SEIZURES IN CHILDHOOD ASSOC PROF DR SUAT BIER

SEIZURES IN CHILDHOOD ASSOC. PROF. DR. SUAT BIÇER

A 2 -year-old previously healthy child with a generalized clonic seizure is brought to the emergency department by 112. The parents estimate that he had been having a seizure for 5 minutes prior to arrival of the paramedics at the scene. Paramedics noted that the child was having a generalized seizure and was blue around the lips. The patient was placed on 100% O 2 by face mask, and a rapid assay for glucose was 80 mg/d. L. Rectal diazepam was administered and the child continues to seize on arrival in the emergency department.

What is the most important initial intervention for this patient? What medications can be used to stop the seizure? What are some of the possible etiologies of this child’s seizure?

Recurrent unprovoked seizures. Often, interictal electroencephalographic changes.

General Considerations sudden, transient disturbance of brain function manifested by involuntary motor, sensory, autonomic, or psychic phenomena, alone or in any combination, often accompanied by alteration or loss of consciousness

Seizures can be caused by any factor that disturbs brain function metabolic, traumatic, anoxic, infectious insult to the brain (classified as symptomatic seizures) or spontaneously without prior known CNS insult: • Genetic mutations
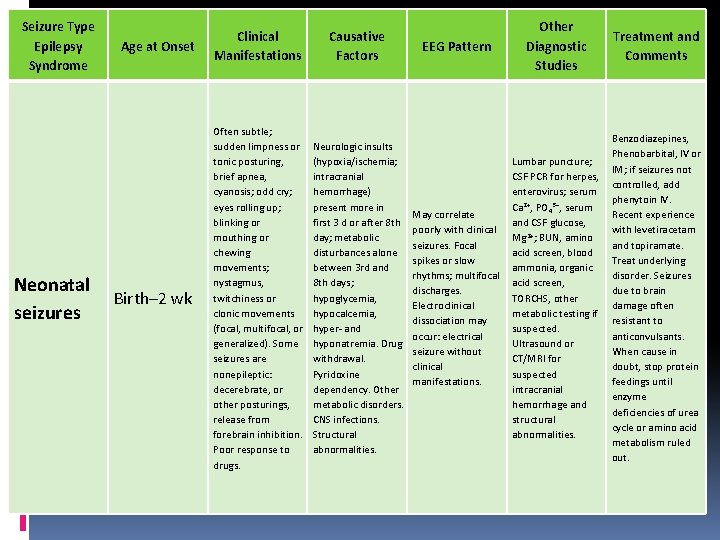
Seizure Type Epilepsy Syndrome Neonatal seizures Age at Onset Clinical Manifestations Causative Factors Birth– 2 wk Often subtle; sudden limpness or tonic posturing, brief apnea, cyanosis; odd cry; eyes rolling up; blinking or mouthing or chewing movements; nystagmus, twitchiness or clonic movements (focal, multifocal, or generalized). Some seizures are nonepileptic: decerebrate, or other posturings, release from forebrain inhibition. Poor response to drugs. Neurologic insults (hypoxia/ischemia; intracranial hemorrhage) present more in first 3 d or after 8 th day; metabolic disturbances alone between 3 rd and 8 th days; hypoglycemia, hypocalcemia, hyper- and hyponatremia. Drug withdrawal. Pyridoxine dependency. Other metabolic disorders. CNS infections. Structural abnormalities. EEG Pattern May correlate poorly with clinical seizures. Focal spikes or slow rhythms; multifocal discharges. Electroclinical dissociation may occur: electrical seizure without clinical manifestations. Other Diagnostic Studies Treatment and Comments Lumbar puncture; CSF PCR for herpes, enterovirus; serum Ca 2+, PO 43−, serum and CSF glucose, Mg 2+; BUN, amino acid screen, blood ammonia, organic acid screen, TORCHS, other metabolic testing if suspected. Ultrasound or CT/MRI for suspected intracranial hemorrhage and structural abnormalities. Benzodiazepines, Phenobarbital, IV or IM; if seizures not controlled, add phenytoin IV. Recent experience with levetiracetam and topiramate. Treat underlying disorder. Seizures due to brain damage often resistant to anticonvulsants. When cause in doubt, stop protein feedings until enzyme deficiencies of urea cycle or amino acid metabolism ruled out.
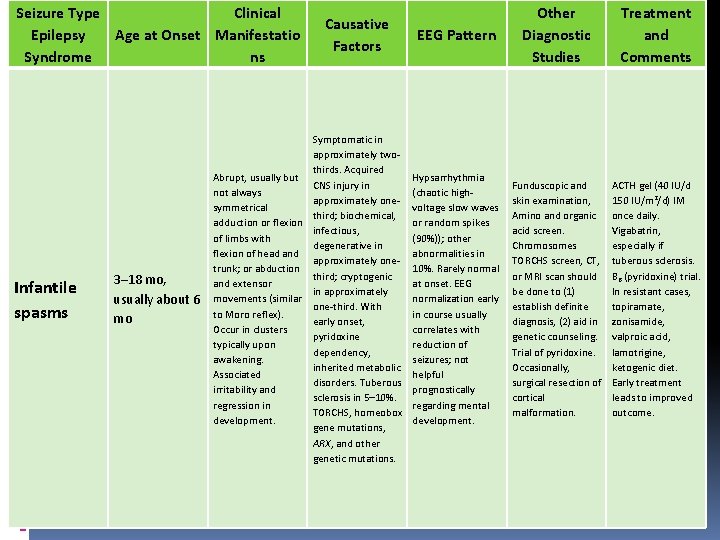
Seizure Type Clinical Epilepsy Age at Onset Manifestatio Syndrome ns Infantile spasms 3– 18 mo, usually about 6 mo Causative Factors Symptomatic in approximately twothirds. Acquired Abrupt, usually but CNS injury in not always approximately onesymmetrical third; biochemical, adduction or flexion infectious, of limbs with degenerative in flexion of head and approximately onetrunk; or abduction third; cryptogenic and extensor in approximately movements (similar one-third. With to Moro reflex). early onset, Occur in clusters pyridoxine typically upon dependency, awakening. inherited metabolic Associated disorders. Tuberous irritability and sclerosis in 5– 10%. regression in TORCHS, homeobox development. gene