Section 4 Aldehydes Ketones Carbonyl Groups Are a
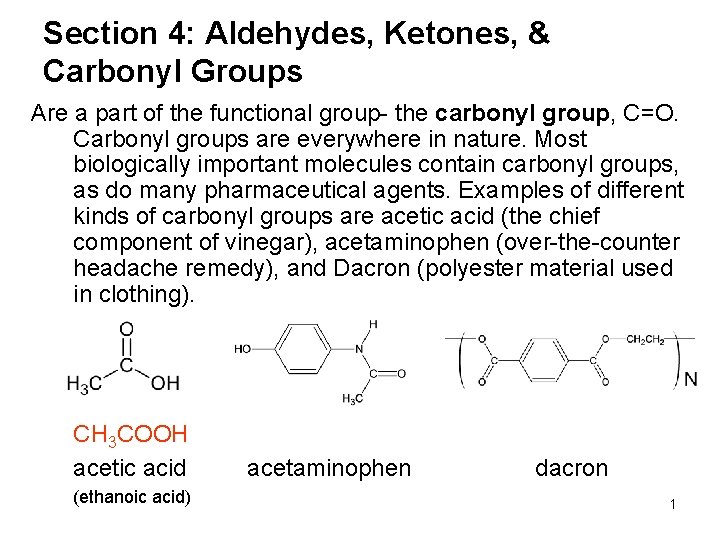
Section 4: Aldehydes, Ketones, & Carbonyl Groups Are a part of the functional group- the carbonyl group, C=O. Carbonyl groups are everywhere in nature. Most biologically important molecules contain carbonyl groups, as do many pharmaceutical agents. Examples of different kinds of carbonyl groups are acetic acid (the chief component of vinegar), acetaminophen (over-the-counter headache remedy), and Dacron (polyester material used in clothing). CH 3 COOH acetic acid (ethanoic acid) acetaminophen dacron 1

Types of Carbonyl Compounds: All carbonyl groups contain an acyl group, R 2 -C=O aldehyde ketone (R≠H) acid anhydride carboxylic acid halide ester (R≠H) amide 2

