Seeing is believing Practical tips on designing your
Seeing is believing? Practical tips on designing your Microscopy experiments CMI Light Microscopy Lecture Series 2015

Question: what is the best way to image my sample? Answer: Choose the right methodology …. . Question: what is the right methodology / to use?
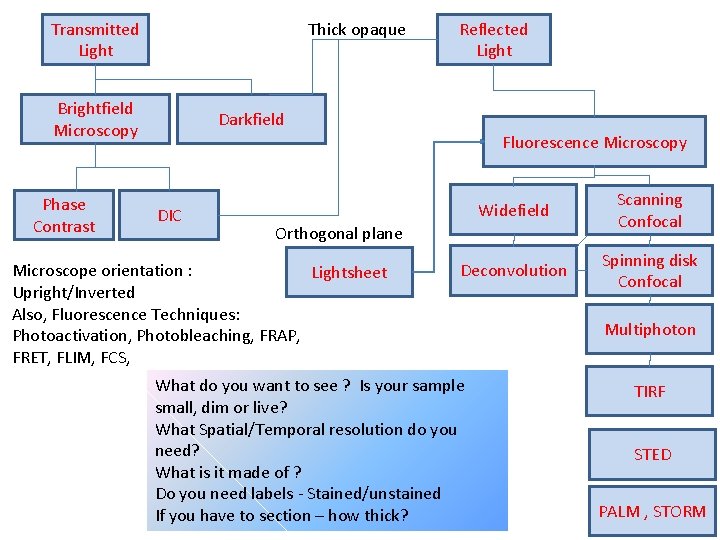
Transmitted Light Thick opaque Brightfield Microscopy Phase Contrast Reflected Light Darkfield Fluorescence Microscopy DIC Widefield Scanning Confocal Deconvolution Spinning disk Confocal Orthogonal plane Microscope orientation : Lightsheet Upright/Inverted Also, Fluorescence Techniques: Photoactivation, Photobleaching, FRAP, FRET, FLIM, FCS, What do you want to see ? Is your sample small, dim or live? What Spatial/Temporal resolution do you need? What is it made of ? Do you need labels - Stained/unstained If you have to section – how thick? Multiphoton TIRF STED PALM , STORM
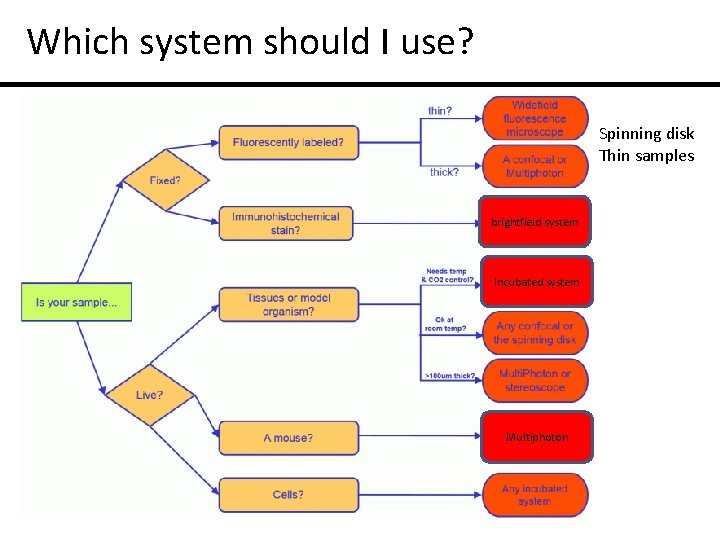
Which system should I use? Spinning disk Thin samples brightfield system Incubated system Multiphoton

Remedi systems • Upright fluorescence Microscope - volocity • Inverted transmitted light microscope and fluorescence • Upright brightfield microscope • contact the CMI for any other imaging • www. imaging. nuigalway. ie

Question: What is the best overall microscope for fluorescent microscopy applications?

• Anyone say confocal? • ok…Sharp clear images , 3 d rendering, good for bright fixed samples. • But everything in imaging is a tradeoff…. • Confocal vs widefield – which to use? • confocal microscopes are still constrained by the same diffraction limit of light as any other conventional (i. e. , non-superresolution) microscope. Obtainable Resolution is the same.
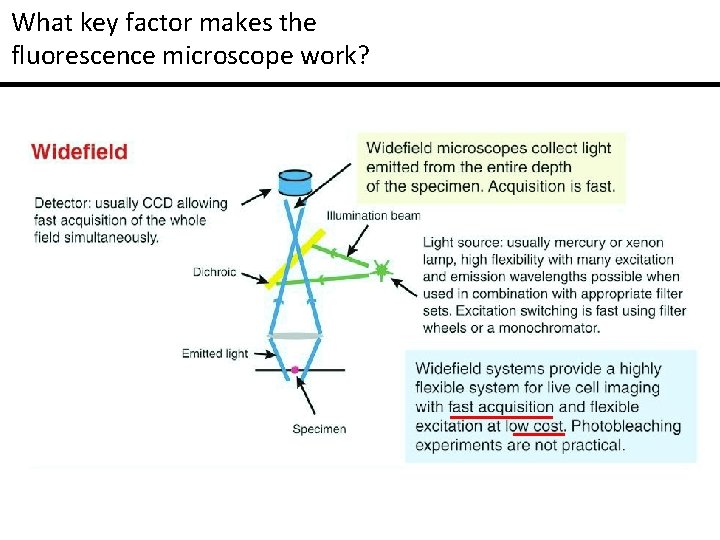
What key factor makes the fluorescence microscope work?
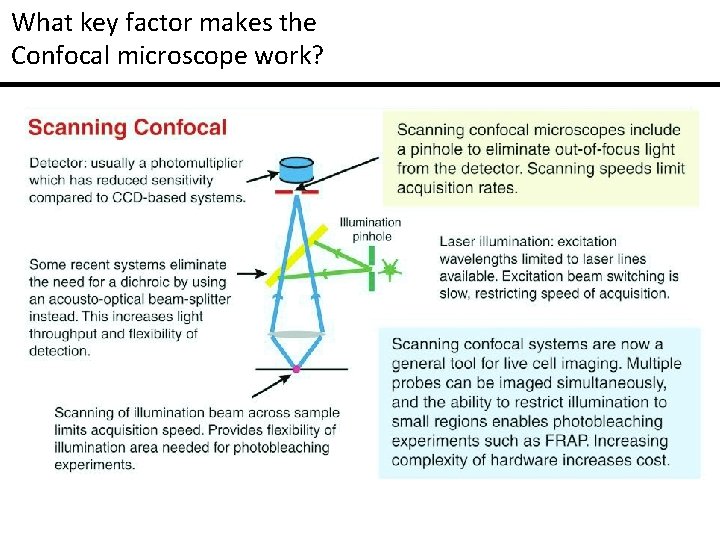
What key factor makes the Confocal microscope work?
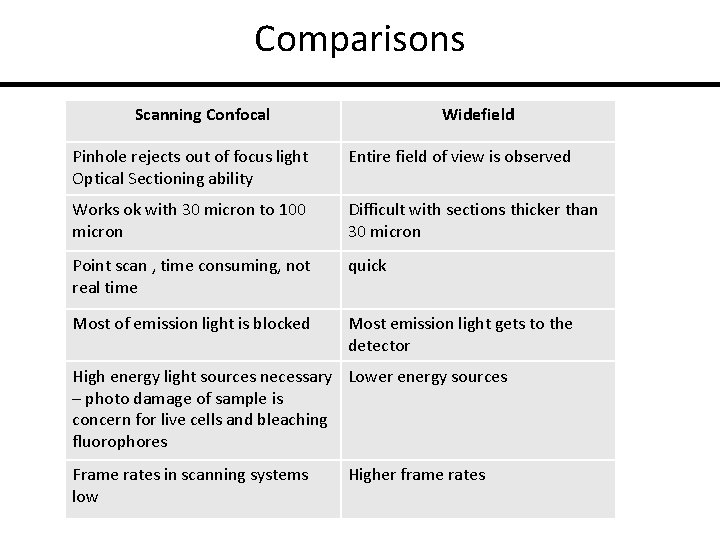
Comparisons Scanning Confocal Widefield Pinhole rejects out of focus light Optical Sectioning ability Entire field of view is observed Works ok with 30 micron to 100 micron Difficult with sections thicker than 30 micron Point scan , time consuming, not real time quick Most of emission light is blocked Most emission light gets to the detector High energy light sources necessary Lower energy sources – photo damage of sample is concern for live cells and bleaching fluorophores Frame rates in scanning systems low Higher frame rates
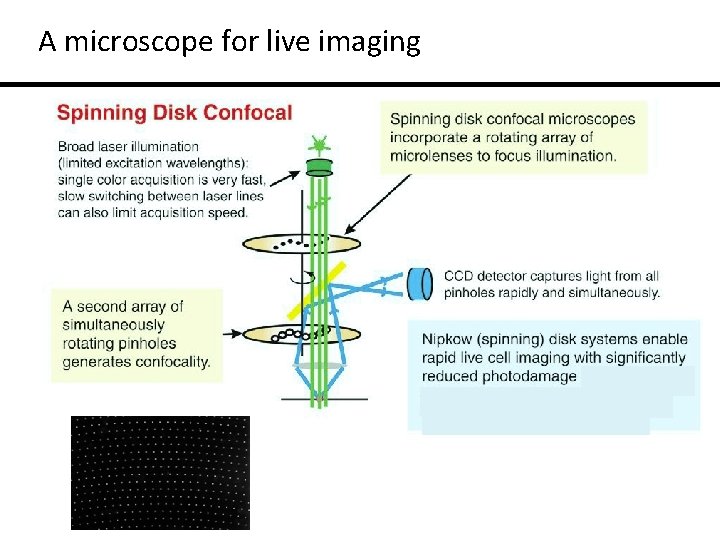
A microscope for live imaging
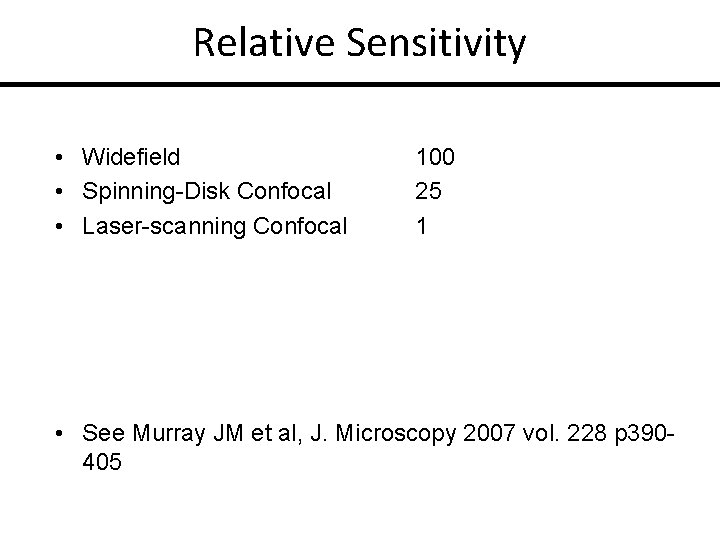
Relative Sensitivity • Widefield • Spinning-Disk Confocal • Laser-scanning Confocal 100 25 1 • See Murray JM et al, J. Microscopy 2007 vol. 228 p 390405

