Section 20 1 Saturated Hydrocarbons Three Objectives SWBAT

Section 20. 1 Saturated Hydrocarbons Three Objectives: SWBAT 1. To understand the types of bonds formed by the carbon atom 2. To learn about the alkanes 3. To learn about some common polymers
Section 20. 1 Saturated Hydrocarbons Carbon Chemistry Carbon is an element that 1. Can bond strongly to itself (C—C) 2. Can form long chains or rings (C—C—C—C…) 3. Can bond to other nonmetals: H, N, O, S, and halogens. 4. Has extraordinary number of carbon compounds existing in nature. Biomolecule – molecule that functions in maintaining and reproducing life Organic compounds – vast majority of carbon compounds Exceptions: oxides of carbon and carbonates are inorganic compounds.
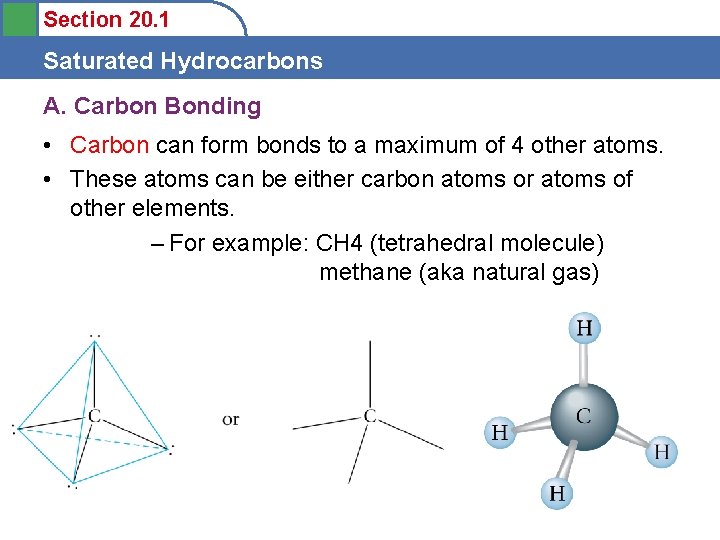
Section 20. 1 Saturated Hydrocarbons A. Carbon Bonding • Carbon can form bonds to a maximum of 4 other atoms. • These atoms can be either carbon atoms or atoms of other elements. – For example: CH 4 (tetrahedral molecule) methane (aka natural gas)
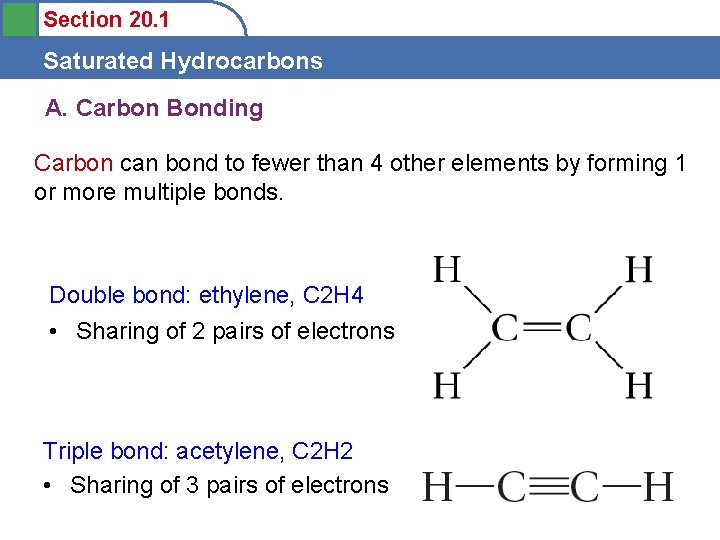
