Seed Funding Health Technologies National SBIR Conference May

Seed Funding Health Technologies National SBIR Conference May 16, 2017 1

Panelists • Matthew Portnoy – NIH/HHS – Moderator • • • Keith L. Woodman - NASA Jesus Soriano – NSF Larry Pollack – Do. D/Chemical and Biological Defense Capt. Sally Hu – Do. D/Defense Health Agency Scott Dockum - USDA 2
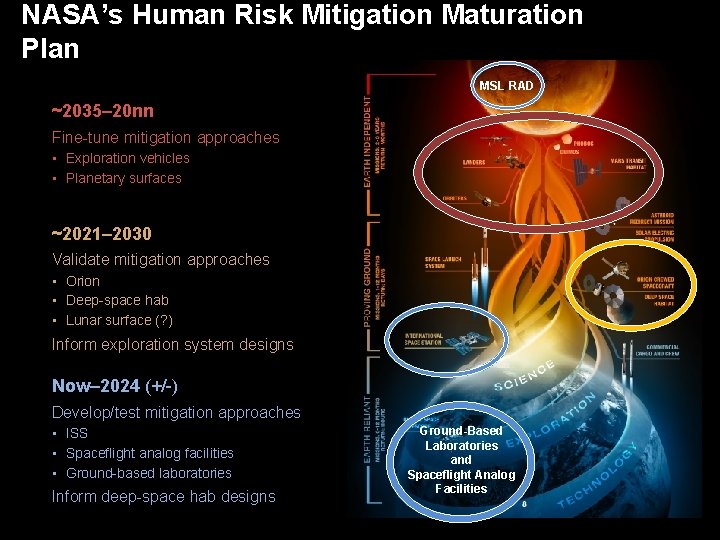
NASA’s Human Risk Mitigation Maturation Plan MSL RAD ~2035– 20 nn Fine-tune mitigation approaches • Exploration vehicles • Planetary surfaces ~2021– 2030 Validate mitigation approaches • Orion • Deep-space hab • Lunar surface (? ) Inform exploration system designs Now– 2024 (+/-) Develop/test mitigation approaches • ISS • Spaceflight analog facilities • Ground-based laboratories Inform deep-space hab designs Ground-Based Laboratories and Spaceflight Analog Facilities

NASA’s Primary Human Research Elements Space Radiation Recommendations to permissible exposure limits, assessment/projection tools/models of crew risk from radiation exposure, and models/tools to assess vehicle design for radiation protection Human Health and Countermeasures Integrated physiological, pharmacological and nutritional countermeasures suite; biomedical expertise for the development and assessment of medical standards and vehicle and spacesuit requirements dictated by human physiological needs Human Factors and Behavioral Performance individual and interpersonal interactions; sleep and stress interfaces between humans and vehicles/habitats/environment Exploration Medical Capability Medical care and crew health maintenance technologies (monitoring, diagnostic, treatment tools and techniques); medical data management; probabilistic risk assessment; technology and informatics development ISS Medical Projects Research integration and operations: ISS, Flight Analog facilities
Some NASA SBIR Human Health Success Stories

Who/What We Fund @NSF SBIR Ø High technical risk innovations with potential for high commercial / societal impact Ø R&D only – you will need to raise money for other activities Ø Focus on start-ups and early stage companies Ø Typical program profile (2014 stats): • 72% of funded companies <5 years old • 90% of funded companies <10 employees • 80% of funded companies had not received a prior Phase II award

What We Don’t Fund @NSF SBIR Ø Basic research Ø Projects lacking technical risk or innovation Ø Incremental/evolutionary improvements to an existing product or service Ø Projects where we don’t see a strong chance of commercial success • This applies to the project AND the proposing company/team Ø Projects where our funding won’t move the needle • If NSF funding can’t make a big impact on the company’s prospects, there are usually better ways to raise money Ø Sales and marketing, customer/market discovery • Will need funding beyond NSF