Other approaches to cleaning up out of focus light • TIRF membrane localised events – 100 nm • Multiphoton: localised excitation, inherent optical sectioning , deep penetration into thick samples, reduced photobleaching • light-sheet microscopes illuminate sample planes one at a time with relatively low-intensity light, imaging in an orthogonal direction. thick samples , whole organisms, live samples, low bleaching. • But are expensive set ups and not available NUIG.

Which imaging technique should I use? 1 -5 mm Wide-field (+deconvolution) Fast Spinning Disk Confocal Line-scanning confocal >20 mm Point scanning Confocal >50 mm 2 -photon confocal Slow 10 -100 mm Sensitivity Sample Thickness 1 -20 mm TIRF (for samples at the coverslip)

Setting up the right experimental conditions • sample prep is key Garbage in = Garbage out • Check your staining is optimised before spending time on a high end microscope • Will you image live cells or fixed cells? • How will you stain them? • what will you grow them on? • What objectives will you use to image your cells, and what tradeoffs does that decision represent?

Setting up the right experimental conditions • Plan experiment carefully • Köhler illumination is recommended to produce equally distributed transmitted light and ensure the microscope reaches its full resolving potential. • Match sample to objective • Use correct immersion oil • Use correct cover slip , plates etc – optical grade • Transmitted light – don’t forget, not all about fluorescence! overlay fluorescence and transmitted light image to generate amazing structural context

The most important piece of the optics in a microscope : the Objective r = 0. 61λ/NAobj NA = nsinθ Finest observable details given by λ/2 NA Enables ‘parfocality’ – changing objectives with minimal refocussing
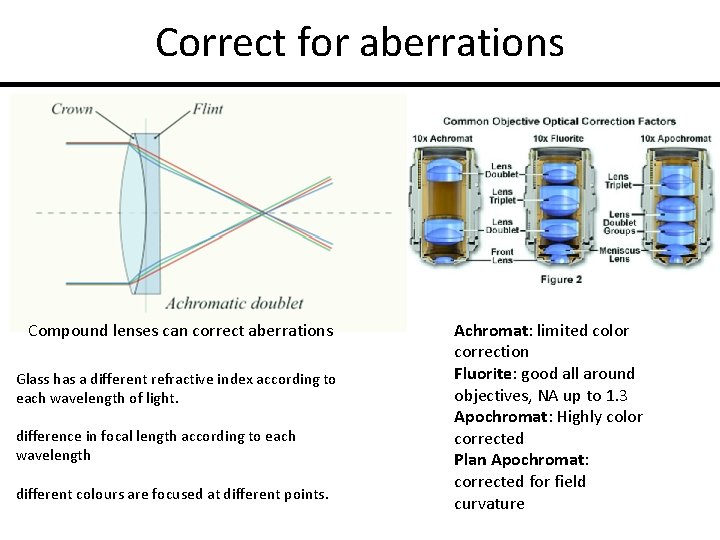
Correct for aberrations Compound lenses can correct aberrations Glass has a different refractive index according to each wavelength of light. difference in focal length according to each wavelength different colours are focused at different points. Achromat: limited color correction Fluorite: good all around objectives, NA up to 1. 3 Apochromat: Highly color corrected Plan Apochromat: corrected for field curvature
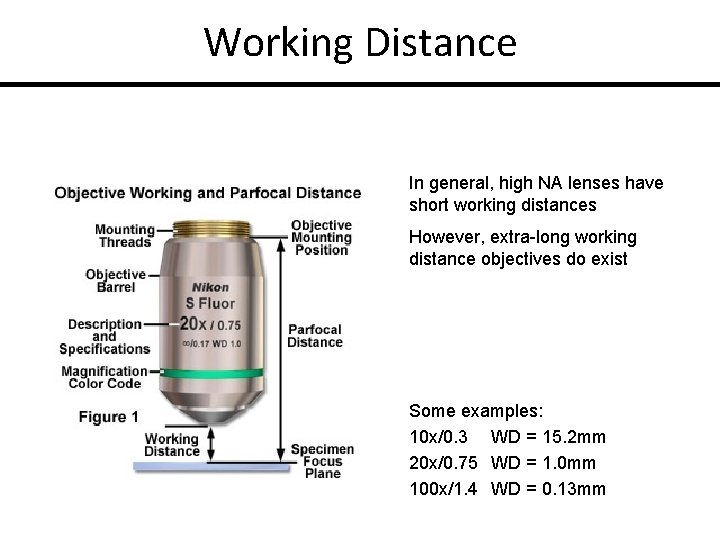
Working Distance In general, high NA lenses have short working distances However, extra-long working distance objectives do exist Some examples: 10 x/0. 3 WD = 15. 2 mm 20 x/0. 75 WD = 1. 0 mm 100 x/1. 4 WD = 0. 13 mm

