Section 4 1 Naming Binary Compounds Section 4

Section 4. 1 Naming Binary Compounds

Section 4. 1 Naming Binary Compounds Objectives 1. To learn to name binary compounds containing only nonmetals (Greek Prefixes) 2. To learn to name Ionic compounds of a metal and nonmetal by balancing charges (Get a little gangsta!) 3. To learn to name Ionic compounds whose cation has more than 1 possible charge (Roman Numeral Notation)

Section 4. 1 Naming Binary Compounds B. Naming Binary Compounds That Contain Only Nonmetals (Greek Prefixes)

Section 4. 1 Naming Binary Compounds B. Naming Binary Compounds That Contain Only Nonmetals (These are the Greek Prefixes)

Section 4. 1 Naming Binary Compounds Binary Compound Practice • BF 3 • • • NO N 2 O 5 H 2 O CCl 4 NO 2 IF 5

Section 4. 1 Naming Binary Compounds More Practice • PCl 5 • P 4 O 6 • SF 6 • SO 3 • SO 2

Section 4. 1 Naming Binary Compounds AND MORE PRACTICE… • N 2 O 3 • Si. O 2 • O 2 F 2 • Xe. F 6

Section 4. 1 Naming Binary Compounds How about the other way? • sulfur hexafluoride • dinitrogen trioxide

Section 4. 1 Naming Binary Compounds A. Naming Compounds That Contain a Metal and a Nonmetal (Ionic Compounds) • Binary ionic compounds contain positive cations and negative anions. – Type I compounds Metal forms only one cation

Section 4. 1 Naming Binary Compounds A. Naming Compounds That Contain a Metal and a Nonmetal Ionic compounds

Section 4. 1 Naming Binary Compounds A. Naming Compounds That Contain a Metal and a Nonmetal

Section 4. 1 Naming Binary Compounds A. Naming Compounds That Contain a Metal and a Nonmetal • For compounds containing both a metal and a nonmetal, the metal is always named first. The nonmetal is named from the root element name.

Section 4. 1 Naming Binary Compounds Naming Ionic Compounds Practice (500 Points Each) • • • KI Ca. S Cs. Br Mg. O Cs. F Al. Cl 3 Mg. I 2 Rb 2 O Sr. I 2 K 2 S

Section 4. 1 Naming Binary Compounds Why don’t we use the Greek Prefixes? • • Charge matters… Na charge? I charge? Only one balanced combination possible, so there is no need for the Greek prefixes… • Ca charge? • O charge? • Ca. O is the only balanced possibility…
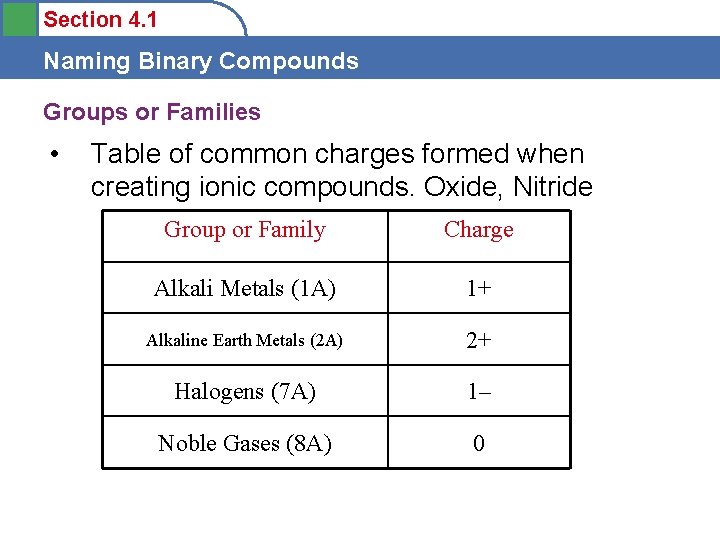
Section 4. 1 Naming Binary Compounds Groups or Families • Table of common charges formed when creating ionic compounds. Oxide, Nitride Group or Family Charge Alkali Metals (1 A) 1+ Alkaline Earth Metals (2 A) 2+ Halogens (7 A) 1– Noble Gases (8 A) 0

