Section 6 4 Lipids Objectives Distinguish among proteins

Section 6. 4 Lipids

Objectives • Distinguish among proteins, carbohydrates, lipids and nucleic acids. (SPI 3210. 1. 3) – Identify a general characteristic of lipids. – Describe the structure and function of fats. – Describe the structure and function of steroids. • Identify positive tests for carbohydrates, lipids and proteins (SPI 3210. 1. 4)

LIPIDS From the Greek lipos meaning fat. Varied structure Hydrophobic (insoluble in water) ─ lack polar groups.

4 Classes of Lipids • Triglycerides (Fats & Oils - Most Familiar Lipids) – Energy reserves – Cushioning – Insulation • Waxes – Waterproof – Resistant to degradation • Phospholipids – Structure similar to fats – Polar – Part membranes in cells • Steroids – Different structure from fats (Carbon ring backbone) – Cell messengers

Functions of Fats Energy Storage Cushioning Insulation

Fats & Oils (Triglycerides) Saturated animal fats are associated with circulatory disorders Plant oils can be substituted for animal fats in the diet.
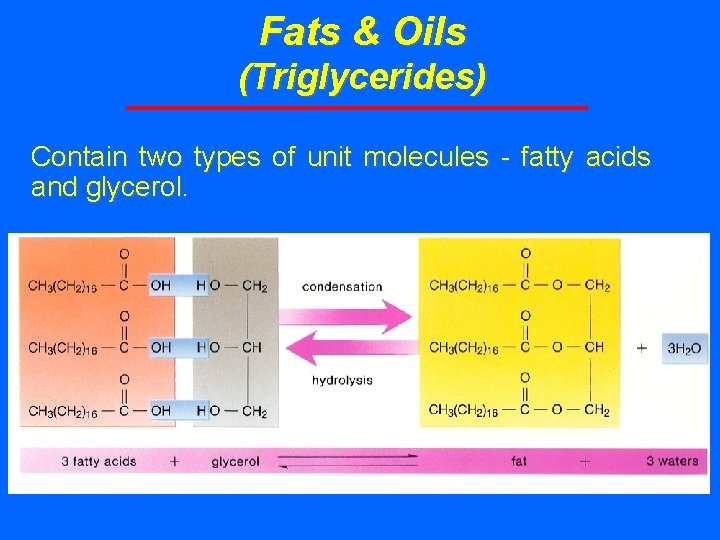
Fats & Oils (Triglycerides) Contain two types of unit molecules - fatty acids and glycerol.

FATTY ACID A lipid unit molecule Fatty acid - long hydrocarbon chain, carboxyl (-COOH), [acid] group at one end. Carboxyl group is polar, fatty acids are soluble in water. Most fatty acids in cells contain 16 to 18 carbons atoms per molecule.
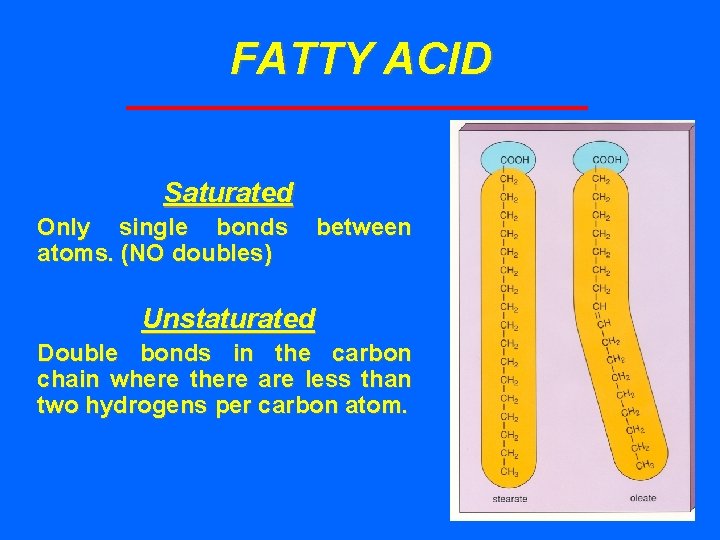
FATTY ACID Saturated Only single bonds atoms. (NO doubles) between Unstaturated Double bonds in the carbon chain where there are less than two hydrogens per carbon atom.
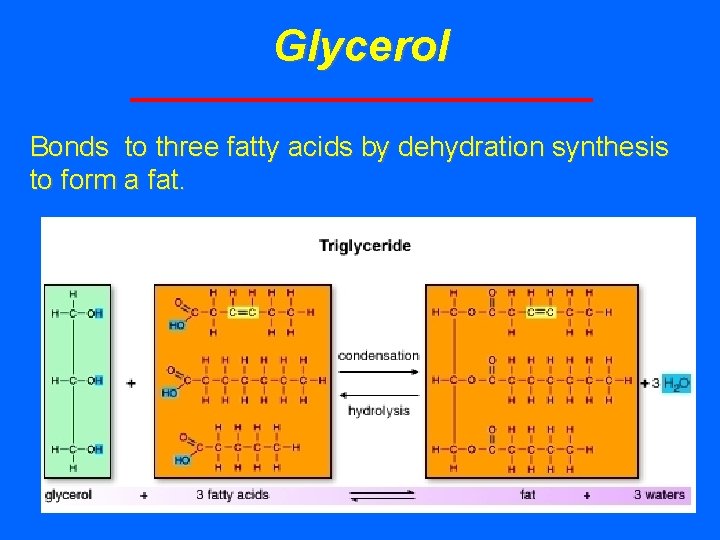
Glycerol Bonds to three fatty acids by dehydration synthesis to form a fat.