Resolution of the Microscope limited by the point-spread function • Microscope objective collects a limited cone of light from the sample Y • This limits the resolution achievable by the microscope • Resolution can be measured by the blurring of a point object → the point-spread function X Z
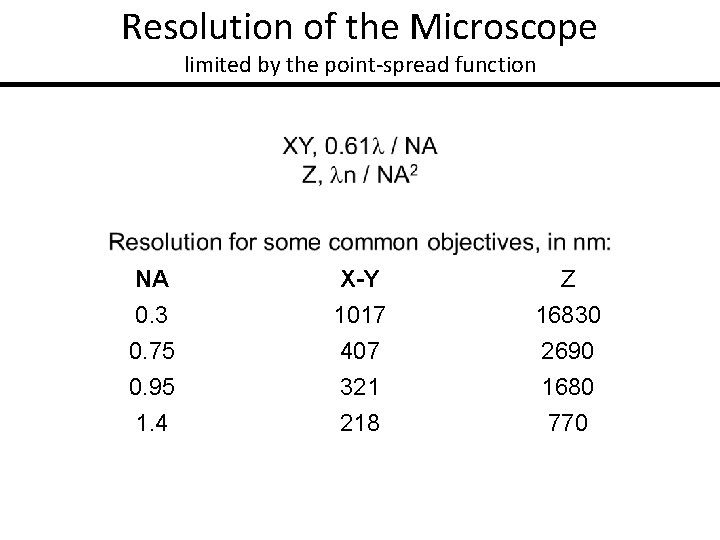
Resolution of the Microscope limited by the point-spread function NA 0. 3 0. 75 0. 95 1. 4 X-Y 1017 407 321 218 Z 16830 2690 1680 770
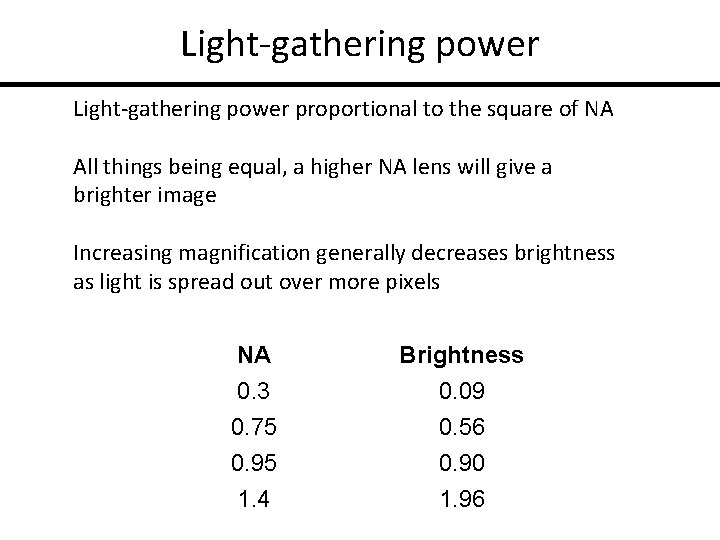
Light-gathering power proportional to the square of NA All things being equal, a higher NA lens will give a brighter image Increasing magnification generally decreases brightness as light is spread out over more pixels NA Brightness 0. 3 0. 75 0. 95 1. 4 0. 09 0. 56 0. 90 1. 96

Choosing an objective • Questions: – What resolution do you need? – How bright is your sample? • For high resolution, you’ll need high NA. • For dim samples, you’ll want high NA, regardless of resolution, to maximize light-gathering. – Dim, low-resolution samples (e. g. protein abundance in nucleus): – Possible solution: bin camera to trade off resolution for brightness

Choosing an objective • Questions: – What resolution do you need? – How bright is your sample? • When to use low NA? – Bright samples at low resolution / low magnification – If you need long working distance – If spherical aberration is a concern – If you want large depth of field to get whole structures in focus at once (avoid Z-stacks)

Cover slips • Most objectives are matched to #1. 5 or 170 micron thickness coverslips • Its all about refractive index matching and reduction of diffraction • Sample RI – Glass RI (1. 52 – Culture medium RI(1. 33) • Some objectives have correction collar, allow for variations in the coverslip thickness.

Immersion media • Five types of objectives are commonly used in biological research: dry, water-immersion, oil-immersion, glycerinimmersion, and water-dipping objectives. • Dry – used in air • Immersion – drop of liquid between sample and lens front surface. • Water dipping allow immersion in media (upright systems) • Oil has an RI (~1. 52) very similar to glass. • oil immersion lens effectively couples the glass coverslip and glass objective in a block of uniform material. • Glycerin has an RI of ~1. 47, making it compatible with some common sample mounting media, • water’s RI is ~1. 33. • Matching the immersion media to the sample is critical for microscopy.

Immersion media cont’ • Example : oil-immersion objective imaging into an aqueous media (e. g. , culture media) will create spherical aberrations due to a mismatch in RIs. • Spherical aberration is an optical distortion that causes objects to appear stretched or compressed, and which also results in an apparent loss of photons. Spherical aberration prop to NA 3 Use correction collar
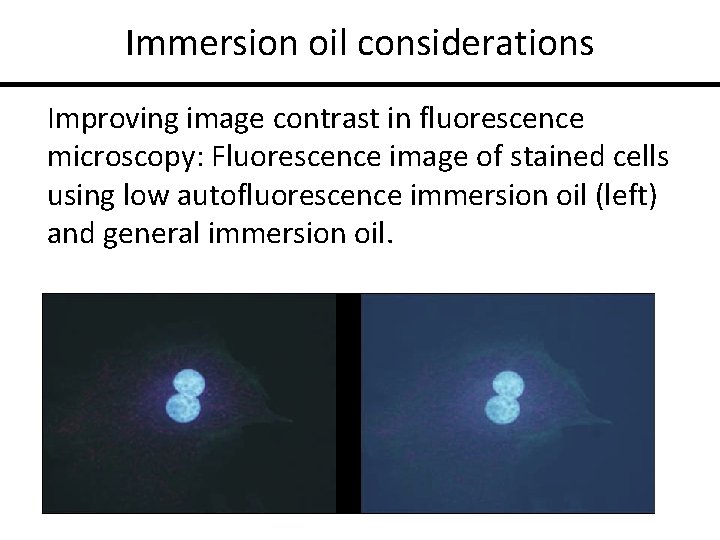
Immersion oil considerations Improving image contrast in fluorescence microscopy: Fluorescence image of stained cells using low autofluorescence immersion oil (left) and general immersion oil.

Which oil immersion lens to use? • 60 x / 1. 2 for samples with RI ≈ 1. 33 • 60 x/1. 3 silicone for samples with RI ≈ 1. 4 • 60 x/1. 42 oil for samples with RI ≈ 1. 52 Methyl Salicylate ( thick specimens) Living cells Fixed Cells Tissue Immersion Oil Glycerol Vectashield DPX Gel/Mount (Biomedia) Coverslip Glass Water Air 1. 53 -1. 54 varies 1. 35 -1. 4 depends on mounting 1. 515 1. 47 1. 45 1. 525 1. 36 1. 52 1. 33 1. 0 Some different refractive indices But η prop T and λ!!!

RI of commonly imaged tissues Biswas and Gaupta 2002

Correct use of immersion oil • See guidelines (http: //imaging. nuigalway. ie/equipment/fluoview 1 000 brb. html) • Never use oil on air objective • Know how to distinguish air/immersion objectives • Always check what objective you are using – know the NA • Know how to clean oil lenses and how to use oil on lenses

Microscope Care • Keeping the microscope optics clean is important for high-quality imaging. Dust, fingerprints, excess immersion oil, or mounting medium on or in a microscope causes reduction in contrast and resolution. DIC is especially sensitive to contamination and scratches on the lens surfaces. • Always keep microscopes covered when not in use. • Make sure that all ports, tubes, and unoccupied positions on the lens turrets are plugged. • Plastic plugs are usually supplied with the microscope.