mutations, ARX, and other genetic mutations. EEG Pattern Other Diagnostic Studies Treatment and Comments Hypsarrhythmia (chaotic highvoltage slow waves or random spikes (90%)); other abnormalities in 10%. Rarely normal at onset. EEG normalization early in course usually correlates with reduction of seizures; not helpful prognostically regarding mental development. Funduscopic and skin examination, Amino and organic acid screen. Chromosomes TORCHS screen, CT, or MRI scan should be done to (1) establish definite diagnosis, (2) aid in genetic counseling. Trial of pyridoxine. Occasionally, surgical resection of cortical malformation. ACTH gel (40 IU/d 150 IU/m 2/d) IM once daily. Vigabatrin, especially if tuberous sclerosis. B 6 (pyridoxine) trial. In resistant cases, topiramate, zonisamide, valproic acid, lamotrigine, ketogenic diet. Early treatment leads to improved outcome.
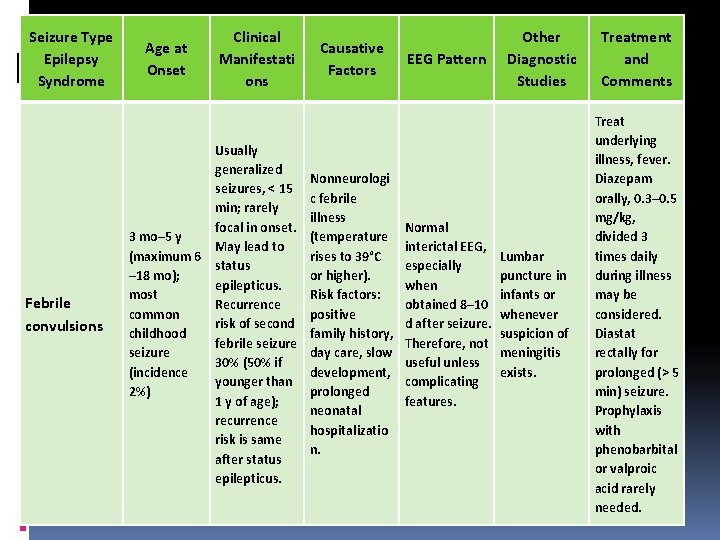
Seizure Type Epilepsy Syndrome Febrile convulsions Age at Onset Clinical Manifestati ons Usually generalized seizures, < 15 min; rarely focal in onset. 3 mo– 5 y May lead to (maximum 6 status – 18 mo); epilepticus. most Recurrence common risk of second childhood febrile seizure 30% (50% if (incidence younger than 2%) 1 y of age); recurrence risk is same after status epilepticus. Causative Factors Nonneurologi c febrile illness (temperature rises to 39°C or higher). Risk factors: positive family history, day care, slow development, prolonged neonatal hospitalizatio n. EEG Pattern Normal interictal EEG, especially when obtained 8– 10 d after seizure. Therefore, not useful unless complicating features. Other Diagnostic Studies Lumbar puncture in infants or whenever suspicion of meningitis exists. Treatment and Comments Treat underlying illness, fever. Diazepam orally, 0. 3– 0. 5 mg/kg, divided 3 times daily during illness may be considered. Diastat rectally for prolonged (> 5 min) seizure. Prophylaxis with phenobarbital or valproic acid rarely needed.
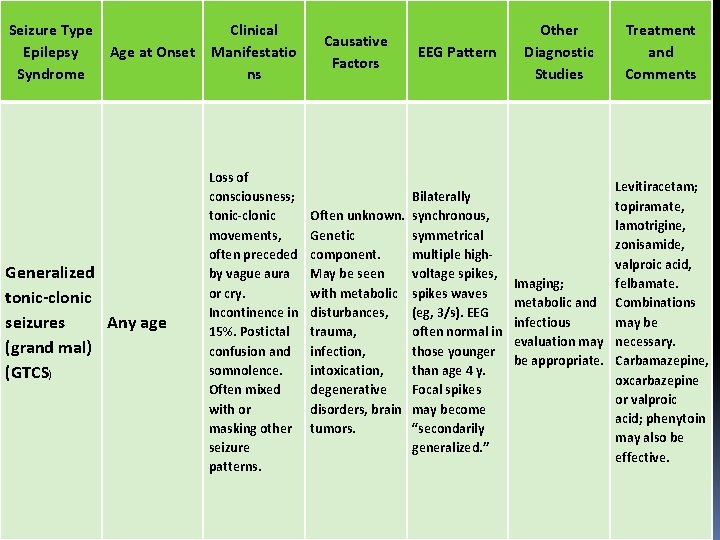
Seizure Type Epilepsy Syndrome Age at Onset Generalized tonic-clonic seizures Any age (grand mal) (GTCS) Clinical Manifestatio ns Loss of consciousness; tonic-clonic movements, often preceded by vague aura or cry. Incontinence in 15%. Postictal confusion and somnolence. Often mixed with or masking other seizure patterns. Causative Factors EEG Pattern Bilaterally Often unknown. synchronous, Genetic symmetrical component. multiple high. May be seen voltage spikes, with metabolic spikes waves disturbances, (eg, 3/s). EEG trauma, often normal in infection, those younger intoxication, than age 4 y. degenerative Focal spikes disorders, brain may become tumors. “secondarily generalized. ” Other Diagnostic Studies Treatment and Comments Levitiracetam; topiramate, lamotrigine, zonisamide, valproic acid, Imaging; felbamate. metabolic and Combinations infectious may be evaluation may necessary. be appropriate. Carbamazepine, oxcarbazepine or valproic acid; phenytoin may also be effective.
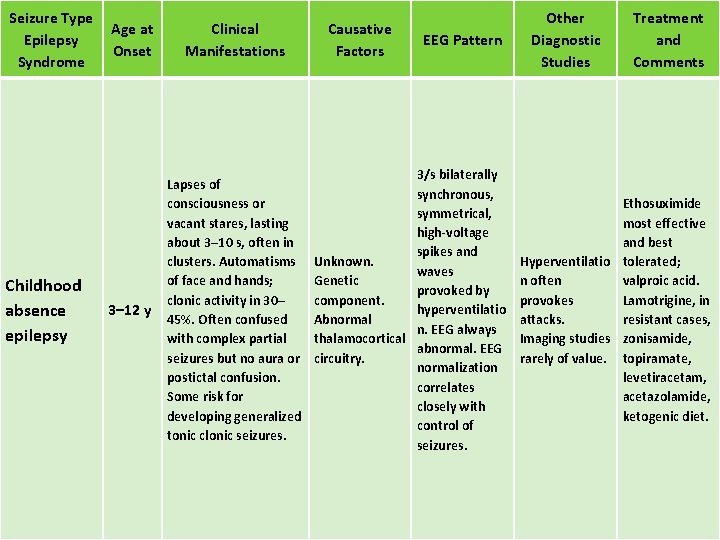
Seizure Type Epilepsy Syndrome Childhood absence epilepsy Age at Onset Clinical Manifestations 3– 12 y Lapses of consciousness or vacant stares, lasting about 3– 10 s, often in clusters. Automatisms of face and hands; clonic activity in 30– 45%. Often confused with complex partial seizures but no aura or postictal confusion. Some risk for developing generalized tonic clonic seizures. Causative Factors EEG Pattern 3/s bilaterally synchronous, symmetrical, high-voltage spikes and Unknown. waves Genetic provoked by component. hyperventilatio Abnormal n. EEG always thalamocortical abnormal. EEG circuitry. normalization correlates closely with control of seizures. Other Diagnostic Studies Treatment and Comments Ethosuximide most effective and best Hyperventilatio tolerated; n often valproic acid. provokes Lamotrigine, in attacks. resistant cases, Imaging studies zonisamide, rarely of value. topiramate, levetiracetam, acetazolamide, ketogenic diet.
