Section 8 1 Understanding Reactions in Aqueous Solutions

















- Slides: 18

Section 8. 1 Understanding Reactions in Aqueous Solutions Objectives 1. To learn more about some of the results of chemical reactions 2. To learn to predict the solid that forms in a precipitation reaction 3. To learn to write molecular, complete ionic, and net ionic equations

Section 8. 1 Understanding Reactions in Aqueous Solutions A. Common Results of a Reaction • Four common events accompany chemical reactions. These often occur when a reaction happens – Formation of a solid – Formation of water – Formation of a gas – Transfer of electrons Can you give examples of each of these?

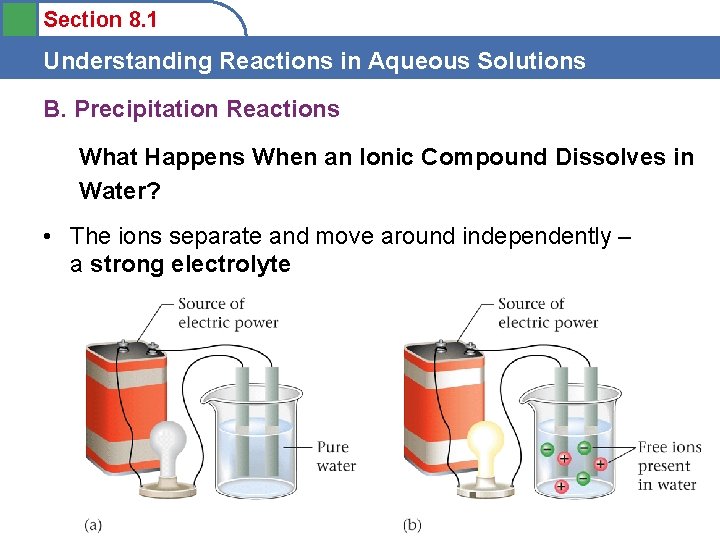
Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions What Happens When an Ionic Compound Dissolves in Water? Na. Cl (s) H 2 O Na. Cl (aq) But what does Na. Cl (aq) mean? Na. Cl molecules in water ? Na and Cl atoms in water? Na+ and Cl- ions in water ?

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions What Happens When an Ionic Compound Dissolves in Water? • The ions separate and move around independently – a strong electrolyte

Section 8. 1 Understanding Reactions in Aqueous Solutions B. A Reaction in Which a Solid Forms • A reaction in which a solid is formed from a solution is called a precipitation reaction – Solid = precipitate = insoluble When positive and negative ions form a precipitate the net charge of the precipitate is zero Pb(NO 3)2 (aq) + 2 Na. I (aq) Pb. I 2 (s) + 2 Na. NO 3 (aq)

Section 8. 1 Understanding Reactions in Aqueous Solutions Stalactites in Yosemite Ca(HCO 3)2(aq) → Ca. CO 3(s) + H 2 O(l) + CO 2(aq)

Section 8. 1 Understanding Reactions in Aqueous Solutions

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions What Happens When Two Ionic Compounds Dissolve in Water? • K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) Products

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions How to Decide What Products Form • Determine the possible products from the ions in the reactants. • In our example K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) Products The possible ion combinations are

Section 8. 1 Understanding Reactions in Aqueous Solutions What are the potential products in the reactions between the following aqueous solutions? – Write balanced equations for each • Na. OH + Ca. Cl 2 • Cu. Br 2 + (NH 4)2 CO 3 • K 2 SO 4 + Fe(NO 3)3

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions How Would We Decide What Product Has Formed? • What is most likely to be the yellow solid formed in the following reaction? K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) • The possible product combinations are KNO 3 and Ba. Cr. O 4 – KNO 3 white solid – Ba. Cr. O 4 yellow solid

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions – Solubility Rules

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions Using Solubility Rules • Predicting Precipitates – Soluble solid – Insoluble solid – (Slightly soluble solid) Why are some compounds soluble and others not?

Section 8. 1 Understanding Reactions in Aqueous Solutions Solubility Rules – Are These Compounds Soluble or Insoluble? Barium Nitrate Potassium Carbonate Sodium Sulfate Copper (II) Hydroxide Mercury (I) Chloride Ammonium Phosphate Chromium (III) Sulfide Lead (II) Sulfate

Section 8. 1 Understanding Reactions in Aqueous Solutions B. Precipitation Reactions Let’s try some predictions: WOC P 271 Q 10

Section 8. 1 Understanding Reactions in Aqueous Solutions C. Describing Precipitation Reactions in Aqueous Solutions • Three types of equations: – Molecular (formula) equation complete formulas of all reactants and products – Complete ionic equation all strong electrolytes are shown as ions – Net ionic equation only those components of the solution that undergo a change • Spectator ion (those that remain unchanged) not shown in the net ionic equation

Section 8. 1 Understanding Reactions in Aqueous Solutions C. Describing Precipitation Reactions in Aqueous Solutions • Molecular Equation Pb(NO 3)2 (aq) + 2 Na. I (aq) Pb. I 2 (s) + 2 Na. NO 3 (aq) • Complete Ionic Equation Pb 2+(aq) + 2 NO 3 -(aq) + 2 Na+(aq) + 2 I-(aq) Pb. I 2(s) + 2 Na+(aq) + 2 NO 3 -(aq)

Section 8. 1 Understanding Reactions in Aqueous Solutions C. Describing Precipitation Reactions in Aqueous Solutions • Net Ionic Equation Pb 2+(aq) + 2 I-(aq) Pb. I 2(s) • What are the spectator ions in this reaction? A solution of sodium chloride in water reacts with aqueous silver nitrate to give a precipitate of silver chloride – write down the three equations that describe this reaction and identify the spectator ions (P 272 Q 14 – all three equations and identify spectators)