Section 2 3 Carbon Compounds Organic Chemistry Organic
Section 2. 3 Carbon Compounds
Organic Chemistry • Organic Chemistry- Study of all compounds that contain bonds between carbon atoms

I. Chemistry of Carbon • Characteristics of Carbon: 1. Carbon atoms have 4 valence electrons 2. Each electron can join with an electron from another atom to form a strong covalent bond 3. Carbon can bond with many elements such as • • • Hydrogen Oxygen Phosphorus Sulfur Nitrogen SPONCH

Characteristics of Carbon 4. Carbon atoms can bond to other carbon atoms forming chains that are almost unlimited in length (macromolecules) 5. Carbon-carbon bonds can be • Single (C-C) • Double (C=C) • Triple (C C)
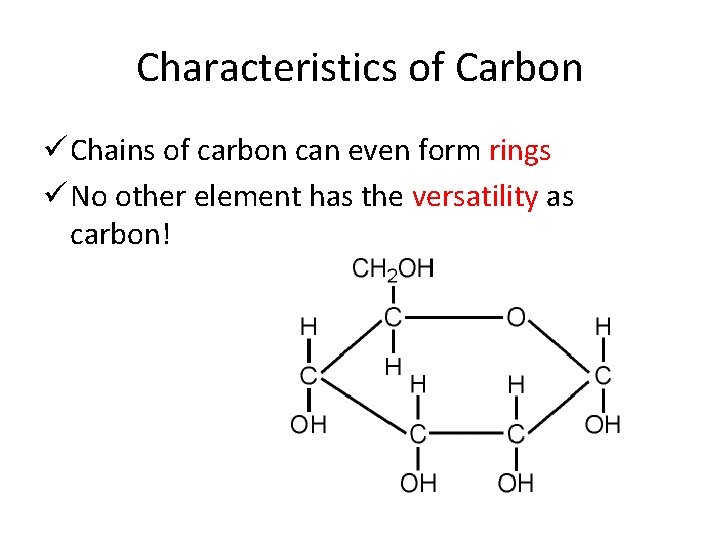
Characteristics of Carbon ü Chains of carbon can even form rings ü No other element has the versatility as carbon!

Macromolecules- large molecules made from 1000 s or 100 s of 1000 s smaller molecules • Made by a process of polymerization • Made of smaller units called monomers joined together to form polymers (the monomers can be identical or different!)

Organic compounds or biomolecules are classified into 4 groups: 1. Carbohydrates 2. Lipids 3. Nucleic Acids 4. Proteins

1. Carbohydrates ü Living things use carbohydrates as their main source of quick energy and structural purposes ü The breakdown of sugars, such as glucose (C 6 H 12 O 6) supplies immediate energy for all cell activity

1. Carbohydrates ü Complex carbohydrates known as starches a) Monosaccharide- single sugar molecules such as glucose, galactose (milk), and fructose (fruits) b) Disaccharide- two monosaccharides linked together
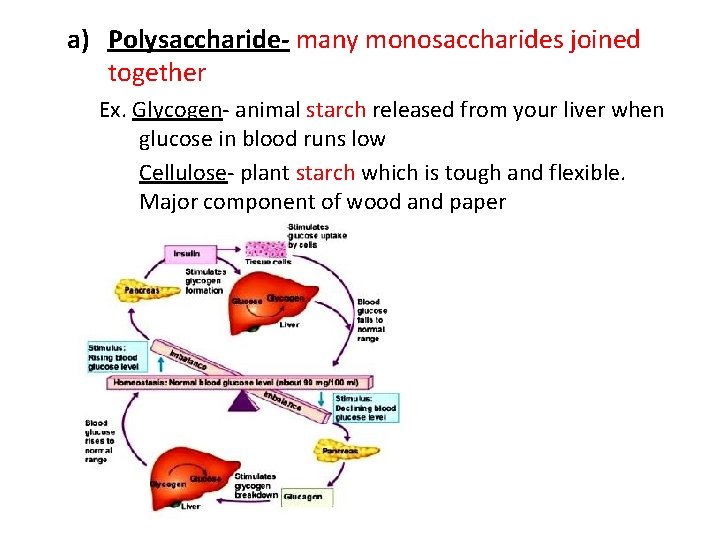
a) Polysaccharide- many monosaccharides joined together Ex. Glycogen- animal starch released from your liver when glucose in blood runs low Cellulose- plant starch which is tough and flexible. Major component of wood and paper

2. Lipids- fats, oils, and waxes ü Compounds made mostly from carbon and hydrogen ü NOT soluble in water! ü Used to store energy

2. Lipids ü Serves an important role in biological membranes and waterproof coverings ü Many lipids are formed when a glycerol molecule combines with compounds called fatty acids
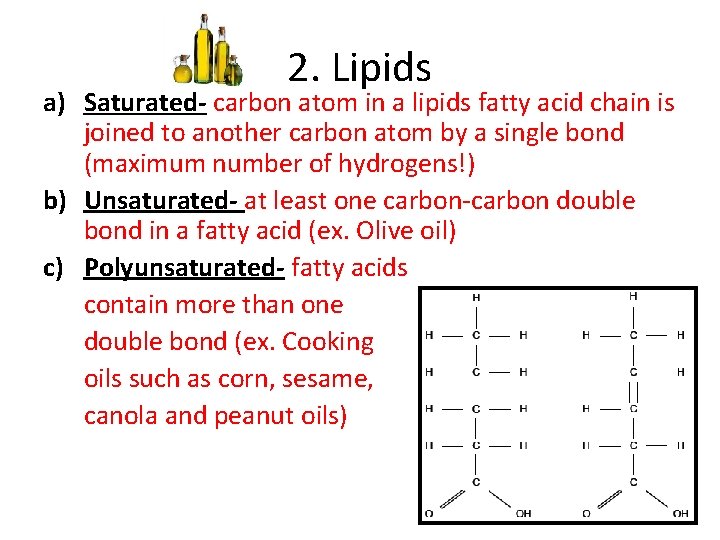
2. Lipids a) Saturated- carbon atom in a lipids fatty acid chain is joined to another carbon atom by a single bond (maximum number of hydrogens!) b) Unsaturated- at least one carbon-carbon double bond in a fatty acid (ex. Olive oil) c) Polyunsaturated- fatty acids contain more than one double bond (ex. Cooking oils such as corn, sesame, canola and peanut oils)