Section 4. 1 Naming Binary Compounds Binary Ionic Compounds (Type I) Charge Matters! • Examples: Gangsta Switch! Potassium chloride Magnesium bromide Calcium iodide

Section 4. 1 Naming Binary Compounds • • • How about writing a formula from a name? CHARGE!! (10, 000 points each) Sodium Chloride Magnesium Sulfide Rubidium Bromide Calcium Oxide Barium Iodide Aluminum Fluoride Lithium Oxide Strontium Chloride Potassium Sulfide

Section 4. 1 Naming Binary Compounds A. What if the cation has more than 1 possible charge? Type II Binary Ionic compounds • Write the formula for Gold Chloride • Since the metal ion can have more than one charge, a Roman numeral is used to specify the charge. • Gold Chloride? Gold can have (+1) or (+3) charge • Au. Cl or Au. Cl 3? • Gold (I) Chloride or Gold (III) Chloride • Silver walks into a schoolyard….

Section 4. 1 Naming Binary Compounds A. Naming Compounds That Contain a Metal and a Nonmetal Type II Binary Ionic compounds (Iron II’s in a circle? ) Iron in the wind?

Section 4. 1 Naming Binary Compounds IONIC Roman Numeral (ONLY if more than 1 possible charge) • Molybdenum (VI) Bromide • Palladium (IV) Oxide

Section 4. 1 Naming Binary Compounds IONIC Roman Numeral (ONLY if more than 1 possible charge) • • • Fe. Cl 2 Fe. Cl 3 Pb. O Cu. Cl Pb. O 2 Hg. O Mn. O 2 Pb. Cl 4 Fe O

Section 4. 1 Naming Binary Compounds Bonus Round 1, 000, 000 points • Name the Compound Formed when Barium, Cobalt, and Nitrogen Combine… • Iron, Lithium, Neon….
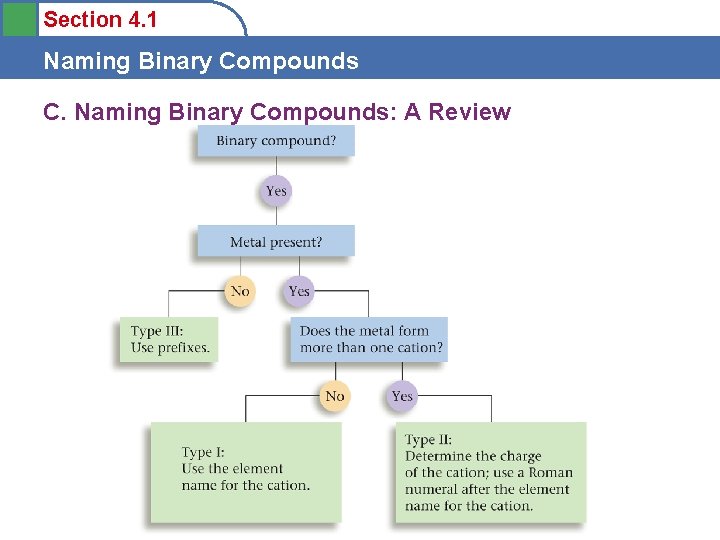
Section 4. 1 Naming Binary Compounds C. Naming Binary Compounds: A Review

Section 4. 1 Naming Binary Compounds Objectives Review 1. To learn to name binary compounds containing only nonmetals (Greek Prefixes) 2. To learn to name Ionic compounds of a metal and nonmetal by balancing charges 3. To learn to name Ionic compounds whose cation has more than 1 possible charge (Roman Numeral Notation) 4. Work Session: Page 118 -122 Greeks: 10, 26 Ionic: 6, 48 skip e, f Roman Numeral: 8

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Objectives 1. To learn the names of common polyatomic ions 2. To learn to name compounds containing polyatomic ions 3. To learn to write the formula for a compound, given its name 4. To get an update on Chuck Norris’ career plans

Section 4. 2 Naming and Writing Formulas for More Complex Compounds A. Naming Compounds That Contain Polyatomic Ions • Polyatomic ions are charged entities composed of several atoms bound together. Page ? ? Digital Version of Table 4. 4

Section 4. 2 Naming and Writing Formulas for More Complex Compounds

Section 4. 2 Naming and Writing Formulas for More Complex Compounds A. Naming Compounds That Contain Polyatomic Ions • Naming ionic compounds containing polyatomic ions follows rules similar to those for binary compounds. – ammonium acetate