What’s Different about NSF SBIR Ø Ø NSF won’t buy anything from you We don’t identify the application or technology space You identify the market need and propose a tech-based solution Broad program topics cover almost every area of technology: • • • Educational Technologies & Applications Information Technologies Semiconductors and Photonic Devices & Materials Internet of Things Electronic Hardware, Robotics & Wireless Technologies Advanced Manufacturing & Nanotechnology Advanced Materials & Instrumentation Chemical & Environmental Technologies Biological Technologies Smart Health and Biomedical Technologies Ø Topic fit is much less important than meeting the technical and commercial requirements of the solicitation

What’s Different about NSF SBIR Ø We expect you to be responsive to market changes Ø Pivots are OK – but must be evidence-based Ø You must develop and fund a viable commercialization approach Ø Program Directors are devoted full-time to SBIR/STTR – most are ex-entrepreneurs or have industry experience Ø Program is managed by a dedicated team focused exclusively on SBIR/STTR – everything from writing solicitation to managing the review process to recommending awards

THANK YOU Contact Us: Jesus Soriano jsoriano@nsf. gov @Jesus. VSoriano sbir@nsf. gov www. nsf. gov/SBIR @NSFSBIR

UNCLASSIFIED Joint Science & Technology Office for Chemical and Biological Defense Chemical and Biological Technologies: Taxonomy of Medical Technology Focus Areas UNCLASSIFIED

UNCLASSIFIED Diagnostics, Detection, Disease Surveillance Focus Areas Assays & Biomarkers Development and verification of rapid, sensitive, and specific tests for the identification of Chemical Biological Warfare Agents (CBWAs) and their expressed pathogens and toxins in clinical specimens from Warfighters for the diagnosis of exposure/infection. Discovery of host biomarkers generated in response to exposure to biological threat agents. Also includes codevelopment of next generation diagnostic technologies including portable diagnostic platforms, highly parallel and informative testing formats, novel assay formats and hardware solutions to enable point of need diagnostic capabilities, allowing for rapid guidance of medical decisions. Diagnostic & Detection Devices Threat Surveillance Focuses on the detection and identification of chemical and biological threats in near real-time. Future programs focus on the improvement of algorithms, excitation sources, and detector elements to increase warning time, reduce false positives, increase sensitivity, and reduce cost. Also includes diagnostic device development to include systems able to harness next generation technologies to revolutionize clinical diagnostics in care facilities and in hospital laboratories. This investment will incorporate capabilities such as next generation sequencing and advanced biomolecular methods to harness both host and pathogen biomarkers in a threat agnostic approach that will serve all echelons of military medical care. Deliver cutting edge Integrated Early Warning, Information Management and Applied Analytic capabilities to the warfighter; virtually connect them to these capabilities and other system users for rapid situational awareness, course of action (Co. A) analysis and decision support. This will include development of a biosurveillance framework, integration and demonstration of standards-based device-to-cloud connectivity, development and testing of Role 0 and Role 1 diagnostic device solutions with capability of data upload, and next generation analytic capabilities for prediction, early warning and forecasting. UNCLASSIFIED

UNCLASSIFIED Translational Medical S&T Division Focus Areas Bio-Therapeutics Branch Bio-Pretreatments Branch Discovery and development of therapeutics for alphaviruses, filoviruses, botulinum toxin, and broadspectrum antibiotics targeting multi-drug resistant bacterial threat pathogens Discovery and development of vaccines for: alphaviruses, filoviruses, Bacillus anthracis (anthrax), Burkholderia mallei (glanders), Burkholderia pseudomallei (meliodosis), Francisella tularensis (tularemia), Coxiella burnetii (Q Fever), and botulinum, ricin and SEB toxin Supporting S&T Investments in animal model development, ex vivo human mimetics, novel manufacturing technologies that exploit readily adaptable expression platforms and leverage flexible biomanufacturing technologies, understanding host-pathogen/toxin interaction for identification of targets and/or biomarkers, adjuvants and stabilization technologies, drug delivery methods, and compound library compilation and characterization UNCLASSIFIED