Carbonyl compounds are classified into two categories based on the kind of chemistry they undergo. 1. Aldehydes & ketones: The aldehydes and ketones are bonded to substituents that cannot stabilize a negative charge and therefore cannot act as leaving groups. Aldehydes and ketones behave similarly and undergo many of the same reactions. 2. Carboxylic acids and their derivatives. The acyl group in the carboxylic acids and their derivatives are bonded to substituents (oxygen, halogen, nitrogen) that can stabilize a negative charge and can serve as leaving groups in substitution reactions. The chemistry of these compounds is therefore similar. 3
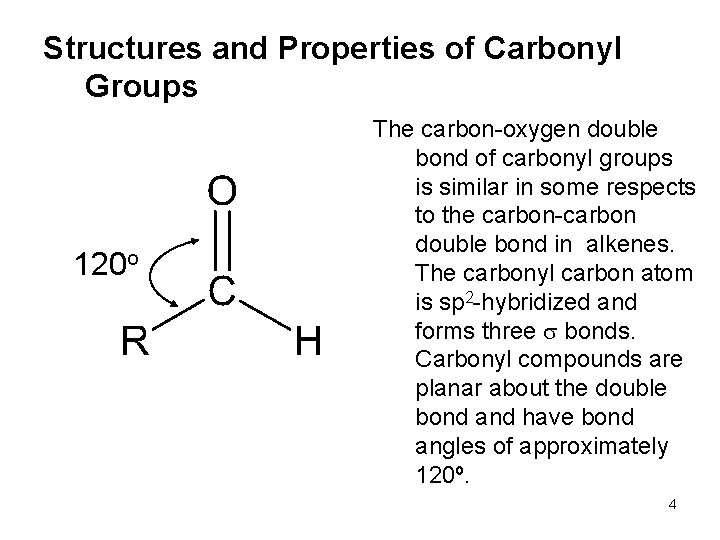
Structures and Properties of Carbonyl Groups 120 o The carbon-oxygen double bond of carbonyl groups is similar in some respects to the carbon-carbon double bond in alkenes. The carbonyl carbon atom is sp 2 -hybridized and forms three bonds. Carbonyl compounds are planar about the double bond and have bond angles of approximately 120º. 4
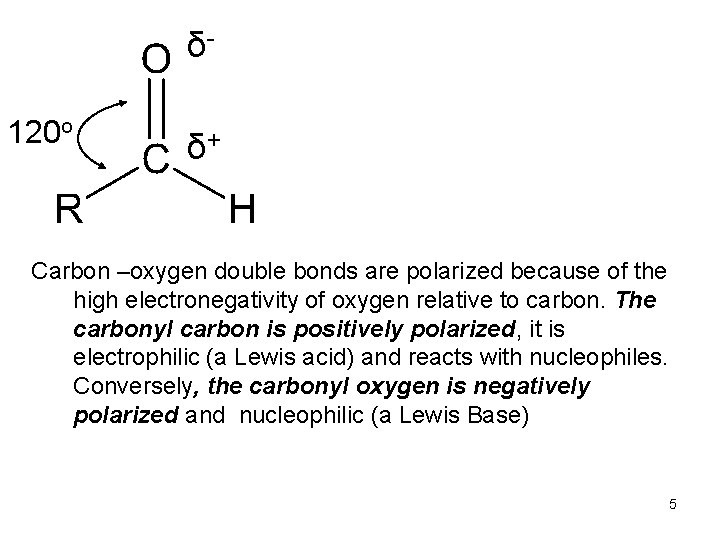
δ 120 o δ+ Carbon –oxygen double bonds are polarized because of the high electronegativity of oxygen relative to carbon. The carbonyl carbon is positively polarized, it is electrophilic (a Lewis acid) and reacts with nucleophiles. Conversely, the carbonyl oxygen is negatively polarized and nucleophilic (a Lewis Base) 5
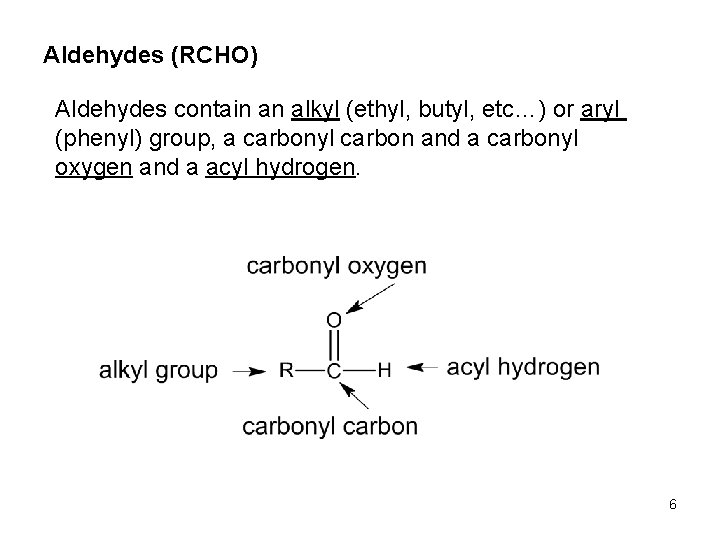
Aldehydes (RCHO) Aldehydes contain an alkyl (ethyl, butyl, etc…) or aryl (phenyl) group, a carbonyl carbon and a carbonyl oxygen and a acyl hydrogen. 6

