Sedimentary Materials Sedimentary rocks cover 80 of the













- Slides: 20

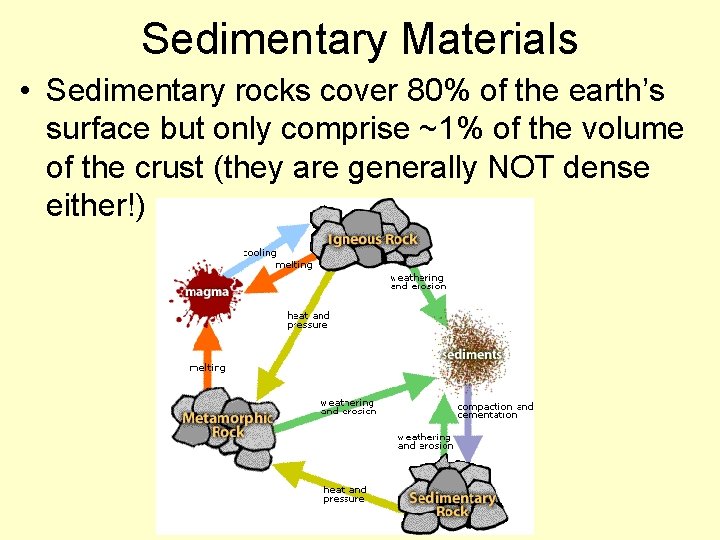
Sedimentary Materials • Sedimentary rocks cover 80% of the earth’s surface but only comprise ~1% of the volume of the crust (they are generally NOT dense either!)

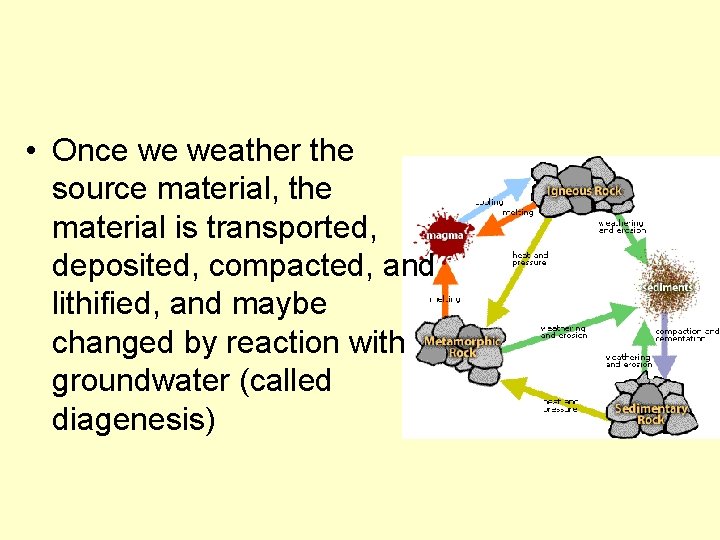
• Once we weather the source material, the material is transported, deposited, compacted, and lithified, and maybe changed by reaction with groundwater (called diagenesis)

Transport • All weathered products can be transported • Dissolved ions are transported until they get to a final destination (such as the ocean) and/ or are precipitated • Physically weathered minerals/ rock fragments How are they transported? – Water, wind, glaciers, gravity • What processes are more selective to the size of the particle

Types of sedimentary rocks • Detrital (a. k. a. clastic) form by compaction and lithification of clastic sediments or lithic fragments – Clasts are little grains or fragments of rocks (i. e. can be made of 1 or more minerals) – Classification based on size • Chemical form by precipitation of minerals from water, or by alteration of pre-existing material – Classification based on chemical composition • Biogenic formed of previously living organic debris • HOWEVER Many sedimentary rocks are combinations of 2 -3 of these types… WHY?

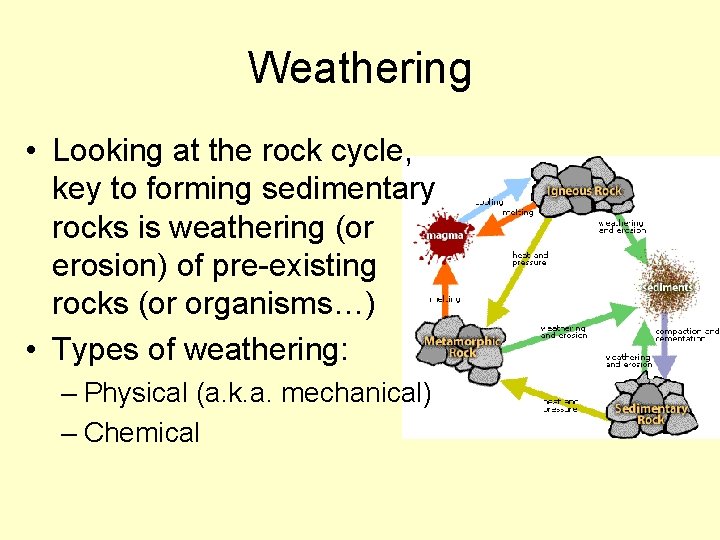
Weathering • Looking at the rock cycle, key to forming sedimentary rocks is weathering (or erosion) of pre-existing rocks (or organisms…) • Types of weathering: – Physical (a. k. a. mechanical) – Chemical