Glycerol (cont’d) Fats Triglycerides containing saturated fatty acids (e. g. butter is solid at room temperature) Oils Triglycerides with unsaturated fatty acids (e. g. Corn oil is liquid at room temperature). Animals use fat rather then glycogen long-term energy storage; fat stores more energy.

WAXES • • • Fatty acids bonded alcohol instead of glycerol. Solid at room temperature High melting point Waterproof Resist degradation. Form protective coverings – Retards water loss in plants – Maintain animal skin and fur.

PHOSPHOLIPIDS Differ from neutral fats - one fatty acid is replaced by phosphate group or a group with both phosphate and nitrogen.
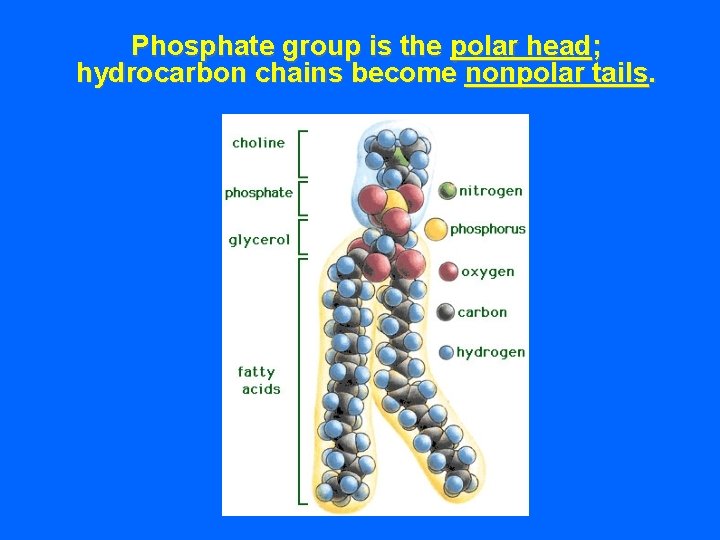
Phosphate group is the polar head; hydrocarbon chains become nonpolar tails.

PHOSPHOLIPIDS In aqueous environments, arrange in a double layer. Polar (hydrophilic) heads toward water molecules. Nonpolar (hydrophobic) tails face toward each other - away from water molecules. Very important property! Forms an interface or separation between two solutions (e. g. The interior and exterior of a cell