Areas to watch out for • The external surface of the front lens of the objective • The surface of the camera sensor and its protective glass cover • Both surfaces of the cover slip • The surface of the microscope slide • The surface of the camera adapter optics • Surface of the upper lens of the condenser • Other glass surfaces in the light path e. g. bulbs of halogen- or high pressure lights, fluorescence filters and beam splitters, collector lenses, contrast and heat filters.
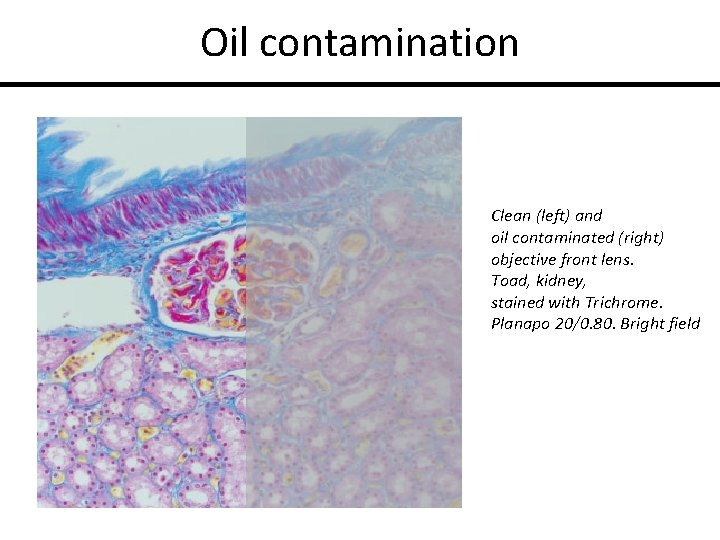
Oil contamination Clean (left) and oil contaminated (right) objective front lens. Toad, kidney, stained with Trichrome. Planapo 20/0. 80. Bright field
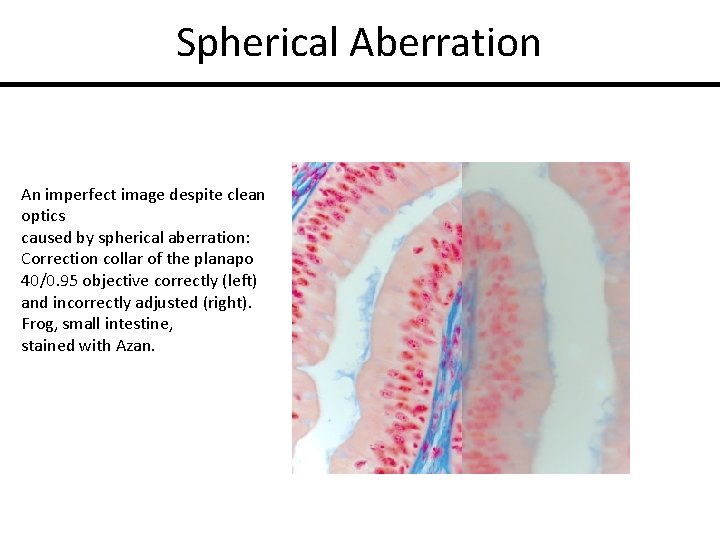
Spherical Aberration An imperfect image despite clean optics caused by spherical aberration: Correction collar of the planapo 40/0. 95 objective correctly (left) and incorrectly adjusted (right). Frog, small intestine, stained with Azan.

Dust on the optics Dust on the internal optics (upper right), extremely dirty camera (bottom right) and clean optics (left). Frog, small intestine, stained with Azan. Planapo 10/0. 45. Bright field.

Lens Cleaning When cleaning lens surfaces, avoid touching the lens surface with anything (even lens paper if possible). IMPORTANT NOTE: Never use Kimwipes or commercial facial tissue, because they may contain a filler that is part Silica material (glass). One pass of a standard tissue could ruin an objective. Some objectives cost in order of € 5 K! Remove dust by using a hand blower. Don’t use compressed air blower. Remove water-soluble contamination using distilled water with a small amount of lens cleaning solution.

Cleaning Oil Clean oil, other contaminants using proper optical micro lens cleaner. Pure Isopropanol , pure petroleum ether or Ted Pella Optical Lens Cleaner • Optical Cleaning Solution L (from Carl Zeiss®) 85% petroleum ether 15% isopropanol Remove most immersion oil by passing a high-quality lens tissue over the objective or condenser front element. Do not leave oil on lens when finished imaging Wipe excess off when changing from objective to objective Be very careful – slow down. Clean objective lenses by holding a piece of doubled lens paper over the objective and placing a few drops of solvent on the paper. Draw the paper across the lens surface so that the solvent flows rapidly in a circular pattern over the recessed lens surface. Finish the stroke with a dry portion of the paper. Repeat as necessary. Avoid soaking a lens with solvent, to prevent damage to lens cements.

Cleaning lenses To clean recessed front elements of dry objective lenses or to remove stubborn dirt, use a cotton-tipped applicator that has been soaked in cleaning solution and then shaken to remove excess fluid. Rotate the cotton tip over the lens surface to clean. Again, first use the optical lens cleaner solution to remove the oil from the surface. Use a detergent solution or ethanol to clean the surfaces of the eyepiece lenses. Do not use xylene as it may solubilize enamel surfaces

Cleaning lenses IMPORTANT NOTE: Do not rub the surface of the lens. No area on the tissue should come in contact with the lens twice. This prevents dust and dirt removed from the lens from coming back and possibly scratching it. Wash hands – minimise oils/crud from fingers getting onto the lens surface.

Cleaning - Spiral motion Cleaning is achieved using a spiral motion from the center to the rim. Never wipe using zig-zag movements as this will only spread the dirt.

Cleaning - To recap 1. When starting to clean, don’t forget to use a dust blower except when fluids (such as immersion oil) are to be removed. 2. Never wipe lenses with dry swabs or tissue – this causes scratches! 3. Do not use abrasive materials e. g. leather wipes, dry linen cloths or polystyrene sticks. 4. Do not apply any solvents before trying distilled water (a film of distilled water can be generated by breathing on the surface), except when grease/oil is to be removed. 5. Do not use any disposable cotton swabs ( e. g. Q-Tip. R) instead of cotton or ITW Texwipe Clean. Tips. R swabs, as the former are not free from contamination. 6. Do not use any of the optical spray cans containing pressurized liquid air. The pressurized air from these sprays leaves a slight, but difficult to remove, residue. 7. Never use acids or ammonia to clean objective front lenses. 8. Never try to clean the internal optical surfaces, cameras or adaptor optics.

Preparation of Slides • Be careful with any mounting media – make sure its set, do not get onto objectives, or use too soon • Make sure sealer (nail varnish) for cover slips has set before using the microscope • Image in good time – don’t leave the slides too long. Store in fridge. Keep away from room light. (for immunohistochemical stains)

Pitfalls in microscopy • Bleaching , NA, filters, aberration • Experimental setup – Fluorophore cross talk – Fluorophore saturation – Detector saturation • Image acquisition – Offset – Binning, under, over sampling • Software resolution issues
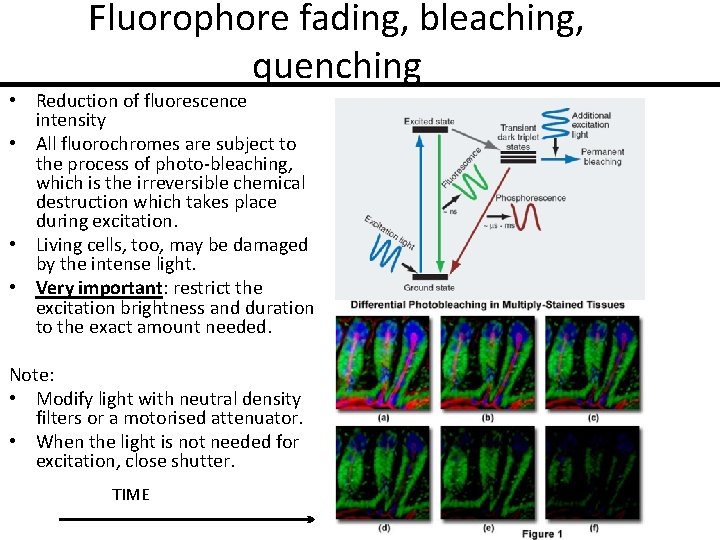
Fluorophore fading, bleaching, quenching • Reduction of fluorescence intensity • All fluorochromes are subject to the process of photo-bleaching, which is the irreversible chemical destruction which takes place during excitation. • Living cells, too, may be damaged by the intense light. • Very important: restrict the excitation brightness and duration to the exact amount needed. Note: • Modify light with neutral density filters or a motorised attenuator. • When the light is not needed for excitation, close shutter. TIME