Physical Weathering • • Joints and sheeting development in rocks Frost wedging, salt wedging, biologic wedging Thermal stress Abrasion – through water, wind, glaciers, gravity, waves

Exfoliation or unloading • Some rocks expand to to pressure release, uplift, heating/ cooling, etc. and break off in sheets

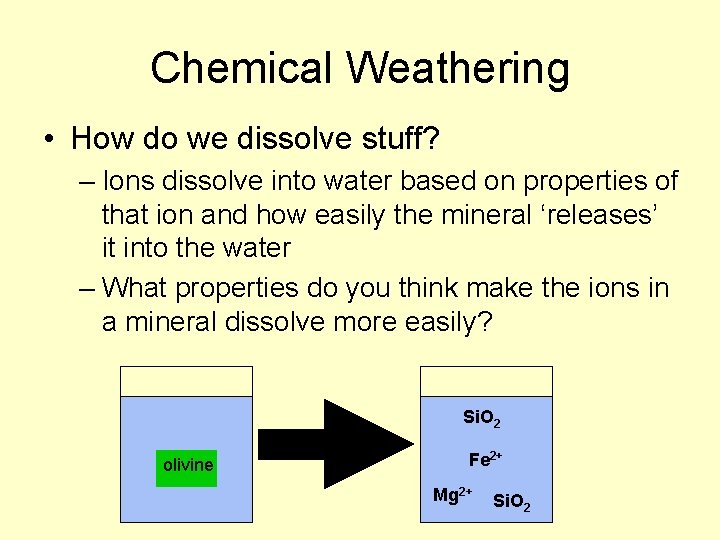
Chemical Weathering • How do we dissolve stuff? – Ions dissolve into water based on properties of that ion and how easily the mineral ‘releases’ it into the water – What properties do you think make the ions in a mineral dissolve more easily? Si. O 2 olivine Fe 2+ Mg 2+ Si. O 2

Chemical Weathering Vocabulary • Hydrolysate – dissolved material • Resistate – solid material left behind (did’t dissolve) – More easily dissolved elements include alkali and alkaline earths (Na+, Ca 2+, K+) • Residual – product of hydrolysis reactions left behind (it can be physically weathered too…)

Mineral Dissolution • Write a reaction: – Mg 0. 5 Fe 0. 5 Si. O 4 + H 2 O 0. 5 Mg 2+ + 0. 5 Fe 2+ + Si. O 44 - • Describe that reaction as an equilibrium expression which defines how much of the mineral can dissolve in a particular fluid – What aspects of fluid composition do you think might affect how much of a mineral can dissolve? – Keq=[products] / [reactants] – Keq=[Mg 2+][Fe 2+][Si. O 44 -] / [olivine][H 2 O]

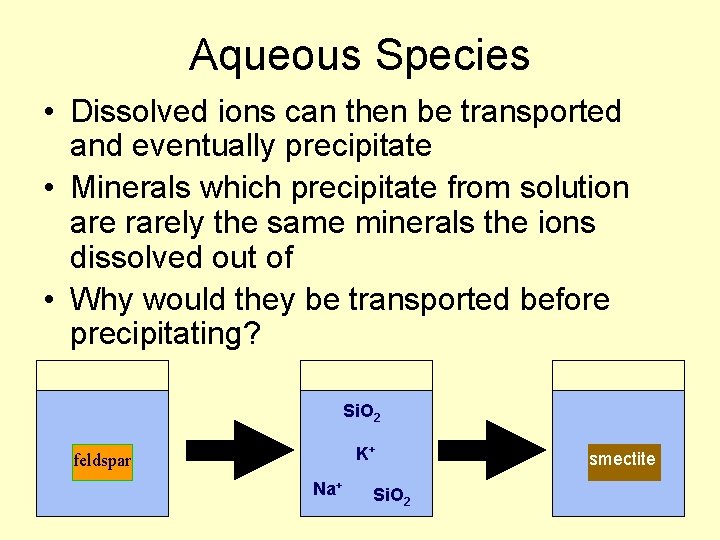
Aqueous Species • Dissolved ions can then be transported and eventually precipitate • Minerals which precipitate from solution are rarely the same minerals the ions dissolved out of • Why would they be transported before precipitating? Si. O 2 K+ feldspar Na+ Si. O 2 smectite

