Section 2 Ionic and Covalent Bonds CHAPTER 4

Section 2: Ionic and Covalent Bonds CHAPTER 4: THE STRUCTURE OF MATTER

Goals/Objectives After completing the lesson, students will be able to: Explain why atoms sometimes join to form bonds; Explain why some atoms transfer their valence electrons to form ionic bonds, while other atoms share valence electrons to form covalent bonds; Differentiate between ionic, covalent, and metallic bonds; Compare the properties of substances with different types of bonds.

The Outermost Energy Level of a Bonded Atom is Full of Electrons When atoms join together, a bond will form. Atoms bond when their valence electrons interact. Atoms join to form bonds so that each atom has a full outermost energy level. When atoms bond, the positive nucleus of one atom attracts the negative electron of the other atom, and vice versa. This pulls the atoms closer, so that their electron clouds cross each other. Essentially, the atoms are receptive of each other as the electrons are filling their energy level.

Bond Can Bend and Stretch Without Breaking Although some bonds are stronger and more rigid than others, all bonds behave more like flexible springs than sticks. The atoms move back and forth a little and their nuclei do not always stay the same distance apart. Although bonds are not rigid, they still hold atoms together tightly.
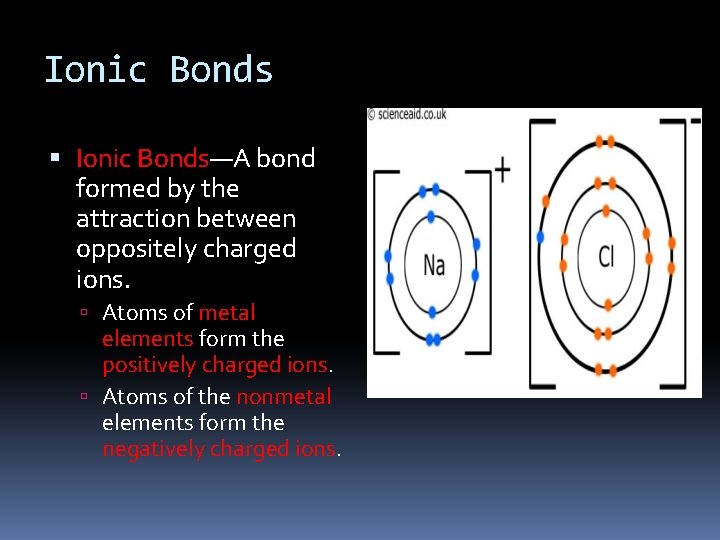
Ionic Bonds Ionic Bonds—A bond formed by the attraction between oppositely charged ions. Atoms of metal elements form the positively charged ions. Atoms of the nonmetal elements form the negatively charged ions.

Ionic Bonds are Formed by the Transfer of Electrons Some atoms do not share electrons to fill their energy level— instead, they transfer electrons. One atom gains the electron that the other atom gave up. Both ions that form usually have filled energy levels The result is a positive ion and a negative ion. The one that “loses” the electron is positive, whereas the one that “gains” the electron is negative. These oppositely charged ions attract each other and form an ionic bond.

Ionic Compounds are in the Form of Networks, Not Molecules Read this section on page 117.

When Melted or Dissolved in Water, Ionic Compounds Conduct Electricity Solid ionic compounds do not conduct electricity, because the charged ions are locked into place, causing the melting points of ionic compounds to be very high. If you dissolve an ionic compound in water or melt it, it can conduct electricity. This is because the ions are free to move.

Metallic Bond Metals can conduct electricity when they are solid. Metal are also flexible, so they can bend and stretch without breaking. Metals are flexible due to the atoms ability to slide past each other without their bonds breaking. Metallic Bond—A bond formed by the attraction between positively charged metal ions and the electrons around them. The attraction between one atom’s nucleus and a neighboring atom’s electrons packs the atom closely together. The close packing causes the outermost energy levels of the atom to overlap. This allows electrons to move from atom to atom freely.

Covalent Bonds Covalent Bond—A bond formed when atoms share one or more pairs of electrons. Covalent bonds are often formed between nonmetal atoms Covalent compounds can be solids, liquids, or gases. Most covalent compounds have a low melting point—usually below 300 degrees Celsius.

Sharing Electrons Atoms Joined by Covalent Bonds Share Electrons Atoms May Share More Than One Pair of Electrons The way electrons are shared depends on which atoms are sharing the electrons. When looking at bonding models, double lines represents two pairs of electrons are shared (double covalent bond), and three lines represents three pairs of electrons shared (triple covalent bond. ) The triple bond will be a stronger bond due to the need of more energy to break it.

Atoms Do Not Always Share Electrons Equally When two different atoms share electrons, the electrons are not shared equally. The shared electrons are attracted to the nucleus of one atom more than the other This is referred to as a polar covalent bond. Usually, electrons are more attracted to atoms of elements that are located farther to the right and closer to the top of the periodic table.

Polyatomic Ions Polyatomic Ion—An ion made of two or more atoms that are covalently bonded and that act like a single ion. Polyatomic ions are groups of covalently bonded atoms that have either lost or gained electrons.
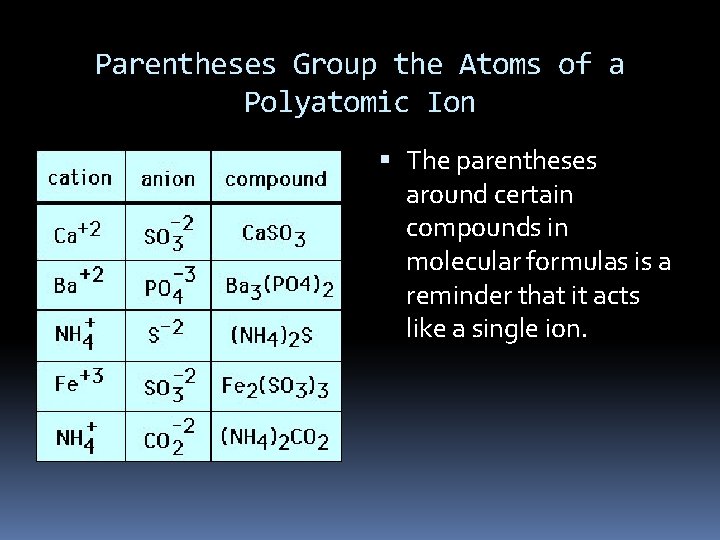
Parentheses Group the Atoms of a Polyatomic Ion The parentheses around certain compounds in molecular formulas is a reminder that it acts like a single ion.

Some Polyatomic Anion Names Relate to Their Oxygen Content Read this section on page 121. An –ate ending is used to name the ion with one more oxygen atom An –ite ending is used to name the ion with on less oxygen

Summary (1) Atoms bond when their valence electrons interact; Cations and anions attract each other to form ionic bonds; When ionic compounds are melted or dissolved in water, moving ions can conduct electricity; Atoms in metals are joined by metallic bonds;

Summary (2) Metals conduct electricity because electrons can move from atom to atom; Covalent bonds form when atoms share electron pairs. Electrons may be shared equally or unequally Polyatomic ions are covalently bonded atoms that have either lost or gained electrons. Their behavior resembles that of simple ions
- Slides: 17