Nomenclature of Aldehydes (IUPAC) A. Aldehydes are named by replacing the terminal –e of the corresponding alkane with an –al B. The longest chain selected as the parent name must contain the –CHO group. C. The –CHO carbon is always numbered as carbon 1 D. When an aldehyde is on a ring, the –e stays and the last name becomes carbaldehyde, the carbon of the ring the aldehyde is attached to is carbon 1 Examples: Formula HCHO CH 3 CH 2 CHO CH 3 CH 2 CH 2 CHO CH 2=CHCHO Common Name formaldehyde acetaldehyde propionaldehyde butyraldehyde valeraldehyde acrolein IUPAC methanal propanal butanal pentanal 2 -propenal 7
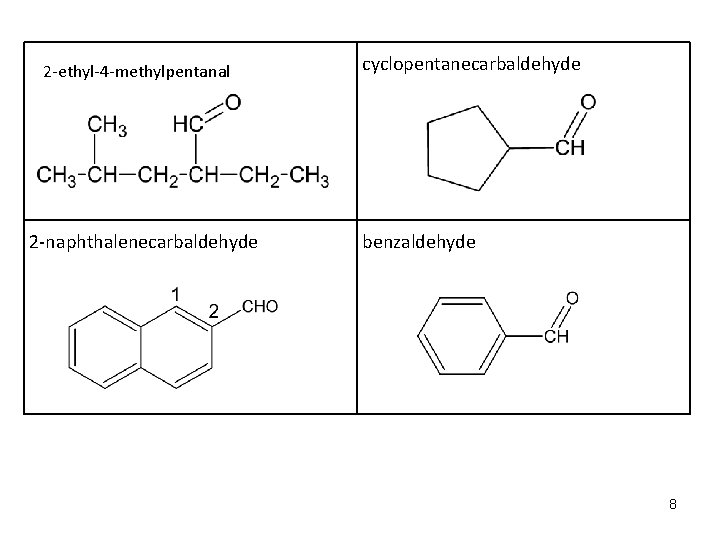
2 -ethyl-4 -methylpentanal 2 -naphthalenecarbaldehyde cyclopentanecarbaldehyde benzaldehyde 8

Nomenclature of Ketones (IUPAC) 1. Ketones are named by replacing the terminal –e of the corresponding alkane name with –one. 2. The parent chain is the longest chain that contains the ketone group. 3. Numbering begins at the end nearer the carbonyl carbon. 4. Common names for ketones are constructed by giving, in alphabetical order, the names of the groups attached, and then adding the word ketone 9

Examples: Propanone (acetone) (dimethyl ketone) 3 -hexanone (ethyl 1 -propyl ketone) 4 -hexen-2 -one 10

Problems: Name or draw the structures of the following: 2 -methyl-3 -pentanone 3 -chlorocyclohexanone 2 -methyl propanal 3 -methylbutanal 2 -bromobenzaldehyde o-bromobenzaldehyde 11

Physical Properties of Aldehydes A. B. The melting point of aldehydes varies nonsystematically with increasing molecular weight. Aldehydes have higher boiling points than the boiling points of alkanes of similar molecular weight, due to dipole–dipole interaction δ+ δ- C. The boiling points of aldehydes are lower than the corresponding alcohol due to the lack of hydrogen bonding interactions. D. There is a systematic increase of the boiling point with molecular weight. 12

Physical Properties of Ketones A. The melting point of ketones is systematic with increasing molecular weight. B. Ketones have higher boiling points than the boiling points of alkanes of similar molecular weight. C. The boiling points of ketones are lower than the corresponding alcohol due to the lack of hydrogen bonding interactions. D. There is a systematic increase of the boiling point with molecular weight. 13

Synthesis of Aldehydes & Ketones The best method of preparing aldehydes and ketones is alcohol oxidation. Primary alcohols are oxidized to give aldehydes, and secondary alcohols are oxidized to give ketones. Pyridinium chlorochromate (PCC) in dichloromethane is usually chosen for making aldehydes, while PCC, Cr. O 3, and Na 2 Cr 2 O 7 are all-effective for making ketones. 1. Oxidation of 1 o alcohol to form aldehydes and carboxylic acids + H 2 O Primary alcohol aldehyde carboxylic acid Mild oxidation stops at aldehyde. Strong or Continued oxidation results in the formation of the corresponding carboxylic acid 14