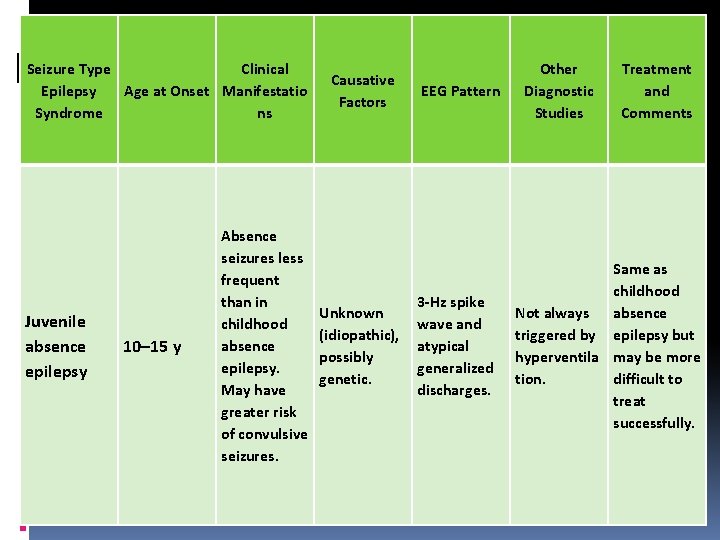
Seizure Type Clinical Epilepsy Age at Onset Manifestatio Syndrome ns Juvenile absence epilepsy 10– 15 y Absence seizures less frequent than in childhood absence epilepsy. May have greater risk of convulsive seizures. Causative Factors Unknown (idiopathic), possibly genetic. EEG Pattern 3 -Hz spike wave and atypical generalized discharges. Other Diagnostic Studies Treatment and Comments Same as childhood Not always absence triggered by epilepsy but hyperventila may be more tion. difficult to treat successfully.
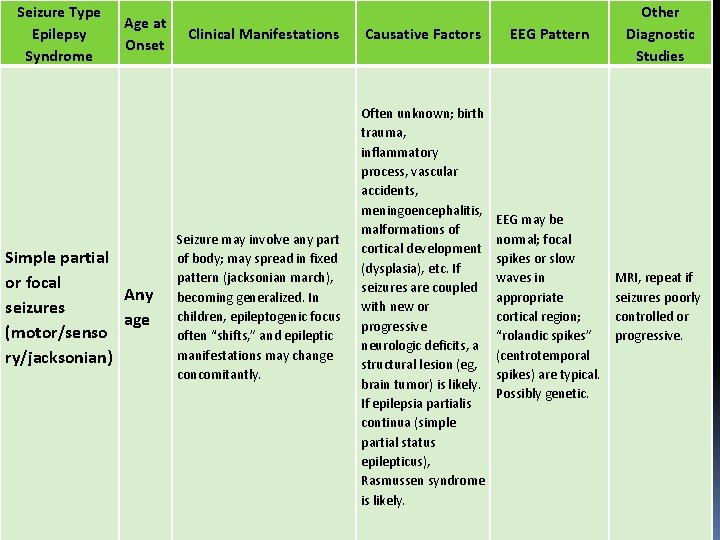
Seizure Type Epilepsy Syndrome Age at Onset Simple partial or focal Any seizures age (motor/senso ry/jacksonian) Clinical Manifestations Causative Factors EEG Pattern Other Diagnostic Studies Seizure may involve any part of body; may spread in fixed pattern (jacksonian march), becoming generalized. In children, epileptogenic focus often “shifts, ” and epileptic manifestations may change concomitantly. Often unknown; birth trauma, inflammatory process, vascular accidents, meningoencephalitis, malformations of cortical development (dysplasia), etc. If seizures are coupled with new or progressive neurologic deficits, a structural lesion (eg, brain tumor) is likely. If epilepsia partialis continua (simple partial status epilepticus), Rasmussen syndrome is likely. EEG may be normal; focal spikes or slow waves in appropriate cortical region; “rolandic spikes” (centrotemporal spikes) are typical. Possibly genetic. MRI, repeat if seizures poorly controlled or progressive.
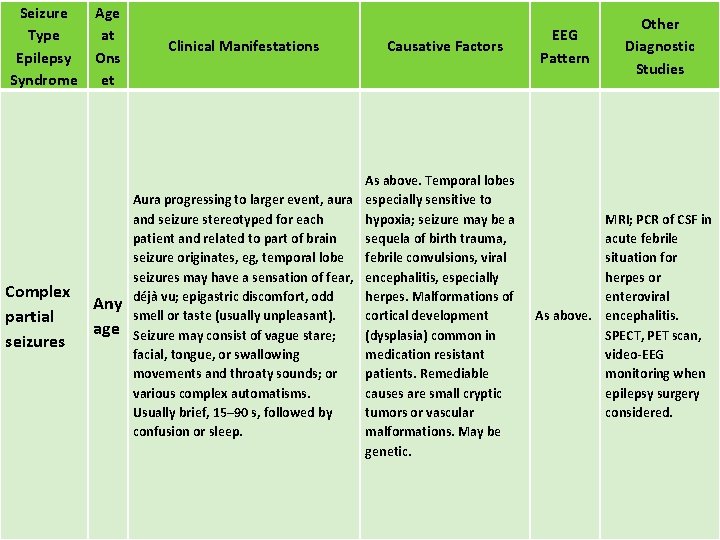
Seizure Type Epilepsy Syndrome Complex partial seizures Age at Ons et Any age Clinical Manifestations Causative Factors As above. Temporal lobes Aura progressing to larger event, aura especially sensitive to and seizure stereotyped for each hypoxia; seizure may be a patient and related to part of brain sequela of birth trauma, seizure originates, eg, temporal lobe febrile convulsions, viral seizures may have a sensation of fear, encephalitis, especially déjà vu; epigastric discomfort, odd herpes. Malformations of cortical development smell or taste (usually unpleasant). Seizure may consist of vague stare; (dysplasia) common in facial, tongue, or swallowing medication resistant movements and throaty sounds; or patients. Remediable causes are small cryptic various complex automatisms. Usually brief, 15– 90 s, followed by tumors or vascular confusion or sleep. malformations. May be genetic. EEG Pattern Other Diagnostic Studies MRI; PCR of CSF in acute febrile situation for herpes or enteroviral As above. encephalitis. SPECT, PET scan, video-EEG monitoring when epilepsy surgery considered.
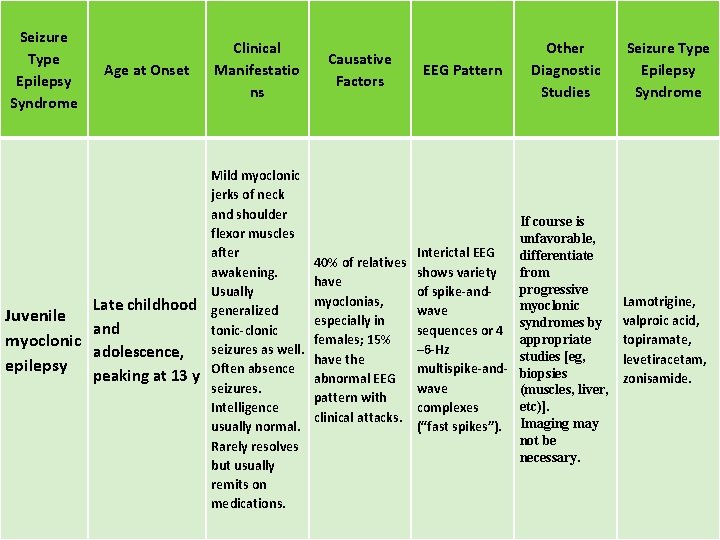
Seizure Type Epilepsy Syndrome Age at Onset Late childhood Juvenile and myoclonic adolescence, epilepsy peaking at 13 y Clinical Manifestatio ns Mild myoclonic jerks of neck and shoulder flexor muscles after awakening. Usually generalized tonic-clonic seizures as well. Often absence seizures. Intelligence usually normal. Rarely resolves but usually remits on medications. Causative Factors EEG Pattern Other Diagnostic Studies If course is unfavorable, Interictal EEG differentiate 40% of relatives shows variety from have progressive of spike-andmyoclonias, myoclonic wave especially in syndromes by sequences or 4 appropriate females; 15% – 6 -Hz studies [eg, have the multispike-and- biopsies abnormal EEG wave (muscles, liver, pattern with etc)]. complexes clinical attacks. (“fast spikes”). Imaging may not be necessary. Seizure Type Epilepsy Syndrome Lamotrigine, valproic acid, topiramate, levetiracetam, zonisamide.