False Data Interpretation Mouse brain tissue slice 18 micron stack, 50 z-sections, 0. 33 micron step 40 x 1. 2 NA Blue Dapi nuclei Green GFP dendrites Red Cy 3 microtubule associated protein MAP 2 Image A: xy merge at top of stack Image B: xy merge at middle of stack Image C: xz slice through the stack GFP and DAPI extend through whole section Cy 3 is restricted to top and bottom z sections Concl: Incomplete penetration of antibody
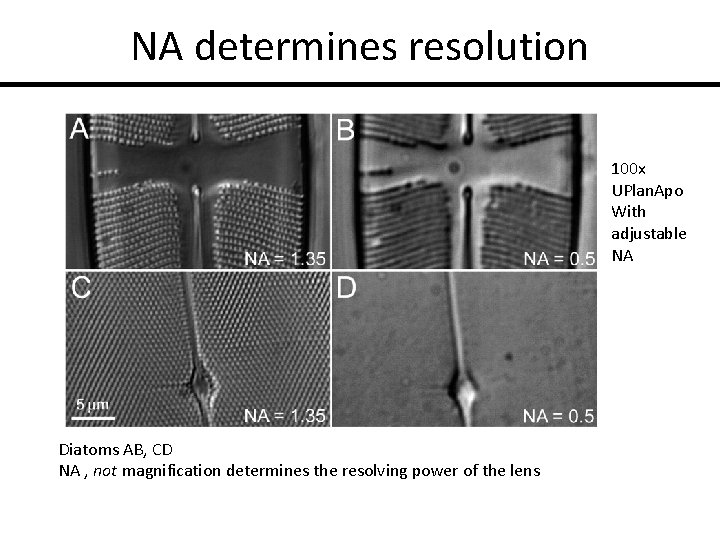
NA determines resolution 100 x UPlan. Apo With adjustable NA Diatoms AB, CD NA , not magnification determines the resolving power of the lens
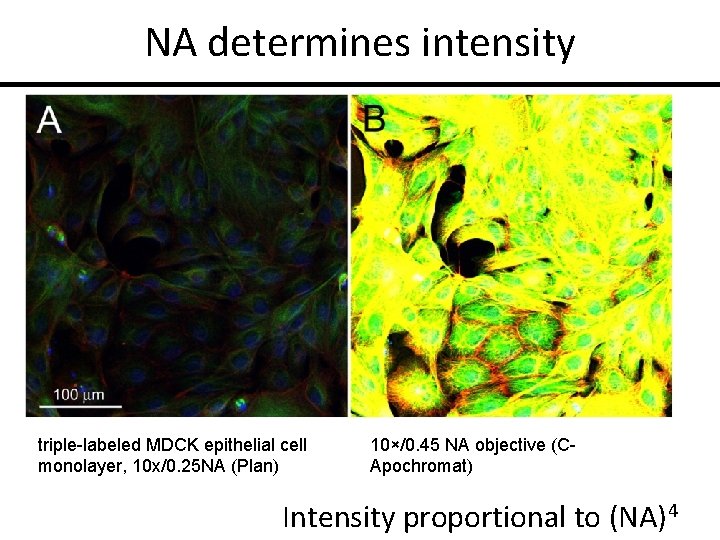
NA determines intensity triple-labeled MDCK epithelial cell monolayer, 10 x/0. 25 NA (Plan) 10×/0. 45 NA objective (CApochromat) Intensity proportional to (NA)4
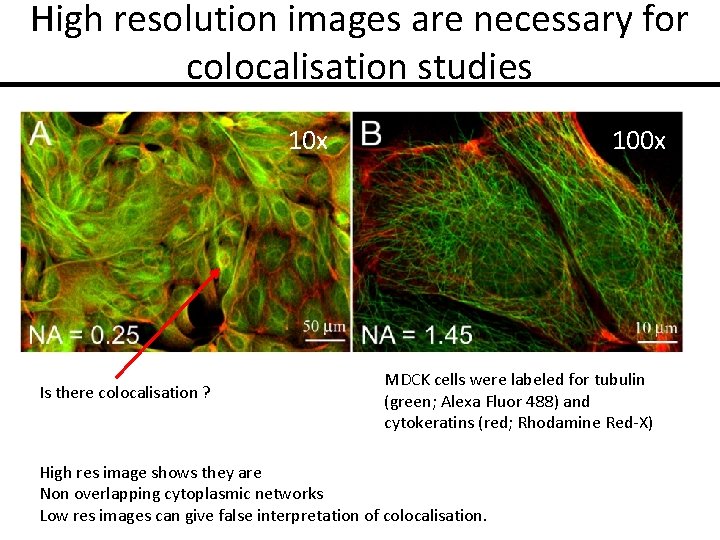
High resolution images are necessary for colocalisation studies 10 x Is there colocalisation ? 100 x MDCK cells were labeled for tubulin (green; Alexa Fluor 488) and cytokeratins (red; Rhodamine Red-X) High res image shows they are Non overlapping cytoplasmic networks Low res images can give false interpretation of colocalisation.
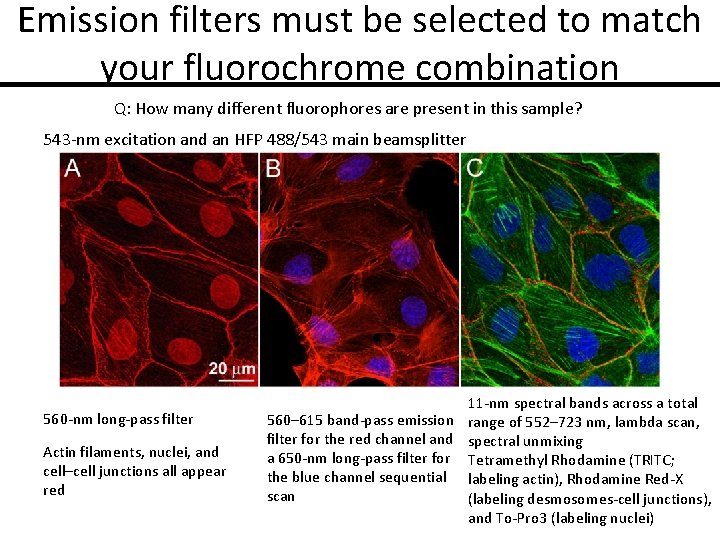
Emission filters must be selected to match your fluorochrome combination Q: How many different fluorophores are present in this sample? 543 -nm excitation and an HFP 488/543 main beamsplitter 560 -nm long-pass filter Actin filaments, nuclei, and cell–cell junctions all appear red 11 -nm spectral bands across a total 560– 615 band-pass emission range of 552– 723 nm, lambda scan, filter for the red channel and spectral unmixing a 650 -nm long-pass filter for Tetramethyl Rhodamine (TRITC; the blue channel sequential labeling actin), Rhodamine Red-X scan (labeling desmosomes-cell junctions), and To-Pro 3 (labeling nuclei)
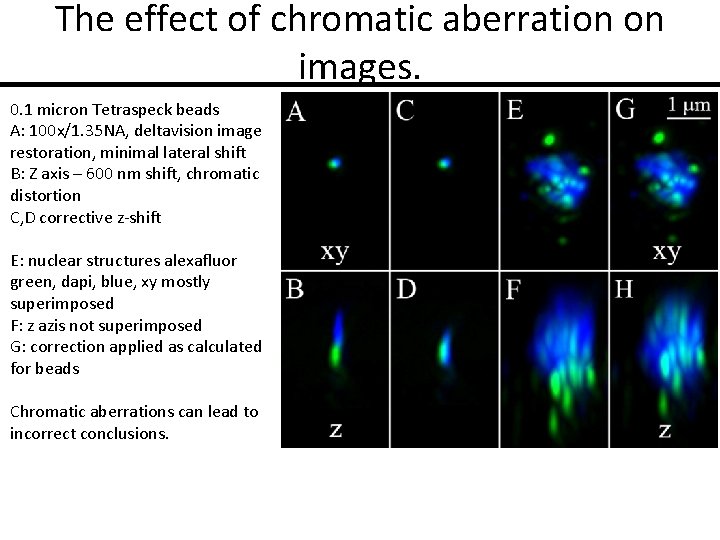
The effect of chromatic aberration on images. 0. 1 micron Tetraspeck beads A: 100 x/1. 35 NA, deltavision image restoration, minimal lateral shift B: Z axis – 600 nm shift, chromatic distortion C, D corrective z-shift E: nuclear structures alexafluor green, dapi, blue, xy mostly superimposed F: z azis not superimposed G: correction applied as calculated for beads Chromatic aberrations can lead to incorrect conclusions.
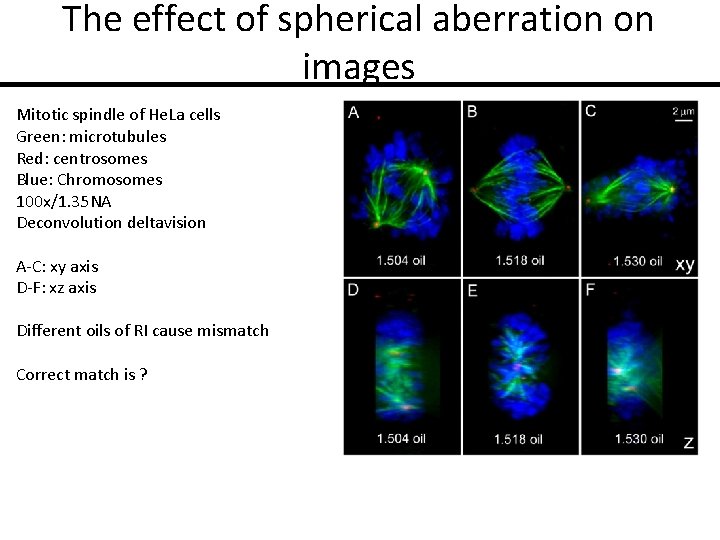
The effect of spherical aberration on images Mitotic spindle of He. La cells Green: microtubules Red: centrosomes Blue: Chromosomes 100 x/1. 35 NA Deconvolution deltavision A-C: xy axis D-F: xz axis Different oils of RI cause mismatch Correct match is ?