2. Oxidation of 2 o alcohol to form ketones + H 2 O 3. Hydration of a terminal alkyne to yield a methyl ketone 4. Friedel-Crafts acylation of an aromatic ring to yield an aryl ketone 15

Problems: Complete the following reactions Prepare pentanal from the appropriate alcohol. + H 2 O Prepare 2 -hexanone from the appropriate alcohol. + H 2 O 16

+ H 2 O 4 -tert-butylcyclohexanol 1 -hexyne benzene acetyl chloride 4 -tert-butylcyclohexanone 2 -hexanone methyl phenyl ketone (acetophenone) 17

Reactions of Aldehydes Oxidation of Aldehydes: (Ketones DO NOT oxidize) • Aldehydes are oxidized to the corresponding carboxylic acid. RCHO RCOOH. This reactivity is consequence of structure. • Aldehydes have a –CHO hydrogen bonded to the carbonyl carbon that can be removed during oxidation, but ketones do not. • Ketones are unreactive towards oxidation. 18

1. Tollens Test: (Qualitative test for aldehydes) One of the simplest methods for oxidizing an aldehyde is to use silver ion, Ag+, in dilute ammonia. (Tollens’ reagent) As the oxidation proceeds, a shiny mirror of silver metal is deposited on the walls of the reaction flask, forming the basis of a simple test to detect the presence of an aldehyde functional group in a sample of unknown. A small amount of unknown is dissolved in ethanol in a test tube, and a few drops of Tollens’ reagent are added. If silver mirror appears, the unknown is an aldehyde. Aldehyde + Ag+(aq) carboxylic acid + Ag (s) 19

2. Benedicts Reagent Oxidation (Qualitative test for aldehydes). Another method for oxidizing an aldehyde is to use copper in a strong base, such as Na. OH. If a red precipitate forms, the unknown is an aldehyde. RCHO + 2 Cu 2+ + 5 OH- RCOO- + Cu 2 O + 3 H 2 O blue solution red ppt 3. Jones oxidation: Jones reagent: Cr. O 3/CH 3 COOH 20

Complete the following reactions: No Reaction 21

Nucleophilic Addition Reactions of Aldehyes and Ketones The most common reaction of aldehydes and ketones is the nucleophilic addition reaction, in which a nucleophile (: Nu or : Nu-) adds to the electrophilic carbon of the carbonyl group. Nucleophilic oxygen reacts with acids and electrophiles (positive species) Aldehydes are generally more reactive than ketones in nucleophilic additions for steric reasons. δδ+ Nucleophilic oxygen reacts with acids and electrophiles (positive species) Electrophilic carbon reacts with bases and nucleophiles (negative species or contain lone pair(s) of e-) 22

Negatively Charged Nucleophiles (Anions) : OH: HR 3 C: RO: : CNhydroxide hydride carbanion alkoxide cyanide Generalized nucleophilic addition reaction for negative nucleophiles. Mechanism for the above reaction: ‡ Aldehyde or ketone tetrahedrally hybridized Intermediate 23

Nucleophilic Addition Reactions for Neutral Nucleophiles HOH RNH 2 (water) 1. Hydration Reaction (alcohol) 2. Acetal Formation (ammonia, 1 o amine) 3. Imine Formation 1. Hydration reactions: Nucleophilic additions of water. Aldehydes and ketones undergo a nucleophilic addition reaction with water to yield 1, 1 -diols, or geminal (gem) diols. The reaction is reversible, and a gem diol can eliminate water to regenerate a ketone or aldehyde. The reaction is slow in pure water but is catalyzed by both acids and bases. Aldehyde or Ketone 1, 1 -diol 24

2. Acetal formation: Nucleophilic addition reactions of alcohols. Aldehydes and ketones react with alcohols in the presence of an acid catalyst to yield acetals R 2 C(OR’)2, compounds that have two ether-like –OR groups bonded to the same carbon. Aldehyde or Ketone acetal 25