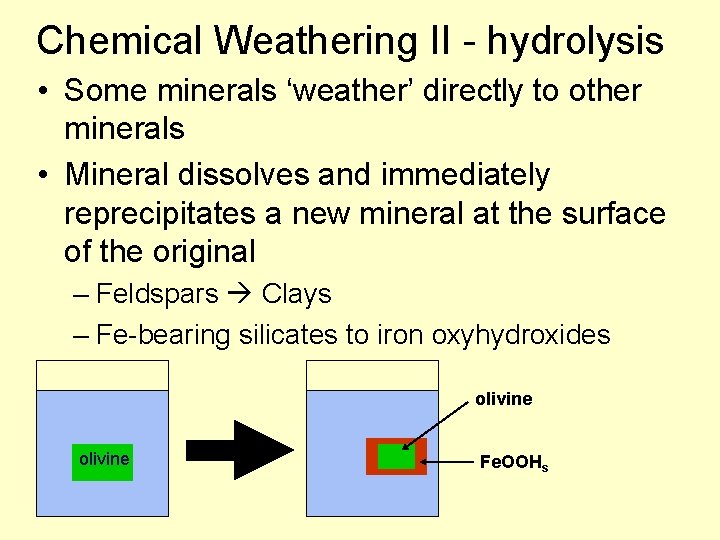
Chemical Weathering II - hydrolysis • Some minerals ‘weather’ directly to other minerals • Mineral dissolves and immediately reprecipitates a new mineral at the surface of the original – Feldspars Clays – Fe-bearing silicates to iron oxyhydroxides olivine Fe. OOHs

Acid/base reactions • Many minerals are affected by the p. H of the solution they are in – some form H+ or OH- when they dissolve – Some dissolve much faster/ better in low or high p. H solutions • Calcite weathering – Ca. CO 3 + H+ + H 2 O H 2 CO 3(g) + Ca. OH+ • Acid/ base chemistry important in mineral dissolution and precipitation!!

Oxidation • Recall that elements exist as different ions in a particular oxidation state • Changing that oxidation state can have a big effect on how well that element will dissolve and what minerals will form after it dissolves • Oxidation (where a reduced ion loses an electron to an oxidant) is important in the weathering of many minerals at the surface of the earth where O 2 is the oxidant • Fe(II)2 Si. O 4 + ½ O 2 + H 2 O 2 Fe(III)OOH + Si. O 2

Chemical Weathering • Recap: How do minerals dissolve? – Dissolution reactions • Ions dissolve in water, do not change – Acid-base reactions • Ions dissolve in water through interaction with H+ or OH- – Redox reactions • Ions dissolve/ precipitate affected by interaction of ions in mineral or in water with O 2

Chemical Weathering and Stability • All minerals are described by a ‘stability’ • Thermodynamics defines this through an energy all energies are relative • Energy changes depending on the conditions i. e. some minerals are more stable than others at high P and T; change the P and T conditions and different minerals are more stable • In weathering environments, minerals that are weathering are not stable, minerals precipitating ARE stable

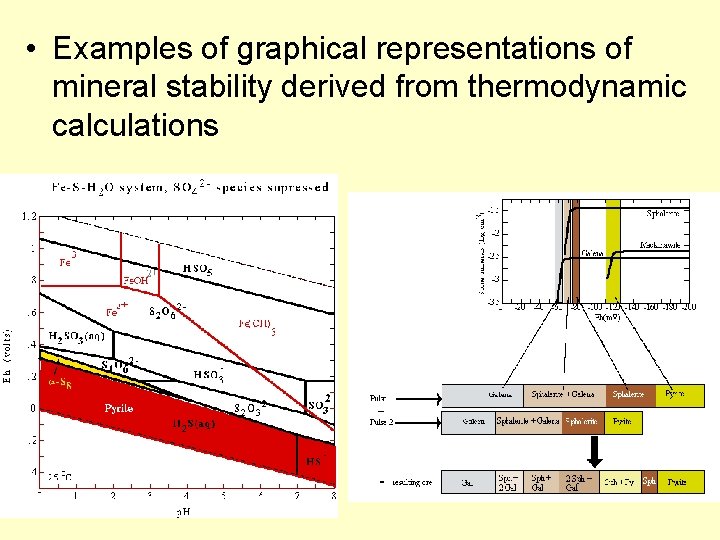
• Examples of graphical representations of mineral stability derived from thermodynamic calculations

Resistance to weathering • Goldrich series empirical observation concerning what minerals weather before others… olivine Ca-plagioclase pyroxene amphibole Na-plagioclase biotite K-feldspar quartz Remind you of anything? ?

What happens when granite is weathered? ? • First, unweathered granite contains these minerals: – – Na Plagioclase feldspar K feldspar Quartz Lesser amounts of biotite, amphibole, or muscovite • What happens when granite is weathered? • The feldspars will undergo hydrolysis to form kaolinite (clay) and Na and K ions • The Na+ and K+ ions will be removed through leaching • The biotite and/or amphibole will undergo hydrolysis to form clay, and oxidation to form iron oxides.

Granite weathering, continued • The quartz (and muscovite, if present) will remain as residual minerals because they are very resistant to weathering. • Weathered rock is called saprolite. • What happens after this? – Quartz grains may be eroded, becoming sediment. The quartz in granite is sand- sized; it becomes quartz sand. The quartz sand will ultimately be transported to the sea (bed load), where it accumulates to form beaches. – Clays will ultimately be eroded and washed out to sea. Clay is finegrained and remains suspended in the water column (suspended load); it may be deposited in quiet water. – Dissolved ions will be transported by rivers to the sea (dissolved load), and will become part of the salts in the sea.