Spectral Bleed/ cross talk Multi-colour image of a GFP/YFP doublelabelled sample. In this sample, GFP was fused with the H 2 B histone protein and YFP with tubulin. Collect images for different fluorophores sequentially whenever possible to avoid crosstalk between image channels when multiple fluorophores are excited simultaneously.
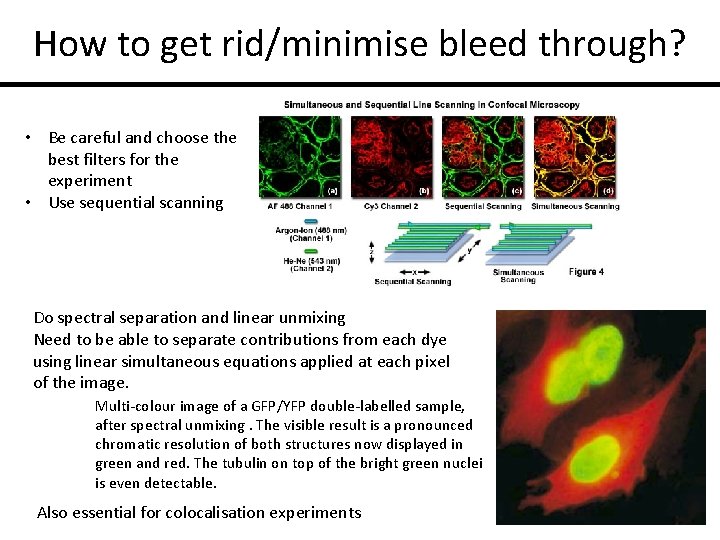
How to get rid/minimise bleed through? • Be careful and choose the best filters for the experiment • Use sequential scanning Do spectral separation and linear unmixing Need to be able to separate contributions from each dye using linear simultaneous equations applied at each pixel of the image. Multi-colour image of a GFP/YFP double-labelled sample, after spectral unmixing. The visible result is a pronounced chromatic resolution of both structures now displayed in green and red. The tubulin on top of the bright green nuclei is even detectable. Also essential for colocalisation experiments
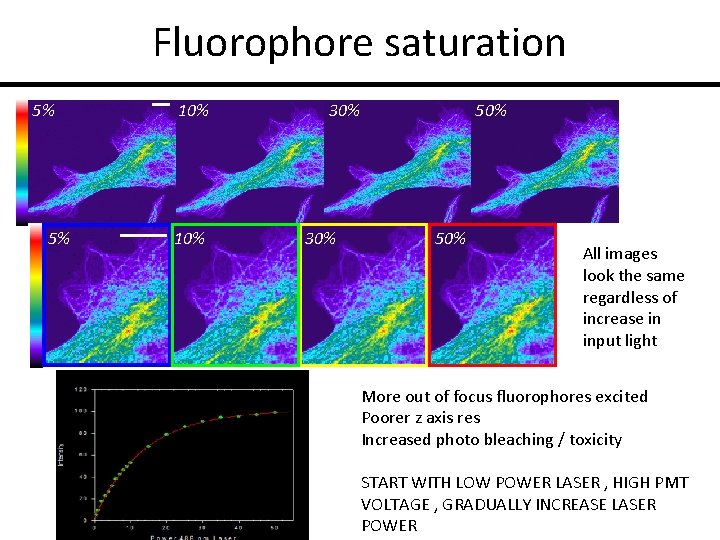
Fluorophore saturation 5% 5% 10% 30% 50% All images look the same regardless of increase in input light More out of focus fluorophores excited Poorer z axis res Increased photo bleaching / toxicity START WITH LOW POWER LASER , HIGH PMT VOLTAGE , GRADUALLY INCREASE LASER POWER
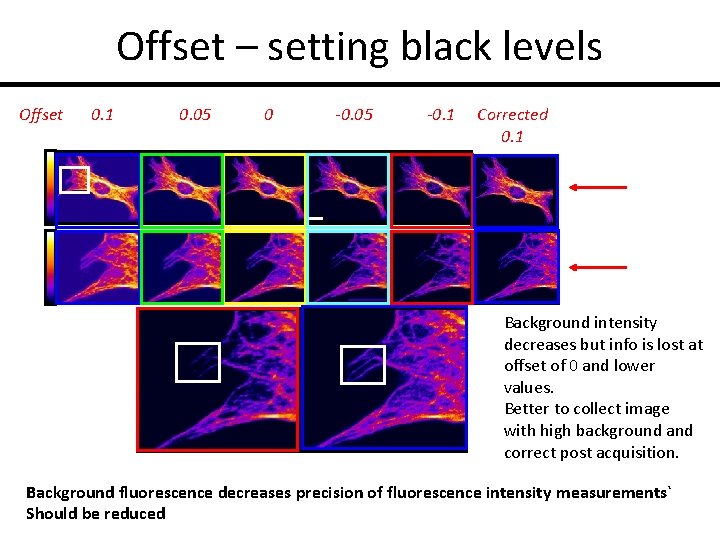
Offset – setting black levels Offset 0. 1 0. 05 0 -0. 05 -0. 1 Corrected 0. 1 Background intensity decreases but info is lost at offset of 0 and lower values. Better to collect image with high background and correct post acquisition. Background fluorescence decreases precision of fluorescence intensity measurements` Should be reduced