3. Imine formation: Nucleophilic addition of amines. Aldehydes and ketones react with ammonia and primary amines, R’NH 2, to yield imines, R 2 C=NR’. Imines are formed by the addition to the carbonyl group of the nucleophilic amine, followed by the loss of water from the amino alcohol product. Aldehyde or Ketone Imine 26

Nucleophilic Addition of Grignard and Hydride Reagents: Alcohol Formation Unlike the nucleophilic additions of HOH and HCN, these are NOT reversible because the carbanion group is too poor of a leaving group Grignard Reactions: Grignard reagents react with aldehydes and ketones to produce alcohol. First step is to make (prepare) the Grignard reagent (R-Mg-X). Preparation of Grignard Reagent: R-Mg-X R-X + Mg 0 Example: CH 3 CH 2 Br + Mgo ether R-Mg-X CH 3 CH 2 Mg. Br 27

Using the Grignard reagent to make 1 o, 2 o & 3 o ROH. Formaldehyde 1 o ROH Aldehydes 2 o ROH Not formaldehyde Ketones 3 o ROH 28
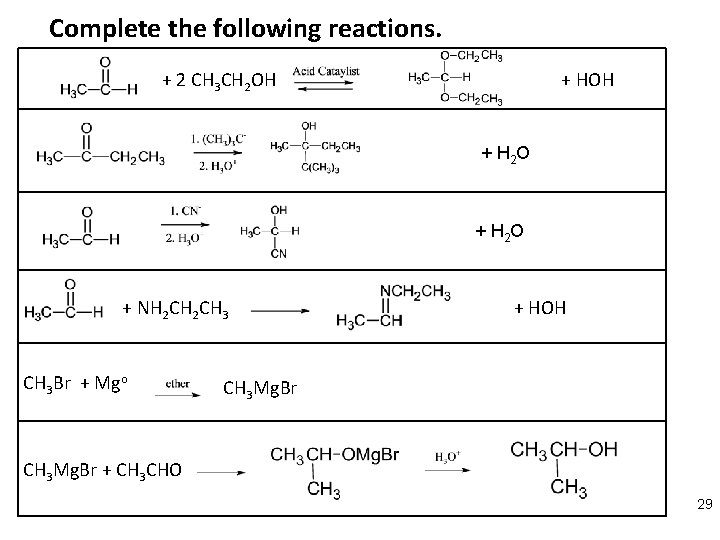
Complete the following reactions. + 2 CH 3 CH 2 OH + H 2 O + NH 2 CH 3 Br + Mgo + HOH CH 3 Mg. Br + CH 3 CHO 29

Carboxylic Acids & Their Derivatives Carboxylic acids and their derivatives are carbonyl compounds in which the acyl group is bonded to electronegative atom such as oxygen, halogen, nitrogen, or sulfur. Although there are many different kinds of carboxylic acid derivatives, will be concerned only with four of the most common types in addition to the acids themselves: acid halides, acid anhydrides, esters, and amides. In contrast to aldehydes and ketones, these compounds contain acyl group bonded to the substituent that can act as a leaving group in substitution reactions. We will also discuss nitrile (R-CΞN), a class of compounds closely related to carboxylic acids. 30

Nomenclature for carboxylic acids: RCOOH 1. Noncyclic carboxylic acids are named by replacing the -e of the corresponding alkane by -oic acid. The -COOH carbon (the carboxylic carbon) is always numbered C 1. 2. Compounds that have a carboxylic acid bonded to a ring are named by using the suffix -carboxylic acid. In this alternate system, the carboxylic acid carbon is attached to C 1 on the ring but is not itself numbered. 3. Many carboxylic acids have historical, non-systematic names. There a large number of carboxylic acids with common names, because many carboxylic acids were among the first organic compounds to be isolated and purified. 31

