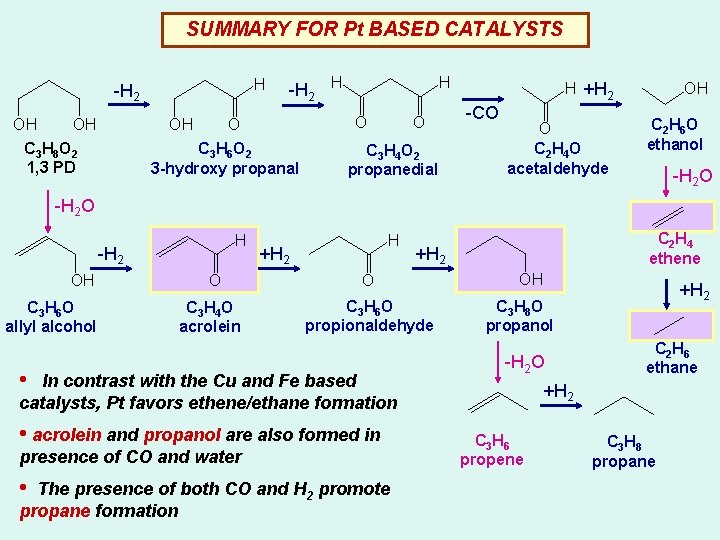
SUMMARY FOR Pt BASED CATALYSTS H -H 2 OH OH OH C 3 H 8 O 2 1, 3 PD -H 2 H H O O C 3 H 6 O 2 3 -hydroxy propanal O C 3 H 4 O 2 propanedial H +H 2 -CO O C 2 H 4 O acetaldehyde OH C 2 H 6 O ethanol -H 2 O H -H 2 OH C 3 H 6 O allyl alcohol O C 3 H 4 O acrolein H +H 2 O C 3 H 6 O propionaldehyde • In contrast with the Cu and Fe based catalysts, Pt favors ethene/ethane formation • acrolein and propanol are also formed in presence of CO and water • The presence of both CO and H 2 promote propane formation C 2 H 4 ethene +H 2 OH +H 2 C 3 H 8 O propanol -H 2 O C 2 H 6 ethane +H 2 C 3 H 6 propene C 3 H 8 propane

CONCLUSIONS AND FUTURE WORK ü We can successfully convert 1, 3 PD into propionaldehyde using Cu. Zn. Al catalysts or into acrolein using Fe. Zn or Pt catalysts. At this point we are able to remove one O from 1, 3 PD. ü So, the next step is to find a catalyst that transforms propionaldehyde or acrolein into propene or propane. ü Our results showed that propane formation from 1, 3 propanediol or propionaldehyde, is favored by using a H 2/CO co-feed mixture, however, feeding only H 2 does no produce propane. ü We are going to carry out more catalytic tests to try to understand this particular behavior.

Thank you very much for your attention!
- Slides: 19