Detector saturation Increasing Laser Power If PMT gain, laser power, lamp intensities of camera exposure times are too high , detector can saturate causing a loss of structural information within bright structures Best to have NO saturated pixels (red) – use HI-LO look up table (LUT).
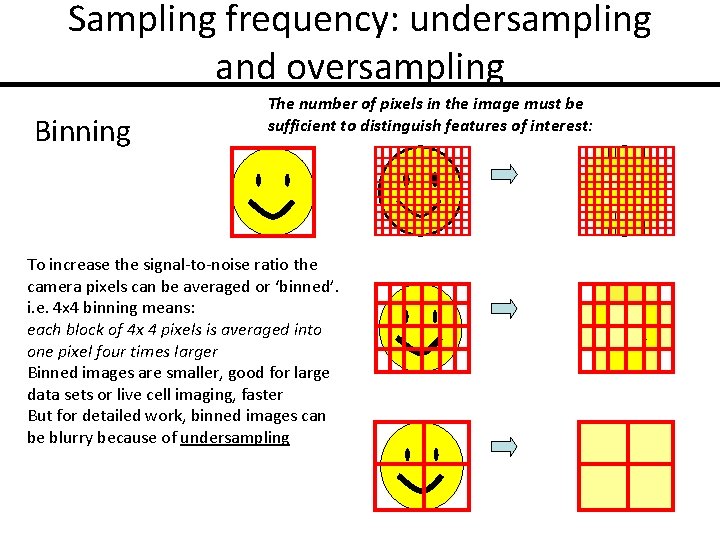
Sampling frequency: undersampling and oversampling Binning The number of pixels in the image must be sufficient to distinguish features of interest: To increase the signal-to-noise ratio the camera pixels can be averaged or ‘binned’. i. e. 4 x 4 binning means: each block of 4 x 4 pixels is averaged into one pixel four times larger Binned images are smaller, good for large data sets or live cell imaging, faster But for detailed work, binned images can be blurry because of undersampling
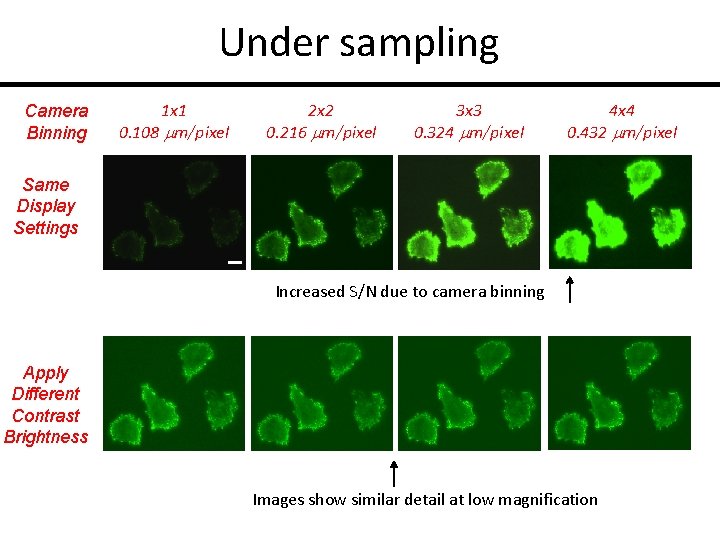
Under sampling Camera Binning 1 x 1 0. 108 mm/pixel 2 x 2 0. 216 mm/pixel 3 x 3 0. 324 mm/pixel 4 x 4 0. 432 mm/pixel Same Display Settings Increased S/N due to camera binning Apply Different Contrast Brightness Images show similar detail at low magnification
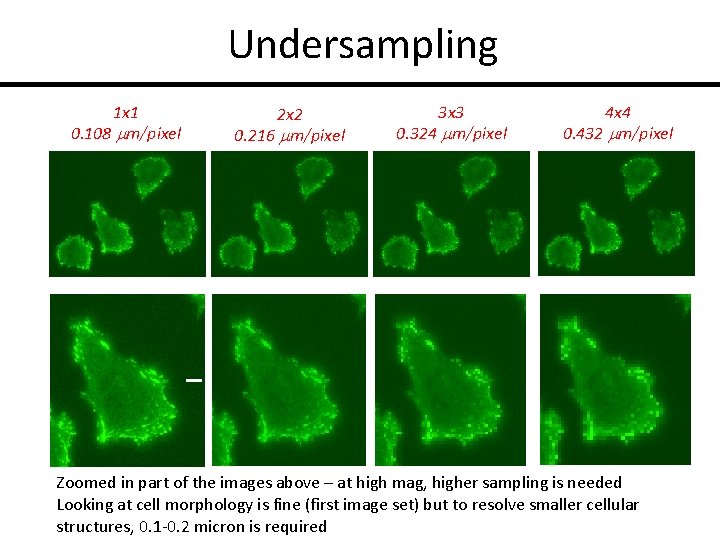
Undersampling 1 x 1 0. 108 mm/pixel 2 x 2 0. 216 mm/pixel 3 x 3 0. 324 mm/pixel 4 x 4 0. 432 mm/pixel Zoomed in part of the images above – at high mag, higher sampling is needed Looking at cell morphology is fine (first image set) but to resolve smaller cellular structures, 0. 1 -0. 2 micron is required
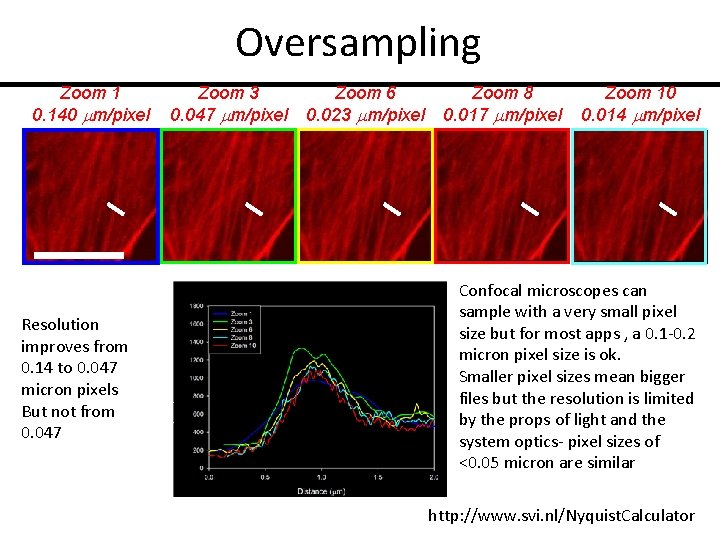
Oversampling Zoom 1 0. 140 mm/pixel Resolution improves from 0. 14 to 0. 047 micron pixels But not from 0. 047 Zoom 3 0. 047 mm/pixel Zoom 6 0. 023 mm/pixel Zoom 8 0. 017 mm/pixel Zoom 10 0. 014 mm/pixel Confocal microscopes can sample with a very small pixel size but for most apps , a 0. 1 -0. 2 micron pixel size is ok. Smaller pixel sizes mean bigger files but the resolution is limited by the props of light and the system optics- pixel sizes of <0. 05 micron are similar http: //www. svi. nl/Nyquist. Calculator

Software settings and Image display 100 ms 400 ms 700 ms Variable exposure times , same image display settings Brightness and contrast adjusted to give similar display intensities Changing image display settings can highlight dim features. Lower exposure times or laser powers may still reveal what you want to see… 1000 ms