Formula Common Name HCOOH formic acid CH 3 COOH acetic acid CH 3 CH 2 COOH propionic acid CH 3 CH 2 COOH butyric acid CH 3(CH 2)3 COOH valeric acid CH 3(CH 2)16 COOH steric acid IUPAC methanoic acid propanoic acid butanoic acid pentanoic acid octadecanoic acid 32
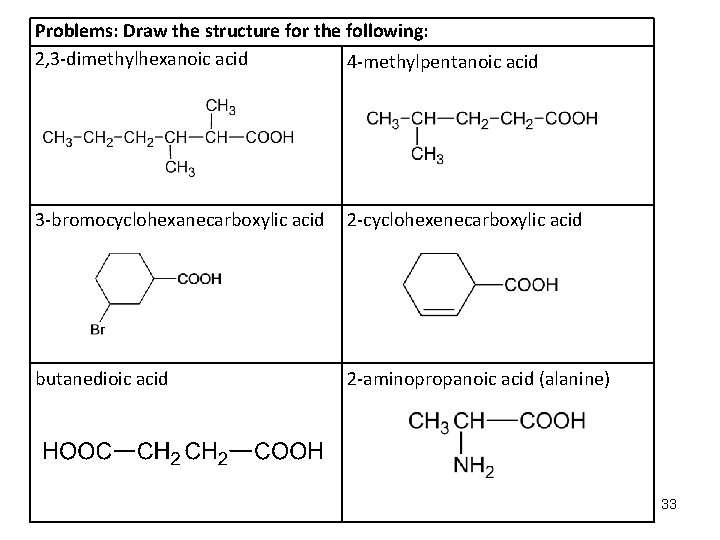
Problems: Draw the structure for the following: 2, 3 -dimethylhexanoic acid 4 -methylpentanoic acid 3 -bromocyclohexanecarboxylic acid 2 -cyclohexenecarboxylic acid butanedioic acid 2 -aminopropanoic acid (alanine) 33

Give the IUPAC name for the following: 4 -hexenoic acid 3 -methylbutanoic acid 2 -hydroxypropanoic acid (lactic acid) 4 -bromopentanoic acid 34

Physical Properties of Carboxylic Acids A. Carboxylic acids are strongly associated because of hydrogen bonding. Most carboxylic acids exist as dimers held together by two hydrogen bonds. This strong hydrogen bonding results in a higher boiling point than the corresponding alcohol. Bond angles are close to 120º Carboxylic acid dimer with hydrogen bonding 35
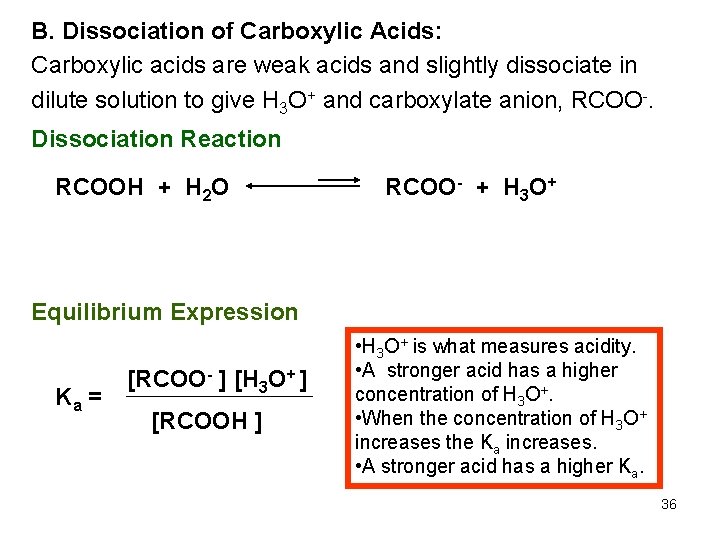
B. Dissociation of Carboxylic Acids: Carboxylic acids are weak acids and slightly dissociate in dilute solution to give H 3 O+ and carboxylate anion, RCOO-. Dissociation Reaction RCOOH + H 2 O RCOO- + H 3 O+ Equilibrium Expression Ka = [RCOO- ] [H 3 O+ ] [RCOOH ] • H 3 O+ is what measures acidity. • A stronger acid has a higher concentration of H 3 O+. • When the concentration of H 3 O+ increases the Ka increases. • A stronger acid has a higher Ka. 36
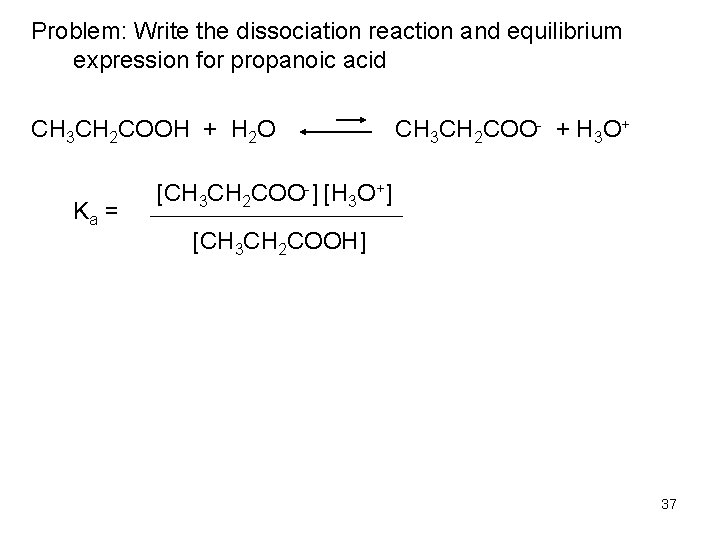
Problem: Write the dissociation reaction and equilibrium expression for propanoic acid CH 3 CH 2 COOH + H 2 O Ka = CH 3 CH 2 COO- + H 3 O+ [CH 3 CH 2 COO-] [H 3 O+] [CH 3 CH 2 COOH] 37

