Section 8 5 Solubility Saturation Saturation of a

Section (8. 5) Solubility & Saturation

Saturation of a Solution • A saturated solution is formed when no more solute will dissolve in a solution, and excess solute is present o It has the maximum amount of solute it can hold • An unsaturated solution is a solution that is not yet saturated. It can still dissolve more solute

Solubility is defined as: The amount of solute that dissolves in a given quantity of solvent, at a certain temperature, to form a saturated solution o Usually stated in grams of solute per 100 g of water at a given temperature. o E. g. the solubility of sodium chloride in water at 20°C is 36 g per 100 m. L of water Insoluble if solubility < 0. 1 g per 100 m. L solvent

Solubility Recall, solubility defined as: The amount of solute that dissolves in a given volume of solvent, at a specific temperature, to form a saturated solution. For most solids: as T increases, solubility increases For most gases: as T increases, solubility decreases as P decreases, solubility decreases

Factors that Affect Rate of Dissolving 1. Particle size: Finely divided substances have more surface area and dissolve more rapidly 2. Motion: Stirring or shaking brings more solvent in contact with more solute particles, increasing solubility 3. Temperature: The solubility of most solids increases with temperature. The solubility of gases decreases with temperature.

Summary of Factors that Affect Solubility 1. The polarity of the substances: LIKE dissolves LIKE 2. The nature of the substances: Intermolecular bonds 3. The temperature: Some substances only dissolve above certain temperatures 4. The pressure: Some substances only dissolve under pressure
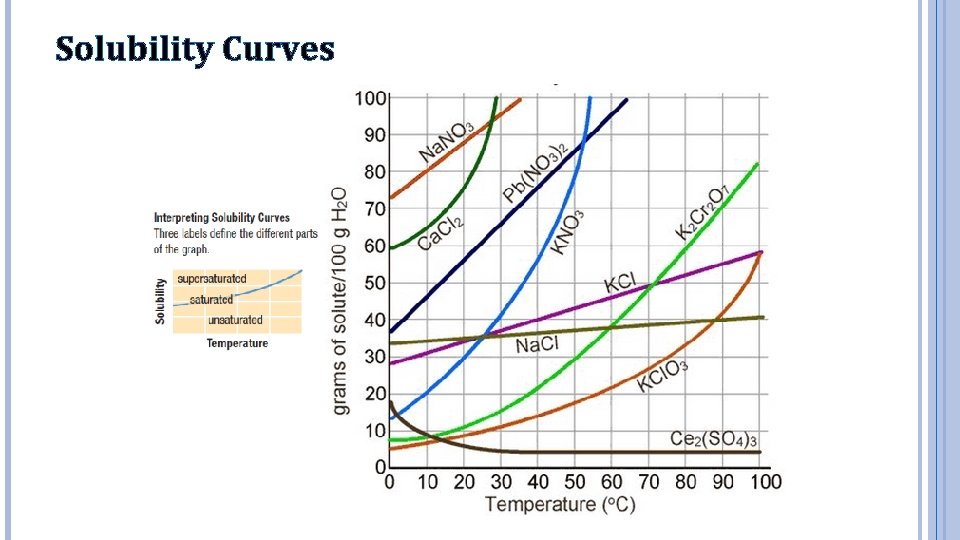
Solubility Curves

Using Solubility Curves 1. Which salt is least soluble at 40°C? a) b) c) d) Na. Cl K 2 Cr 2 O 7 KCl. O 3 KNO 3 2. Which of the following decreases in solubility as the temperature increases? a) b) c) d) KCl Ce 2(SO 4)3 Na. NO 3 KCl. O 3

Using the Solubility Graph 3. How many grams of lead (II) nitrate, Pb(NO 3)2 are soluble in 100 g of water at 30°C? a) b) c) d) 40 g 45 g 65 g 130 g 4. At what temperature does the solubility of sodium chloride match the solubility of potassium chlorate? a) b) c) d) 25°C 60°C 90°C 100°C

Using the Solubility Graph 5. When 70 g of calcium chloride are dissolved in 100 g of water at 20°C, the solution can be described as: a) saturated b) unsaturated 6. A solution of potassium chloride has 35 g of the salt dissolved in 200 g of water at 60°C. How many more grams of the salt can be added to the solution before reaching the saturation point? a) b) c) d) none, it is already saturated 15 g 25 g 50 g
- Slides: 10