UNCLASSIFIED Advanced & Emerging Threat Division Focus Areas Threat Agent Science Provide rapid validated chemical and biological agent property and reactivity data and determine toxicological mechanisms to inform risk assessment. Medical Chemical Countermeasures Deliver novel medical concepts and technologies to address current and emerging threats to protect the lives of our warfighters CB Research Center of Excellence Provide capabilities and competency to the Do. D labs through DTRA Fellowship program, explore collaborative opportunities, and recruit and mentor young talent through scholarly programs (e. g. STEM and NRC Post doc) UNCLASSIFIED

Defense Health Agency (DHA) SBIR/STTR Programs DHA MISSION The Defense Health Agency (DHA) is a joint, integrated Combat Support Agency that enables the Army, Navy, and Air Force medical services to provide a medically ready force and ready medical force to Combatant Commands in both peacetime and wartime. The DHA supports the delivery of integrated, affordable, and high quality health services to Military Health System (MHS) beneficiaries and is responsible for driving greater integration of clinical and business processes across the MHS. CAPT Sally Hu, DHA SBIR/STTR Program Director
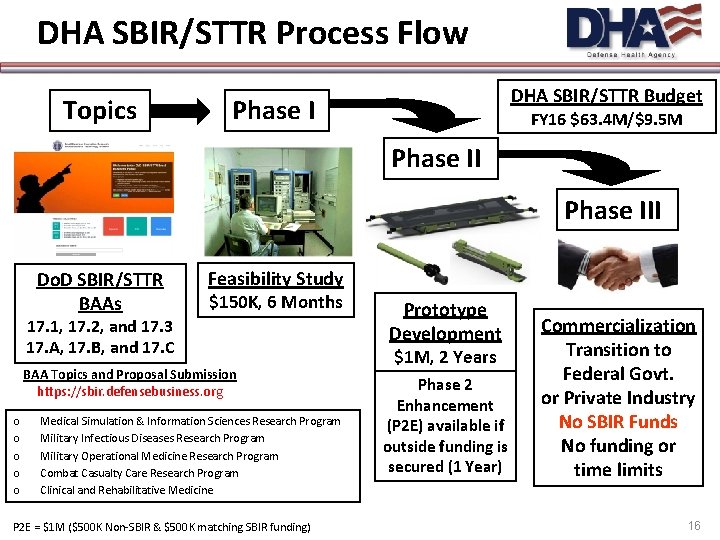
DHA SBIR/STTR Process Flow Topics DHA SBIR/STTR Budget Phase I FY 16 $63. 4 M/$9. 5 M Phase III Do. D SBIR/STTR BAAs Feasibility Study $150 K, 6 Months 17. 1, 17. 2, and 17. 3 17. A, 17. B, and 17. C BAA Topics and Proposal Submission https: //sbir. defensebusiness. org o o o Medical Simulation & Information Sciences Research Program Military Infectious Diseases Research Program Military Operational Medicine Research Program Combat Casualty Care Research Program Clinical and Rehabilitative Medicine P 2 E = $1 M ($500 K Non-SBIR & $500 K matching SBIR funding) Prototype Development $1 M, 2 Years Phase 2 Enhancement (P 2 E) available if outside funding is secured (1 Year) Commercialization Transition to Federal Govt. or Private Industry No SBIR Funds No funding or time limits 16

DHA SBIR Success Story Small Business: Operative Experience, Inc. 2017 Tibbetts Awardee Problem to be Solved: Deploying surgeons are often trauma-naive and ill prepared for the rigors of combat surgery. Previous simulation-based training methods are low fidelity with unrealistic tissue properties, and live tissue training options are not anatomically accurate. Project Objective: The goal of this project is to develop simulation-based methods to rapidly train trauma-naive, deploying surgeons in critical combat surgery skills. Project Description: Operative Experience, Inc. has developed a series of high-fidelity, physical models characterized by artificial skin that can be incised and sutured, subcutaneous tissue, embedded periosteum-covered bones, articulated joints, major muscle groups, integrated dissection planes, fascia, organs, and branching hollow blood vessels. All Models have been Commercialized. Customers include: Special Operations Forces, U. S. Marine Corps, and State Department security. 17