Image display - colour Same image with variable colour displays : 3 D intensity plot Blue, red channels can be difficult to visualise; better to use greyscale images or pseudo colour LUTs to bring out dim features within images.
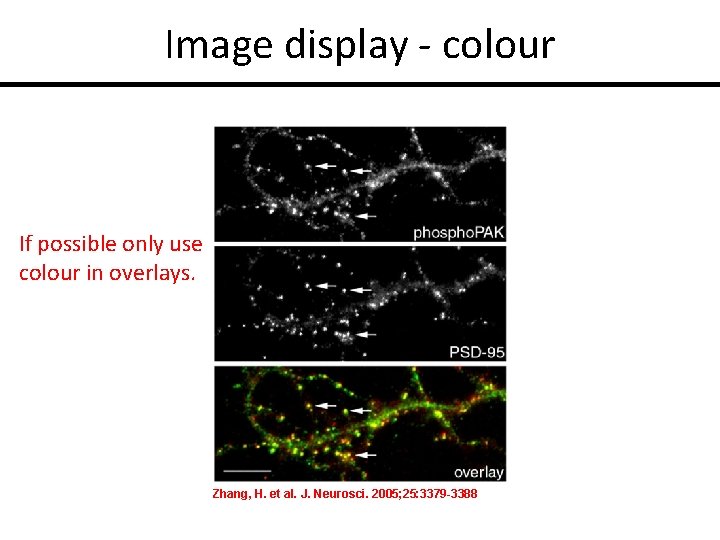
Image display - colour If possible only use colour in overlays. Zhang, H. et al. J. Neurosci. 2005; 25: 3379 -3388

Image display – gamma factor Display Intensity Same image with varying display settings: our eyes process image non-linearly contrast All Three linear gamma 2 gamma 0. 5 brightness Image Intensity Using the gamma correction brings out cell edges and dim features

Software tools • Volocity (here on campus via CMI) • Photoshop • Image. J • Incarnations of Image J: • FIJI (is just image J…. Fiji is an image processing package, a distribution of Image. J (and soon Image. J 2) together with Java, Java 3 D and a lot of plugins organized into a coherent menu structure. Fiji compares to Image. J as Ubuntu compares to Linux. • Also …Mac Biophotonics Image J, another collection of useful plugins wrapped up into the main image J container.

Deconvolution • No microscope is perfect, and some distortion is inevitable. • But it is possible to create a mathematical model of the microscope’s optical characteristics using the instrument’s so-called “point spread function” (PSF), which describes the behavior of an infinitely small fluorescent point. • Deconvolution uses this model to back-calculate the original appearance of the imaged field of view. • Deconvolution thus cleans up a pre-existing image by digitally reconstructing it. • With good deconvolution and z scanner, possible to generate 3 d images with widefield
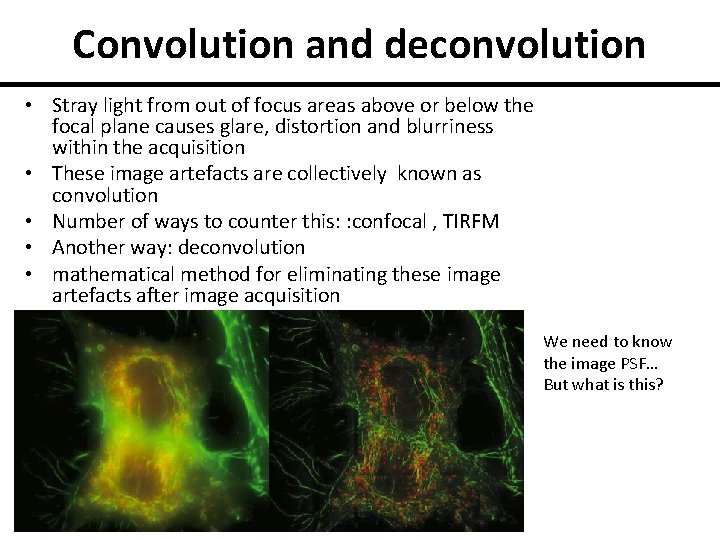
Convolution and deconvolution • Stray light from out of focus areas above or below the focal plane causes glare, distortion and blurriness within the acquisition • These image artefacts are collectively known as convolution • Number of ways to counter this: : confocal , TIRFM • Another way: deconvolution • mathematical method for eliminating these image artefacts after image acquisition We need to know the image PSF… But what is this?

What is the PSF ? • The PSF is the image of a point source of light from the specimen projected by the microscope objective onto the intermediate image plane, i. e. the point spread function is represented by the Airy disk pattern • PSF of an individual objective or a lens system depends on NA, objective, λillumination, contrast mode (e. g. brightfield, phase, DIC). • Models spread/distortion/diffraction of light in 3 dimensions

PSF Stack of images: convolution After imaging and If we know the psf… deconvolution Glare , distortion , bluriness
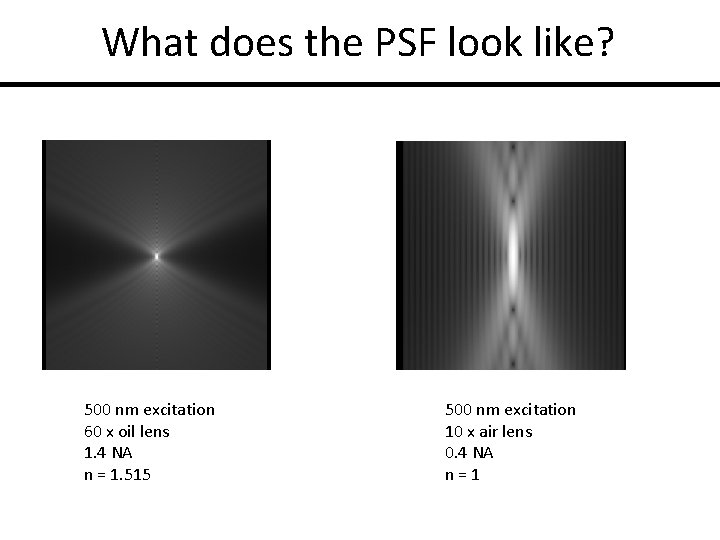
What does the PSF look like? 500 nm excitation 60 x oil lens 1. 4 NA n = 1. 515 500 nm excitation 10 x air lens 0. 4 NA n=1

Volocity and deconvolution • Can deconvolute images from widefield, confocal, spinning disk systems • Create calculated psf using input data or/ • Use measured PSF (from fluorescent beads) • Deconvolute from image • Easy to do and worth it! • Volocity available to all on campus – see imaging. nuigalway. ie

Checklist for optimizing images for quantitation Increase signal: ✓ Choose a bright (high quantum yield, high extinction coefficient) and photostable fluorophore ✓ Image through a clean No. 1. 5 coverslip ✓ Mount specimen as close to the coverslip as possible ✓ Use high NA clean objective lens with lowest acceptable magnification ✓ Choose fluorescence filter sets that match fluorophore spectra ✓ Align lamp for Koehler illumination ✓ For fixed specimens, use a glycerol-based mounting medium containing anti-photobleaching inhibitors ✓ Remove DIC Wollaston prism and analyzer from light path ✓ Use a cooled CCD camera with at least 60% quantum efficiency ✓ Use camera binning

Checklist for optimizing images for quantitation Decrease noise: ✓ Use a cooled CCD camera with less than 8 electrons readout noise and negligible dark noise ✓ Use amplification (e. g. , EM-CCDs) only when signal is limiting ✓ Increase signal (see above) to reduce relative contribution of Poisson noise

Checklist for optimizing images for quantitation Decrease background: ✓ Clean coverslips and optics ✓ Optimise fluorophore labeling protocol to minimize nonspecific labeling ✓ Mount specimens in minimally fluorescent medium (e. g. , without phenol red) ✓ Use band-pass filter sets that block autofluorescence ✓ Turn off the room lights ✓ Close down the field diaphragm to illuminate only the object of interest ✓ When out-of-focus fluorescence is high, consider using deconvolution, confocal, or TIRF

To recap , we talked about… • • What systems to use Widefield vs confocal Care of your microscope, how to clean Sample prep Practical tips for generating good images Pitfalls when generating images Deconvolution
- Slides: 76