Clinical Findings Seizures are stereotyped paroxysmal clinical events; the key to diagnosis is usually in the history. Not all paroxysmal events are epileptic. A detailed description of seizure onset is important in determining if an event is a seizure and if there is localized onset (partial or focal seizure). Events prior to, during, and after the seizure need to be described (although observers often recall little except generalized convulsive activity because of its dramatic appearance). An aura (actually a simple partial seizure) may precede the clinically apparent seizure and indicates focal onset. The patient may describe a feeling of fear, numbness or tingling in the fingers, or bright lights in one visual field. The specific symptoms may help define the location of seizure onset (eg, déjà vu suggests temporal lobe onset).

Diagnostic Evaluation the child’s age, the severity and type of seizure, the child is ill or injured, the clinician’s suspicion about the underlying cause. Seizures in early infancy are often symptomatic. Therefore, the younger the child, the more extensive must be the diagnostic assessment
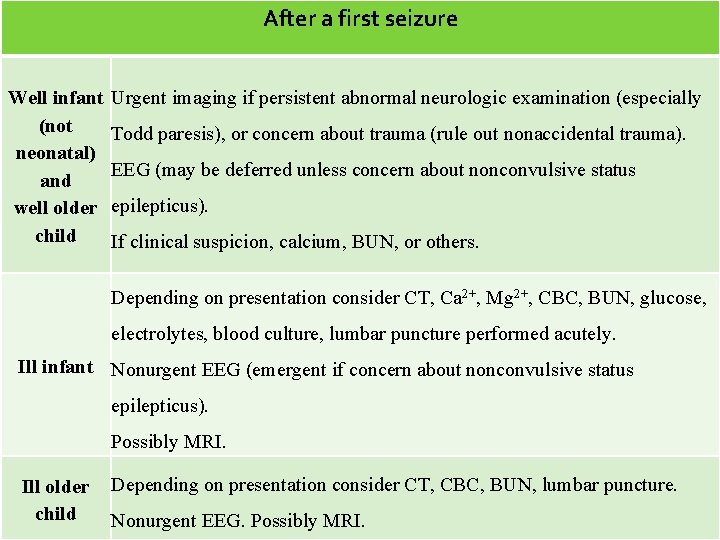
After a first seizure Well infant (not neonatal) and well older child Urgent imaging if persistent abnormal neurologic examination (especially Todd paresis), or concern about trauma (rule out nonaccidental trauma). EEG (may be deferred unless concern about nonconvulsive status epilepticus). If clinical suspicion, calcium, BUN, or others. Depending on presentation consider CT, Ca 2+, Mg 2+, CBC, BUN, glucose, electrolytes, blood culture, lumbar puncture performed acutely. Ill infant Nonurgent EEG (emergent if concern about nonconvulsive status epilepticus). Possibly MRI. Ill older child Depending on presentation consider CT, CBC, BUN, lumbar puncture. Nonurgent EEG. Possibly MRI.