Synthesis of Carboxylic Acids Oxidation of Primary Alcohols 1 o alcohol aldehyde carboxylic acid Oxidation of Aldehydes aldehyde carboxylic acid 38

Grignard Reactions: Grignard reagents act as carbon nucleophiles toward carbonyl compounds. Making carboxylic acid via Grignard. The R group of the Grignard reagent is added irreversibly to the carbon on CO 2, forming a new carbon-to-carbon bond, increasing the chain by one carbon. Recall the Preparation of Grignard Reagent: R-Mg-X R-X + Mg 0 ether R-Mg-X This reaction is typically shown in two steps, first the preparation of the Grignard Reagent, and the second step the reaction of the Grignard Reagent with CO 2. CH 3 CH 2 -Br + Mgo CH 3 CH 2 -Mg-Br

Reactions of Carboxylic Acids Ester Formation: Fisher Esterification All steps are reversible, and the position of equilibrium can be driven forward or backward depending on the reaction conditions. Ester formation is favored when alcohol is used as the solvent, but carboxylic acid is favored when water is used as the solvent. Carboxylic acids undergo acid catalyzed reactions with alcohols to yield esters: Ester hydrolysis, the reverse of the reaction above, is the acid catalyzed reverse reaction of an ester with water to yield an alcohol and a carboxylic acid. 40
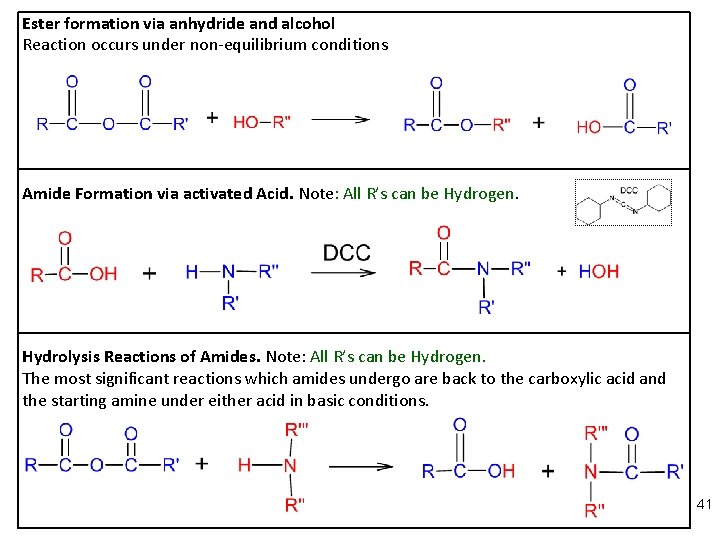
Ester formation via anhydride and alcohol Reaction occurs under non-equilibrium conditions Amide Formation via activated Acid. Note: All R’s can be Hydrogen. Hydrolysis Reactions of Amides. Note: All R’s can be Hydrogen. The most significant reactions which amides undergo are back to the carboxylic acid and the starting amine under either acid in basic conditions. 41