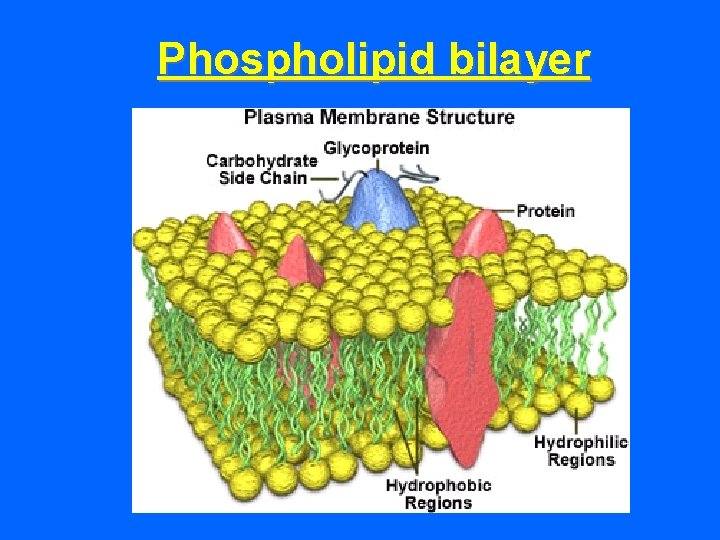
Phospholipid bilayer

STEROIDS Differ from neutral fats - backbone of four fused carbon rings Functions vary according to attached functional groups.
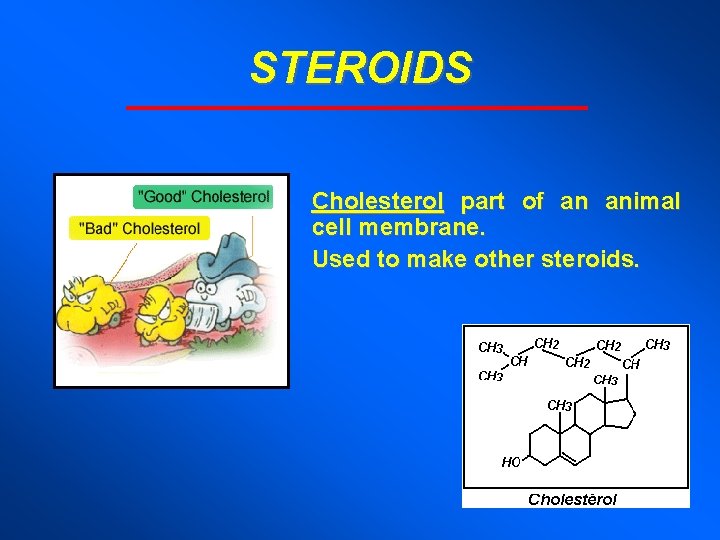
STEROIDS Cholesterol part of an animal cell membrane. Used to make other steroids.
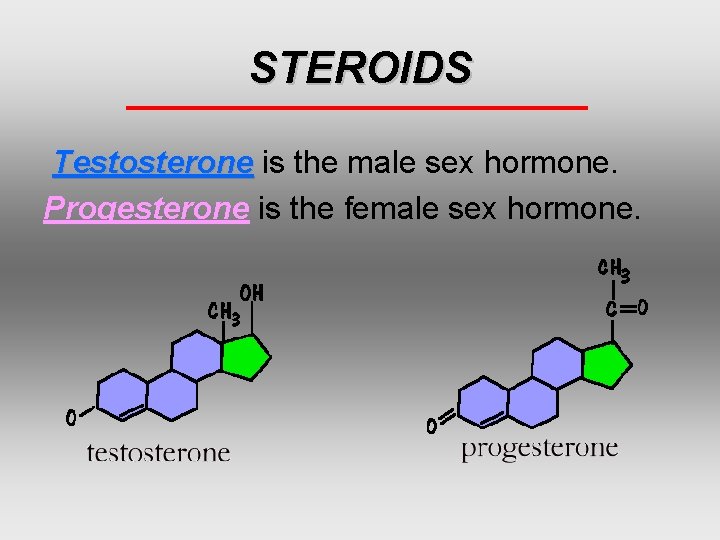
STEROIDS Testosterone is the male sex hormone. Progesterone is the female sex hormone.

Brown Paper Test for Lipids • Put sample on brown paper and let stand for several minutes. • Positive test – translucent spot remains.

Review • Identify a general characteristic of lipids. • Describe the structure and function of fats. • Describe the structure and function of steroids.
- Slides: 22