After a first seizure Practice parameter: EEG only. Generalized tonicclonic seizure Circumstances may suggest need for toxicology screen, calcium, glucose, CBC, BUN, electrolytes, lumbar puncture, and others if concern about infection (especially HSV).

For new-onset epilepsy (2 or more unprovoked seizures) Imaging (MRI is preferred study); EEG. Consider evaluation for inherited metabolic disorder: lumbar Infant (< 1 y) puncture including CSF (and serum) glucose, lactate, glycine; serum and urine amino and organic acids; high-resolution chromosome analysis; other genetic testing. EEG; imaging (MRI). Older child Other evaluation dependant on epilepsy syndrome. Consider CBC and AST before initiation of treatment. Childhood absence seizure EEG only.

For new-onset epilepsy (2 or more unprovoked seizures) EEG. Symptomatic/cryptogenic generalized epilepsy (Lennox-Gastaut and variants; Dravet syndrome and other myoclonic epilepsies) Evaluation for causes of mental retardation: MRI; serum and urine amino and organic acids; chromosomal microarray vs high-resolution chromosome analysis; other genetic testing as indicated per patient, consider biopsy (skin, conjunctiva) for neuronal ceroid lipofuscinosis (NCL). Consider lysosomal enzymes, lumbar puncture including glucose (CSF and serum) lactate, glycine, neurotransmitters, long-chain fatty acids, skin or conjunctival biopsies. Consider tuberous sclerosis complex (including Wood lamp, MRI, renal Infantile spasms and cardiac scans). Evaluation as for symptomatic generalized epilepsy.

For new-onset epilepsy (2 or more unprovoked seizures) EEG including video-EEG monitoring if Cryptogenic focal epilepsy necessary for clarification of diagnosis. MRI with attention to possible focal cortical dysplasia. Idiopathic focal epilepsy (BECTS = benign epilepsy with EEG. centrotemporal spikes)

Electroencephalography++ A routine EEG captures electrical activity during a very short period of time, usually 20– 30 minutes. Thus, it is useful primarily for defining interictal activity (except for the fortuitous recording of a clinical seizure or in the case seizures that are easily provoked such as childhood absence epilepsy). A seizure is a clinical phenomenon; an EEG showing epileptiform activity may confirm and clarify the clinical diagnosis (for instance, defining an epilepsy syndrome), but it is only occasionally diagnostic.