Problem: Complete the reactions: 42

Acid Anhydrides: How they are made? With a 3 step reaction. 43

Problem: Prepare 44
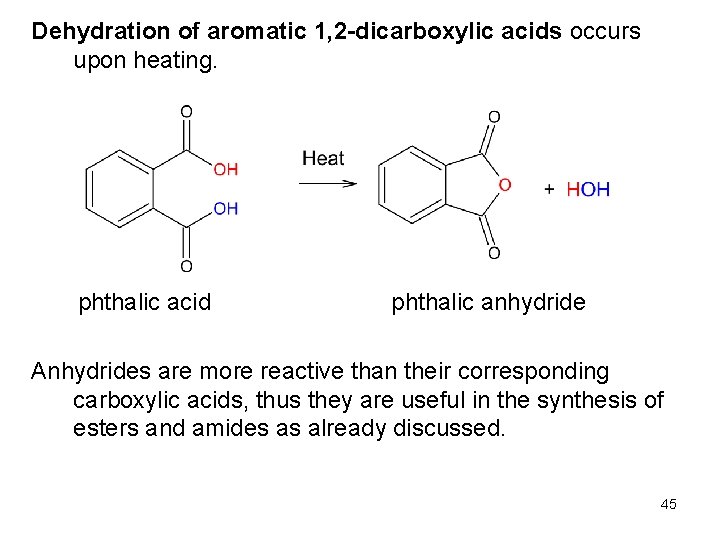
Dehydration of aromatic 1, 2 -dicarboxylic acids occurs upon heating. phthalic acid phthalic anhydride Anhydrides are more reactive than their corresponding carboxylic acids, thus they are useful in the synthesis of esters and amides as already discussed. 45
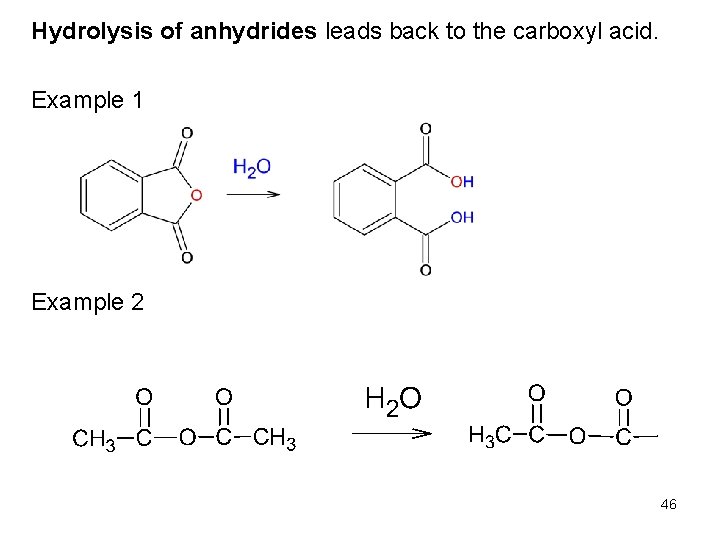
Hydrolysis of anhydrides leads back to the carboxyl acid. Example 1 Example 2 46

Problem: Complete the hydrolysis reaction. 47
- Slides: 47