Section 20. 1 Saturated Hydrocarbons A. Carbon Bonding Carbon can bond to fewer than 4 other elements by forming 1 or more multiple bonds. Double bond: ethylene, C 2 H 4 • Sharing of 2 pairs of electrons Triple bond: acetylene, C 2 H 2 • Sharing of 3 pairs of electrons
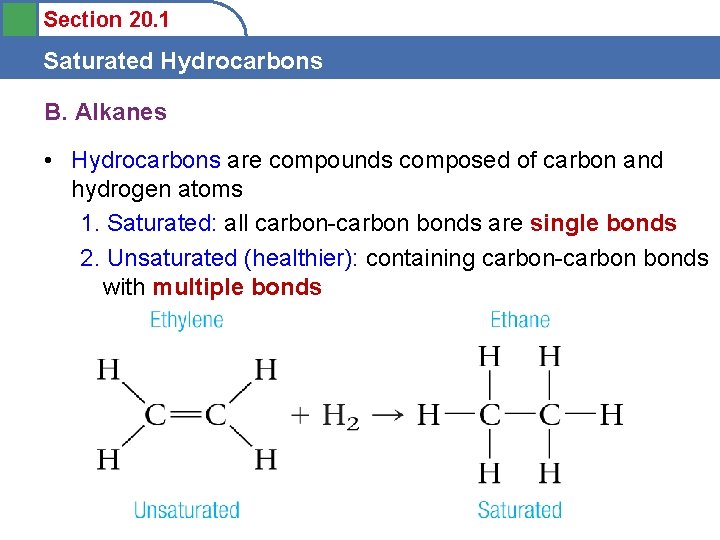
Section 20. 1 Saturated Hydrocarbons B. Alkanes • Hydrocarbons are compounds composed of carbon and hydrogen atoms 1. Saturated: all carbon-carbon bonds are single bonds 2. Unsaturated (healthier): containing carbon-carbon bonds with multiple bonds
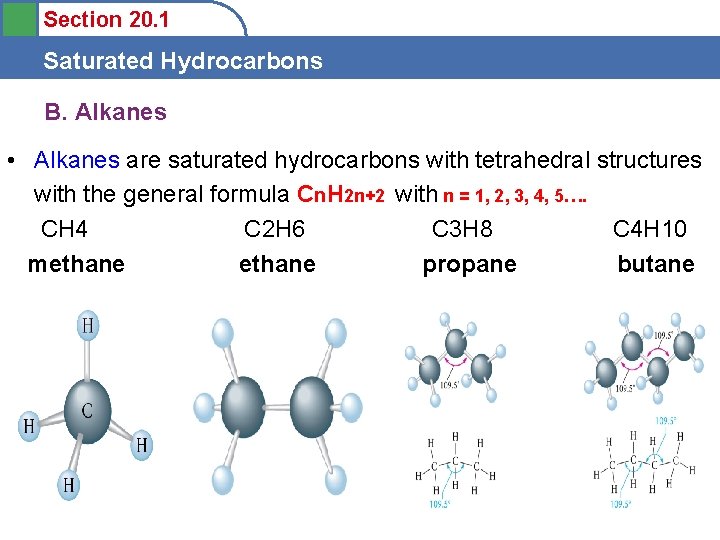
Section 20. 1 Saturated Hydrocarbons B. Alkanes • Alkanes are saturated hydrocarbons with tetrahedral structures with the general formula Cn. H 2 n+2 with n = 1, 2, 3, 4, 5…. CH 4 C 2 H 6 C 3 H 8 C 4 H 10 methane propane butane
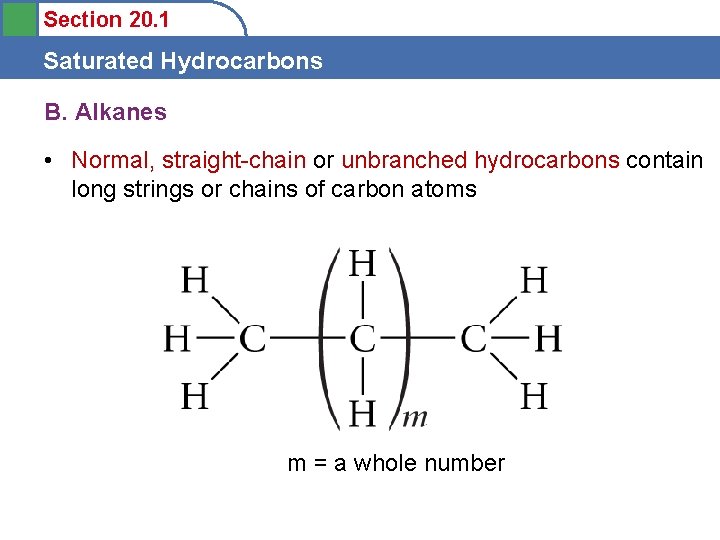
Section 20. 1 Saturated Hydrocarbons B. Alkanes • Normal, straight-chain or unbranched hydrocarbons contain long strings or chains of carbon atoms m = a whole number