Diagnostic Value The greatest value of the EEG in convulsive disorders is to help classify seizure types and epilepsy syndromes. This can aid in prognostication and in selecting appropriate therapy The differing EEG patterns of these seizures will then prove most helpful. The presence of a mixed seizure EEG pattern in a child with clinically generalized convulsive seizures or only focal seizures may lead to identification of specific epilepsy syndromes and help the clinician select anticonvulsants effective for the seizure types identified by the EEG Similarly, the EEG may help in diagnosing seizures in a young infant with minimal or atypical clinical manifestations; it may show hypsarrhythmia (high-amplitude spikes and slow waves with a chaotic background) in infantile spasms or the 1– 4/s slow spike-wave pattern of the Lennox-Gastaut syndrome. The EEG may show focal slowing that, if constant, particularly when corresponding focal seizure manifestations and abnormal neurologic findings, will alert the physician to the presence of a structural lesion. In this case, brain imaging may establish the cause and help determine further investigation and treatment.

The EEG need not be abnormal in a child with epilepsy. Normal EEGs are seen following a first generalized seizure in one-third of children younger than age 4 years. The initial EEG is normal in about 20% of older children with epilepsy and in about 10% of adults with epilepsy. These percentages are reduced when serial tracings are obtained especially if sleep-deprived. Focal spikes and generalized spike-wave discharges are seen in 30% of close nonepileptic relatives of patients with epilepsy.

Prognostic Value EEG following febrile seizures is almost always normal and is not clearly predictive of subsequent seizures and therefore is not useful in these situations. Hypsarrhythmia or slow spike and wave patterns support the diagnosis of infantile spasms and Lennox-Gastaut syndrome, respectively. Both are expressions of diffuse brain dysfunction (epileptic encephalopathy) and are generally of grave significance. Central-temporal (rolandic spikes) and occipital spike-wave activity (occipital paroxysms) are the EEG correlates of idiopathic focal epilepsies of childhood. Following successful treatment, abnormal EEGs may become normal and may aid in the decision to discontinue medications. The normalization of the EEG in childhood absence epilepsy confirms the efficacy of treatment. Normalization can also be seen in infants with infantile spasms who have been successfully treated and, less commonly, in children with epileptic encephalopathies.

EEG should be repeated when the severity and frequency of seizures increase despite adequate anticonvulsant therapy, when the clinical seizure pattern changes significantly, or when progressive neurologic deficits develop. Emergence of new focal or diffuse slowing may indicate a progressive lesion or neurodegenerative disorder.

discontinue anticonvulsant therapy The EEG may be helpful in determining when to discontinue anticonvulsant therapy. The presence or absence of epileptiform activity on the EEG prior to withdrawal of anticonvulsants after a seizure-free period of 2 years on medications has been shown to correlate with the degree of risk of seizure recurrence. However, persistent focal epileptiform discharges are common in children with so-called benign epilepsies until they resolve spontaneously in adolescence.

Differential Diagnosis Misinterpretation of behaviors in children is the most common reason for misdiagnosis. Psychogenic seizures are much less common in children than in adults but must be considered even in the young or cognitively impaired child. The most commonly misinterpreted behaviors are inattention in school-aged children with attention disorders, stereotypies in children with autistic spectrum disorder, sleep-related movements, habit movements such as head-banging and socalled infantile masturbation (sometimes referred to as gratification movements), and gastroesophageal reflux in very young (often impaired) infants.

Nonepileptic Paroxysmal Events Breath-holding attacks (cyanotic and pallid) Tics (Tourette syndrome) Gastroesophageal reflux (Sandifer syndrome) Infantile masturbation/gratification movements Parasomnias (night terrors, sleep talking, walking, “sit-ups”) Conversion reaction/psychogenic nonepileptic seizures Nightmares Migraine Benign paroxysmal vertigo Benign nocturnal myoclonus Staring spells Shuddering

Complications & Sequelae Psychosocial Impact Cognitive Delay Injury and Death

Treatment The ideal treatment of acute seizures is the correction of specific causes. However, even when a biochemical disorder, a tumor, meningitis, or another specific cause is being treated, anticonvulsant drugs are often still required.

First Aid Caregivers should be instructed to protect the patient against self-injury. Turning the child to the side is useful for preventing aspiration. Thrusting a spoon handle, tongue depressor, or finger into the clenched mouth of a convulsing patient or trying to restrain tonic-clonic movements may cause worse injuries than a bitten tongue or bruised limb and could potentially become a choking hazard. Parents are often concerned that cyanosis will occur during generalized convulsive seizures but it is rare for clinically significant hypoxia to occur. Mouth-to-mouth resuscitation is rarely necessary and is unlikely to be effective

For prolonged seizures (> 5 minutes), acute home treatment with benzodiazepines such as rectal diazepam gel or intranasal midazolam may be administered to prevent the development of status epilepticus. Rectal diazepam has proven to be safe even when administered by nonmedical professionals, including teachers and day care providers, when appropriately instructed.

The child with a single seizure has a 50% chance of recurrence. The seizure type and epilepsy syndrome as well as potential side effects will determine which drug to initiate. Start with one drug in moderate dosage and increase the dosage until seizures are controlled. If seizures are not controlled on the maximal tolerated dosage of one major AED, gradually switch to another before using two-drug therapy. Polytherapy (ie, the use of more than two medications concurrently) is rarely sufficiently effective to warrant the considerable risk of adverse side effects from the synergistic impact of multiple medications.

Follow-Up Regular follow-up evaluations of the patient are appropriate at intervals that depend on the degree of control, underlying cause of the seizures, and toxic properties of the AEDs used. Blood counts and liver function tests (aspartate aminotransferase; AST) are often obtained periodically in patients taking the older AEDs, such as valproate, phenytoin, and carbamazepine. Routine determination of serum AED levels is not necessary in patients with controlled seizures but levels should be obtained to direct care, for instance, confirming adherence to medication recommendations. Periodic neurologic reevaluation is important. Repeat EEGs are useful for confirming seizure-freedom in children with absence of epilepsy and West syndrome, but otherwise are usually not useful as measures of medication efficacy.