DHA SBIR/STTR Points of Contact Defense Health Agency – Falls Church, VA https: //health. mil/About-MHS/Defense-Health-Agency CAPT Sally Hu, DHA SBIR/STTR Program Director (sally. h. hu. mil@mail. mil, 703 -681 -8221) US Army Medical Research and Materiel Command – Fort Detrick, MD http: //mrmc. amedd. army. mil/ Mr. J. R. Myers, SBIR/STTR Project Manager (james. r. myers. 38. civ@mail. mil, 301 -619 -7377) Ms. Colleen Gibney, SBIR/STTR Deputy Project Manager (colleen. n. gibney. civ@mail. mil, 301 -619 -3719) Ms. Susan Dael, SBIR/STTR Operations Manager (susan. j. dael. ctr@mail. mil, 301 -619 -5047) Ms. Lauren Lynch, DHA SBIR/STTR Program Analyst (lauren. a. lynch 6. ctr@mail. mil, 301 -619 -5146) 18

National Institute of Food And Agriculture USDA SBIR Program Health Innovations and Opportunities SBIR

USDA SBIR Health Focused Topic Areas Food Science and Nutrition Research focusing on developing new and improved processes, technologies, or services that address emerging food safety, food processing and nutrition issues. • Fiberstar Bio-Ingredient Technologies, Inc. : Fat Replacement System from Natural Plant Materials for Improving Food Nutrition, Quality, and Reducing Costs • • • Deliver a fat substitute that is more nutritious, has improved eating properties, and costs less than current alternatives Citri-Fi is a line of clean label, natural functional fibers derived from citrus that offer hydrocolloidal properties produced by significant high water holding capabilities. These benefits improve food texture, enhance health and/or provide cost savings. . Prosperity Organic Foods, Inc. : Development and Expansion of Melt Organic Baking Fat for Oxidative Stability and Reducing Child Obesity 2013 -03922 • • MELT Organic spreads are North America’s first line of luscious butter improvements that are truly good for you. Each variety of MELT Organic features the Perfect Blendtm of the healthiest oils that we’ve carefully chosen for you. Our Perfect Blend is made from natural, fruit- and plant-based organic oils: virgin coconut, palm fruit, flaxseed, sunflower and canola. Can be purchased in major grocery stores across the nation including Whole Foods, Wegmans and Vons. SBIR 20

USDA SBIR Health Focused Topic Areas Rural and Community Development Applications may be submitted for the development of new technology, or for the utilization of existing technology, that address important economic and social development issues or problems in rural America. • Hubble Telemedical, Inc. : Metadata assisted Management of Diabetes in Rural Health Disparity Communities • • • Offers remote diabetic retinopathy screening and analysis services. The company’s services are used to screen various causes of blindness, such as diabetic retinopathy, certain types of cancer, infection, central nervous system/immune disorders, atherosclerosis/risk of stroke, hypertension/cardiovascular disease, glaucoma, age-related macular degeneration, and other retinal defects worldwide. January 20, 2015 Welch Allyn, Inc. , a leading medical diagnostic device company that delivers practical innovation at the point of care, today announced the acquisition of Knoxville, TN-based Hubble Telemedical, Inc. , a privately-held healthcare company that enables remote diabetic retinopathy screening and analysis in primary care and other convenient settings. Createability Concepts, Inc. : Rural-Health-Mate: A Tele-Monitoring System That Improves Rural Senior's Healthcare Via Monitoring, Communication & Stakeholder Interaction 201504161 • • Rural-Health-Mate technology successfully demonstrated the technical merit, feasibility and cost efficiency of combining commercially available technology with sophisticated in-home wireless technology and cloud-based intelligence. Provides rural seniors with improved coordination of care with healthcare professionals, increased medication adherence, reduced risk or impact of falls, detection of vital signs deviation from baseline. SBIR 21

U. S. Department of Agriculture Small Business Innovation Research Program Scott Dockum – Program Coordinator Waterfront Centre 800 9 th Street, SW Washington, DC 20024 Phone: (202) 720 -6346 Fax: (202) 401 -6070 E-mail: sdockum@nifa. usda. gov Web Site: www. nifa. usda. gov/fo/sbir SBIR

ANY QUESTIONS? SBIR
- Slides: 23