Section 20. 1 Saturated Hydrocarbons Example 1: Normal Hydrocarbons 1. Alkanes with 6 carbon atoms CH 3—(CH 2)4—CH 3 CH 3 CH 2 CH 2 CH 3 m=4 2. Alkanes with 8 carbon atoms CH 3—(CH 2)6—CH 3 m=6 CH 3 CH 2 CH 2 CH 3

Section 20. 1 Saturated Hydrocarbons B. Alkanes

Section 20. 1 Saturated Hydrocarbons Write the chain structure and formula for… An alkane with 6 carbon atoms C 6 H 14 CH 3 – (CH 4)4 – CH 3 An alkane with 8 carbon atoms C 8 H 18 CH 3 – (CH 4)6 – CH 3

Section 20. 1 Saturated Hydrocarbons Homework • Read Sec. 1 p. 700 -704 only • Read Sec 4. p. 738 -741 only

Section 20. 1 Chapter 21 Saturated Hydrocarbons Objectives: SWBAT 1. To learn about some common polymers
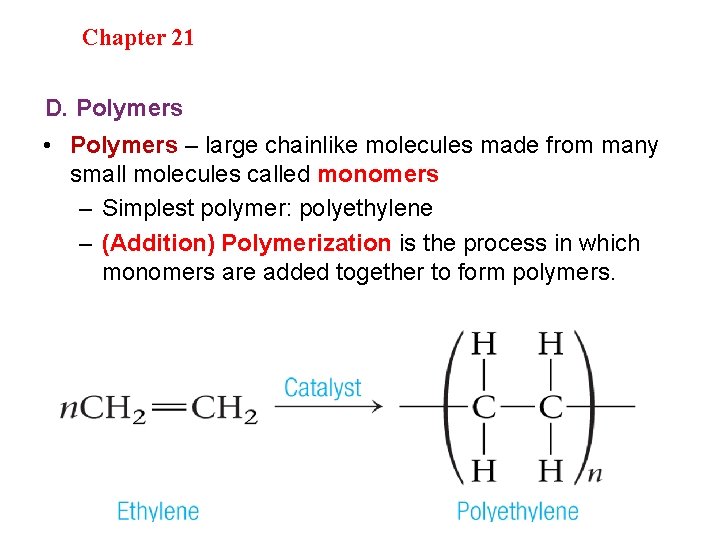
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons D. Polymers • Polymers – large chainlike molecules made from many small molecules called monomers – Simplest polymer: polyethylene – (Addition) Polymerization is the process in which monomers are added together to form polymers.
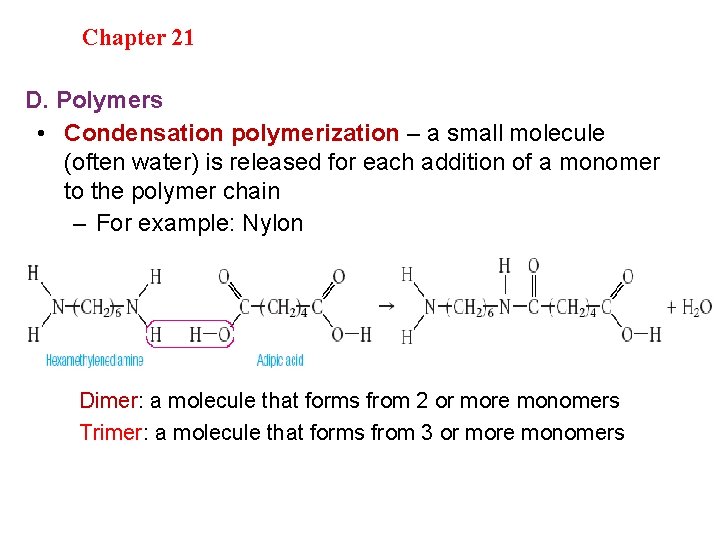
Section 20. 1 Chapter 21 Saturated Hydrocarbons D. Polymers • Condensation polymerization – a small molecule (often water) is released for each addition of a monomer to the polymer chain – For example: Nylon Dimer: a molecule that forms from 2 or more monomers Trimer: a molecule that forms from 3 or more monomers
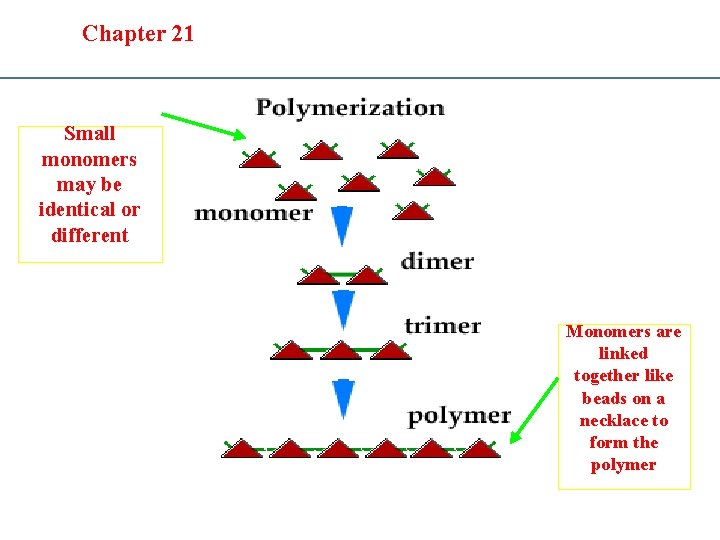
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Small monomers may be identical or different Monomers are linked together like beads on a necklace to form the polymer

Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons • 4 Types of Biomolecules 1. Lipids 2. Carbohydrates 3. Proteins 4. Nucleic Acids

Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Lipids • Examples: Fats, oils, waxes & steroids • Monomer: 3 fatty-acids and 1 glycerol • Polymer: Lipids – Unsaturated Fats: Liquid at room temperature due to double bonds (bent structure) – Saturated Fats: Solid at room temperature and no double bonds (full of hydrogen)
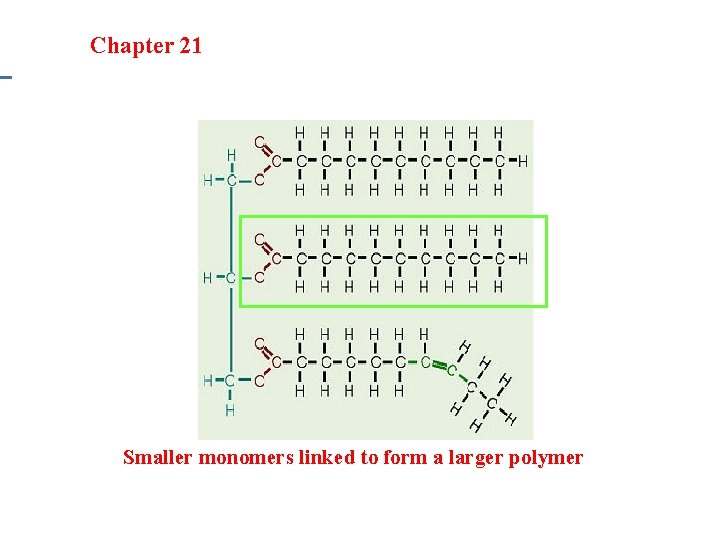
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Smaller monomers linked to form a larger polymer

Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Carbohydrates • Examples: Sugars & Starches • Monomer: Monosaccharides – Simple sugars like glucose, fructose, and galactose • Polymer: Polysaccharides (starches) – Cellulose: Plant starch – Glycogen: Animal Starch
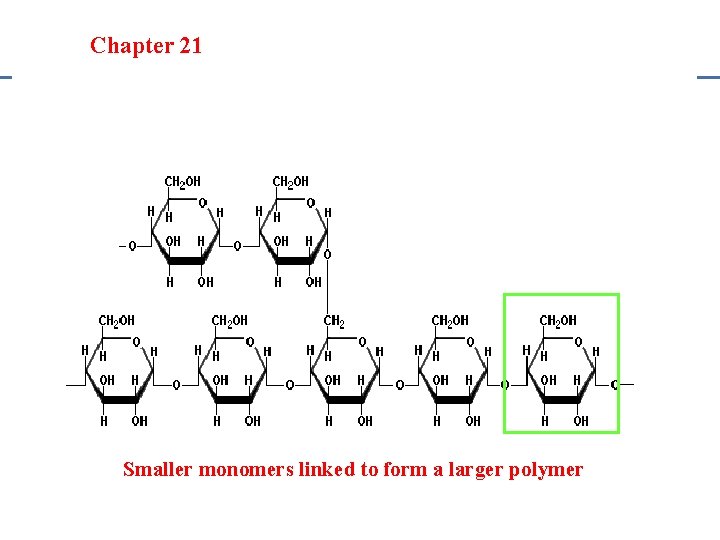
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Smaller monomers linked to form a larger polymer

Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Proteins • • Examples: Hair, nails and enzymes Monomer: Amino Acids Polymer: Protein Structure: Long chains of amino acids
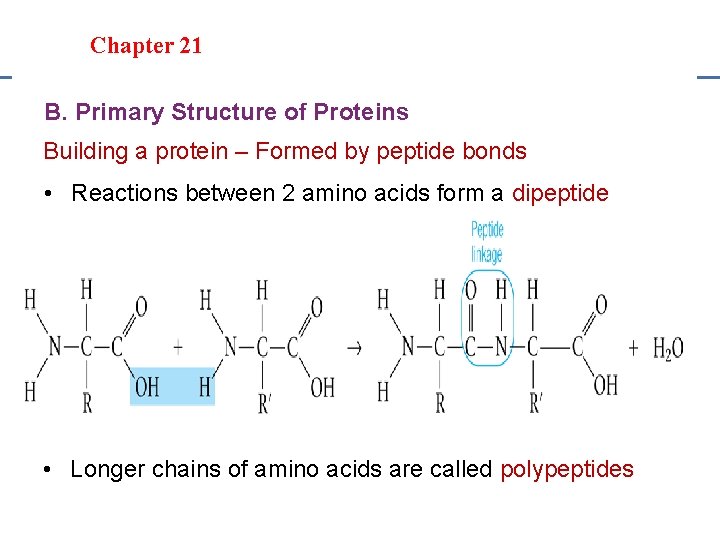
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons B. Primary Structure of Proteins Building a protein – Formed by peptide bonds • Reactions between 2 amino acids form a dipeptide • Longer chains of amino acids are called polypeptides
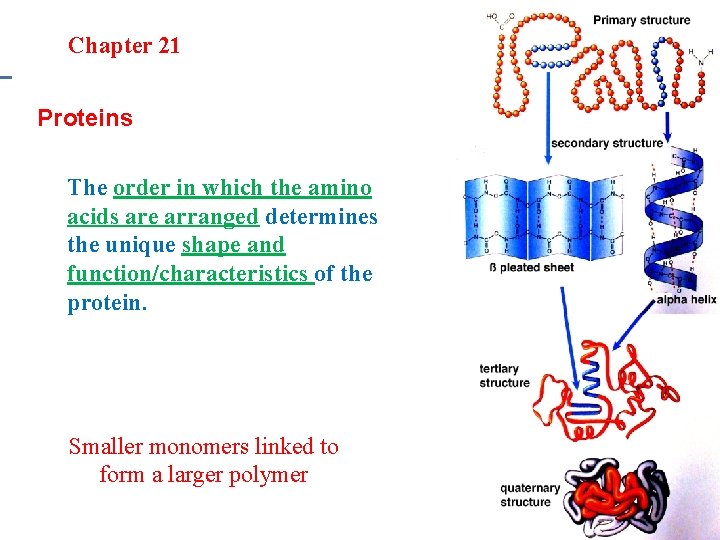
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Proteins The order in which the amino acids are arranged determines the unique shape and function/characteristics of the protein. Smaller monomers linked to form a larger polymer

Section 20. 1 Chapter 21 Saturated Hydrocarbons Nucleic Acids • • Examples: DNA & RNA Monomer: Nucleotide (sugar, phosphate & nitrogen base) Polymer: Nucleic Acid Structure: Long chains of nucleotides found in a twisted or folded structure
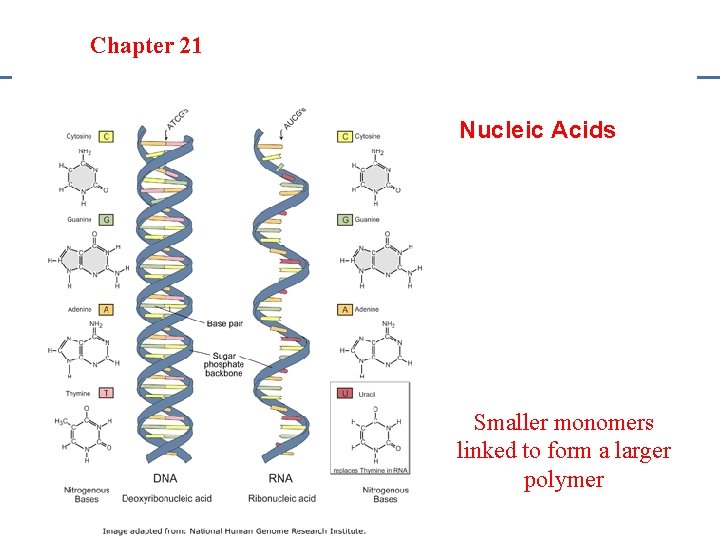
Section 20. 1 Section 20 -4 Chapter 21 Saturated Hydrocarbons Nucleic Acids Smaller monomers linked to form a larger polymer

Section 20. 1 Chapter 21 Saturated Hydrocarbons Homework • Read p. 752 -760 only • Read p. 762 -775 only
- Slides: 26