Status Epilepticus SE is usually defined as a clinical or electrical seizure lasting at least 15 minutes, or a series of seizures without complete recovery over a 30 -minute period. After 30 minutes of seizure activity, hypoxia and acidosis occur, with depletion of energy stores, cerebral edema, and structural damage. Eventually, high fever, hypotension, respiratory depression, and even death may occur. Aggressive treatment of prolonged seizures may prevent development SE. It is generally recommended that treatment with benzodiazepines at home for prolonged seizures be initiated, 5 minutes after onset of a seizure.

SE is classified as: (1) convulsive (the common tonic-clonic, or grand mal, status epilepticus) (2) nonconvulsive (characterized by altered mental status or behavior with subtle or absent motor components). Absence status, or spike-wave stupor, and complex partial SE are examples of the nonconvulsive type. An EEG may be necessary to aid in diagnosing nonconvulsive status because patients sometimes appear merely stuporous and lack typical convulsive movements.

A child with SE may have a high fever with or without intracranial infection. SE can be a reflection of an acute or remote insult. Tumor and stroke, which are common causes of status epilepticus in adults, are uncommon causes in childhood. Fifty percent of pediatric status epilepticus is due to acute (25%) or chronic (25%) CNS disorders. Infection and metabolic disorders are common causes of status epilepticus in children. The cause is unknown in 50% of patients.

Treatment ABCs �Airway: maintain oral airway; intubation may be necessary. �Breathing: oxygen. �Circulation: assess pulse, blood pressure; support with IV fluids, drugs. Monitor vital signs. Start glucose-containing IV (unless patient is on ketogenic diet); evaluate serum glucose; electrolytes, CBC, BUN, anticonvulsant levels. Consider arterial blood gases, p. H. Give 50% glucose if serum glucose low (1– 2 m. L/kg).

Begin IV drug therapy; goal is to control status epilepticus in 20– 60 min. �Diazepam, 0. 3– 0. 5 mg/kg over 1– 5 min (20 mg max); may repeat in 5 – 20 min; or lorazepam, 0. 05– 0. 2 mg/kg (less effective with repeated doses, longer-acting than diazepam); or midazolam: IV, 0. 1– 0. 2 mg/kg; intranasally, 0. 2 mg/kg. �Phenytoin, 10– 20 mg/kg IV (not IM) over 5– 20 min; (1000 mg maximum); monitor with blood pressure and ECG. Fosphenytoin may be given more rapidly in the same dosage and can be given IM; order 10– 20 mg/kg of “phenytoin equivalent” (PE) �Phenobarbital, 5– 20 mg/kg (sometimes higher in newborns or refractory status in intubated patients).

Correct metabolic perturbations (eg, lowsodium, acidosis). Administer fluids judiciously.

Other drug approaches in refractory status: �Repeat phenytoin, phenobarbital (10 mg/kg). Monitor blood levels. Support respiration, blood pressure as necessary. �Other medications: Valproate sodium, available as 100 mg/m. L for IV use; give 15– 30 mg/kg over 5– 20 min. �Levetiracetam may be helpful (20– 40 mg/kg/dose IV) �For patients who fail initial intervention consider: midazolam drip: 1– 5 mcg/kg/min (even to 20 kg/min); pentobarbital coma; Propofol and general anesthetic.

Consider underlying causes: �Structural disorders or trauma: MRI or CT scan. �Infection: lumbar puncture, blood culture, antibiotics. �Metabolic disorders: consider lactic acidosis, toxins, and uremia if child is being treated with chronic AEDs, obtain medication levels. Toxin screen.

Initiate maintenance drug treatment with IV medications: phenytoin (10 mg/kg); phenobarbital (5 mg/kg); valproate IV 30 mg/kg; levetiracetam 20– 30 mg/kg/. Transition to oral medication when patient can safely take them.

Febrile Seizures (1) age 3 months to 6 years (most occur between ages 6 and 18 months), (2) fever of greater than 38. 8°C, and (3) non-CNS infection. More than 90% of febrile seizures are generalized, last less than 5 minutes, and occur early in the illness causing the fever.

Febrile seizures occur in 2%– 3% of children. Acute respiratory illnesses are most commonly associated with febrile seizures. Gastroenteritis, especially when caused by Shigella or Campylobacter, and urinary tract infections are less common causes. Roseola infantum is a rare but classic cause. One study implicated viral causes in 86% of cases. Immunizations may be a cause. It is very important, especially in younger children, to exclude CNS infection as a source; these children are not classified as having a febrile seizure

Rarely febrile seizures may present with status epilepticus. Febrile seizures rarely (1%– 3%) lead to recurrent unprovoked seizures (epilepsy) in later childhood and adult life (risk is increased two- to fivefold compared with children who do not have febrile seizures). The chance of later epilepsy is higher if the febrile seizures have complex features, such as duration longer than 15 minutes, more than one seizure in the same day, or focal features. Other adverse factors are an abnormal neurologic status preceding the seizures (eg, cerebral palsy or mental retardation), early onset of febrile seizure (before age 1 year), and a family history of epilepsy. Even with adverse factors, the risk of epilepsy after febrile seizures is still only in the range of 15%– 20%, although it is increased if more than one risk factor is present. Recurrent febrile seizures occur in 30%– 50% of cases. Therefore, families should be prepared to expect more seizures. In general, recurrence of febrile seizures does not worsen the long-term outlook