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Polyatomic Ions Class Practice (Use Table 4. 4) • • Na 2(SO 4) or Na 2 SO 4 KH 2 PO 4 (NH 4)2 SO 4 Li 2 SO 3 Fe(NO 3)3 Mn(OH)2 Na 2 CO 3

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Polyatomic Ions Independent Practice (Use Table 4. 4) • • Ca(OH)2 Na 3 PO 4 KMn. O 4 (NH 4)2 Cr 2 O 7 Co(Cl. O 4)2 KCl. O 3 Cu(NO 2)2

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Formation of Ionic Compounds

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Polyatomic Ions Class Practice (Use Table 4. 4) • • • Potassium hydroxide Sodium carbonate Cobalt (III) nitrate Calcium sulfite Lead (IV) acetate Ammonium perchlorate

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Mixed Independent Practice (Use Table 4. 4) • • • ammonium sulfate Vanadium (V) fluoride disulfur dichloride Rubidium peroxide Aluminum oxide

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Mixed Independent Practice (Use Table 4. 4 and Periodic Table) • • • Na. HCO 3 Na. Br Zn 3(PO 4)2 Ba. SO 4 Br. F 5 Cu. SO 4
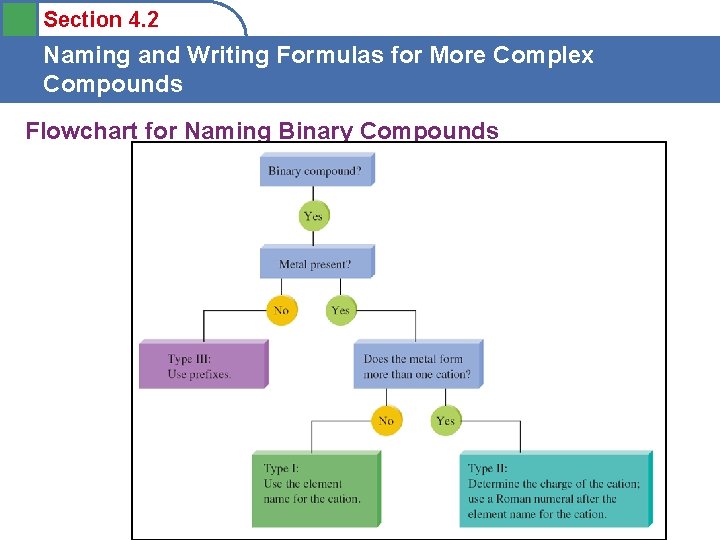
Section 4. 2 Naming and Writing Formulas for More Complex Compounds Flowchart for Naming Binary Compounds
Section 4. 2 Naming and Writing Formulas for More Complex Compounds Objectives Review 1. To learn the names of common polyatomic ions 2. To learn to name compounds containing polyatomic ions 3. To learn to write the formula for a compound, given its name 4. Chuck Norris was considering a career in History… 5. Work Session: Page 118 -122 Polyatomic: 17, 19, 21, 27, Mixed: 50 Digital Version of Table 4. 4

Section 4. 2 Naming and Writing Formulas for More Complex Compounds Objectives 1. This is the end of the required lessons for Chapter 4.

Section 4. 2 Naming and Writing Formulas for More Complex Compounds B. Naming Acids

Section 4. 2 Naming and Writing Formulas for More Complex Compounds B. Naming Acids • An acid is a molecule with one or more H+ ions attached to an anion.

Section 4. 2 Naming and Writing Formulas for More Complex Compounds B. Naming Acids

Section 4. 2 Naming and Writing Formulas for More Complex Compounds B. Naming Acids

Section 4. 2 Naming and Writing Formulas for More Complex Compounds B. Naming Acids

Section 4. 2 Naming and Writing Formulas for More Complex Compounds C. Writing Formulas from Names • Sodium hydroxide – Na. OH • Potassium carbonate – K 2 CO 3 • Sulfuric acid – H 2 SO 4 • Dinitrogen pentoxide – N 2 O 5 • Cobalt(III) nitrate – Co(NO 3)3
- Slides: 43