Abend NS, Gutierrez-Colina AM, Dlugos DJ: Medical treatment of pediatric status epilepticus. Semin Pediatr Neurol 2010; 17: 169– 175. [Pub. Med: 20727486] ++ Aldenkamp AP et al: Optimizing therapy of seizures in children and adolescents with ADHD. Neurology 2006; 67(Suppl 4): S 49. [Pub. Med: 17190923] ++ Arya R, Kabra M, Gulati S: Epilepsy in children with Down syndrome. Epileptic Disord 2011; 13(1): 1– 7. [Pub. Med: 21398208] ++ Bartha AI et al: Neonatal seizures: Multicenter variability in current treatment practices. Pediatr Neurol 2007; 37: 85. [Pub. Med: 17675022] ++ Berg AT, Berkovic SF, Brodie MT: Revised terminology and concepts for organization of seizures and epilepsies: Report of the ILAE Commission on Classification and Terminology, 2005– 2009. Epilepsia 2010; 51: 676– 685 [PMID: 20196795]. ++ Bough KJ, Rho JM: Anticonvulsant mechanisms of the ketogenic diet. Epilepsia 2007; 48: 43. [Pub. Med: 17241207] ++ Braakman HM, Vaessen MJ, Hofman PA et al: Cognitive and behavioral complications of frontal lobe epilepsy in children: A review of the literature. Epilepsia 2011; 52: 849– 856. [Pub. Med: 21480882] ++ Bruhn K et al: Screen sensitivity in photosensitive children and adolescents: Patient-dependant and stimulus-dependant factors. Epileptic Disord 2007; 9: 57. [Pub. Med: 17307713] ++ Caraballo RH et al: Ketogenic diet in patients with myoclonic-astatic epilepsy. Epileptic Disord 2006; 8: 151. [Pub. Med: 16793577] ++ Ceulemans B: Overall management of patients with Dravet syndrome. Dev Med Child Neurol 2011; 53(Suppl 2): 19– 23. [Pub. Med: 21504428] ++ Chahine LM, Mikati MA: Benign pediatric localization-related epilepsies. Epileptic Disord 2006; 8: 243. [Pub. Med: 17150437] ++ Chin RF et al: Meningitis is a common cause of convulsive status epilepticus with fever. Arch Dis Child 2005; 90: 66. [Pub. Med: 15613516] ++ Chu-Shore CJ, Thiele EA: Semin New drugs for pediatric epilepsy. Pediatr Neurol. 2010; 17: 214– 223. [Pub. Med: 21183127] ++ Deltour L et al: Children with benign epilepsy with centrotemporal spikes (BECTS) show impaired attentional control: Evidence from an attentional capture paradigm. Epileptic Disord 2007; 9: 32. [Pub. Med: 17307709] ++ Desai J, Mitchell WG: Does one more medication help? Effect of adding another anticonvulsant in childhood epilepsy. J Child Neurol 2011; 26: 329– 333. [Pub. Med: 21183723] ++ Hughes JR: Benign epilepsy of childhood with centrotemporal spikes (BECTS): To treat or not to treat, that is the question. Epilepsy Behav 2010; 19: 197– 203. [Pub. Med: 20797913] ++ Ostrowsky K: Outcome and prognosis of status epilepticus in children. Semin Pediatr Neurol 2010; 17: 195 [PMID: 20727490]. ++ Dion MH et al: Lamotrigine therapy of epilepsy with Angelman’s syndrome. Epilepsia 2007; 48: 593. [Pub. Med: 17326790] ++ Evans D et al: Anticonvulsants for preventing mortality and morbidity in full-term newborns with perinatal asphyxia. Cochrane Database Syst Rev 2007: CD 001240. [Pub. Med: 17636659] ++ Ferrie C et al: Panayiotopoulos syndrome: A consensus view. Dev Med Child Neurol 2006; 48: 236. [Pub. Med: 16483404] ++ Freeman JM, Kossoff EH: Ketosis and the ketogenic diet, 2010: Advances in treating epilepsy and other disorders. Adv Pediatr 2010; 57: 315– 329. [Pub. Med: 21056745] ++ Fischer JW, Cho CS: Pediatric syncope: Cases from the emergency department. Emerg Med Clin North Am 2010; 28: 501– 516. [Pub. Med: 20709241] ++ Gerstner T et al: Valproic acid induced encephalopathy– 19 new cases in Germany from 1994 to 2003–a side effect associated to VPA-therapy not only in young children. Seizure 2006; 15: 443. [Pub. Med: 16787750] ++ Glauser TA et al: Pharmacokinetics of levetiracetam in infants and young children with epilepsy. Epilepsia 2007; 48: 1117. [Pub. Med: 17442002] ++ Glauser TA et al: Topiramate monotherapy in newly diagnosed epilepsy in children and adolescents. J Child Neurol 2007; 22: 693. [Pub. Med: 17641254] ++ Grosso S et al: Efficacy and safety of levetiracetam in infants and young children with refractory epilepsy. Seizure 2007; 16: 345. [Pub. Med: 17368928] ++ Guillet R, Kwon J: Seizure recurrence and developmental disabilities after neonatal seizures: Outcomes are unrelated to use of phenobarbital prophylaxis. J Child Neurol 2007; 22: 389. [Pub. Med: 17621516] ++ Hedera P et al: Identification of a novel locus for febrile seizures and epilepsy on chromosome 21 q 22. Epilepsia 2006; 47: 1622. [Pub. Med: 17054683] ++ Hirtz D et al: Practice parameter: Treatment of the child with a first unprovoked seizure: Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2003; 60: 166. [Pub. Med: 12552027] + Holmes GL, Stafstrom CE, Tuberous Sclerosis Study Group: Tuberous sclerosis complex and epilepsy: Recent developments and future challenges. Epilepsia 2007; 48: 617. [Pub. Med: 17386056]
- Slides: 49