3. Nucleic Acids ü Nucleic Acids- Macromolecules that contain hydrogen, oxygen, nitrogen, carbon and phosphorus a) Nucleotide- individual monomer consisting of a 5 -carbon sugar, a phosphate group, and a nitrogenous base (A, T, C, or G)

3. Nucleic Acids – Individual nucleotides can be joined by covalent bonds to form a nucleic acid – Nucleic acids store and transmit heredity or genetic information 1. DNA- deoxyribonucleic acid (sugar=deoxyribose) 2. RNA- ribonucleic acid (sugar = ribose)
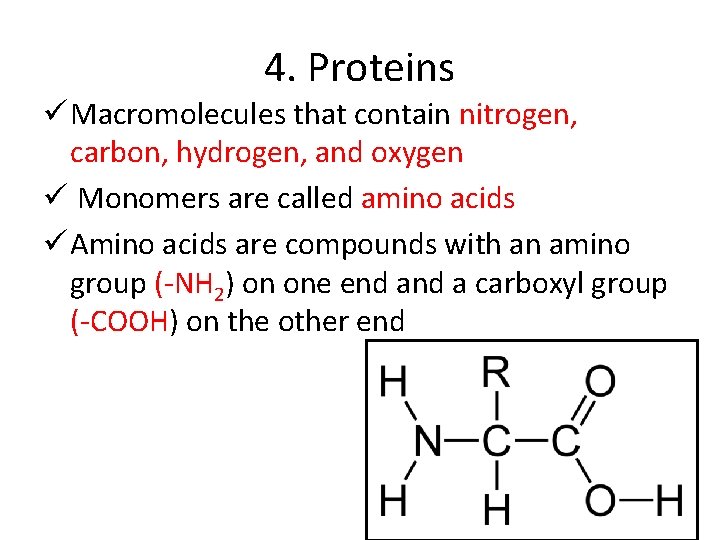
4. Proteins ü Macromolecules that contain nitrogen, carbon, hydrogen, and oxygen ü Monomers are called amino acids ü Amino acids are compounds with an amino group (-NH 2) on one end a carboxyl group (-COOH) on the other end
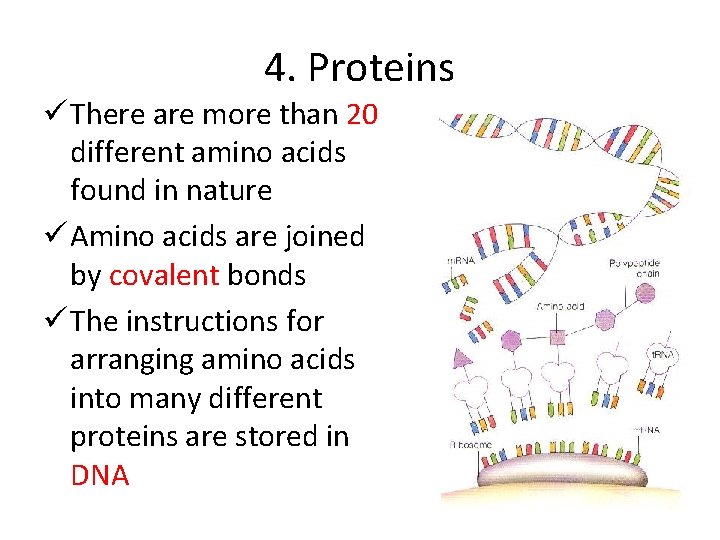
4. Proteins ü There are more than 20 different amino acids found in nature ü Amino acids are joined by covalent bonds ü The instructions for arranging amino acids into many different proteins are stored in DNA
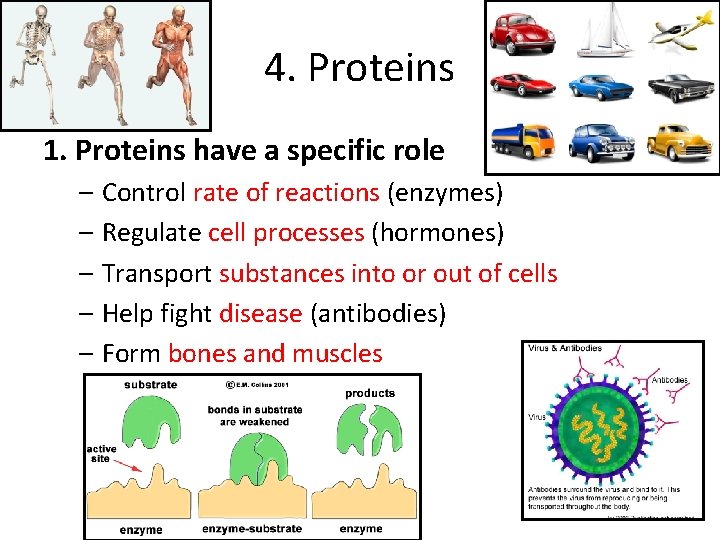
4. Proteins 1. Proteins have a specific role – Control rate of reactions (enzymes) – Regulate cell processes (hormones) – Transport substances into or out of cells – Help fight disease (antibodies) – Form bones and muscles
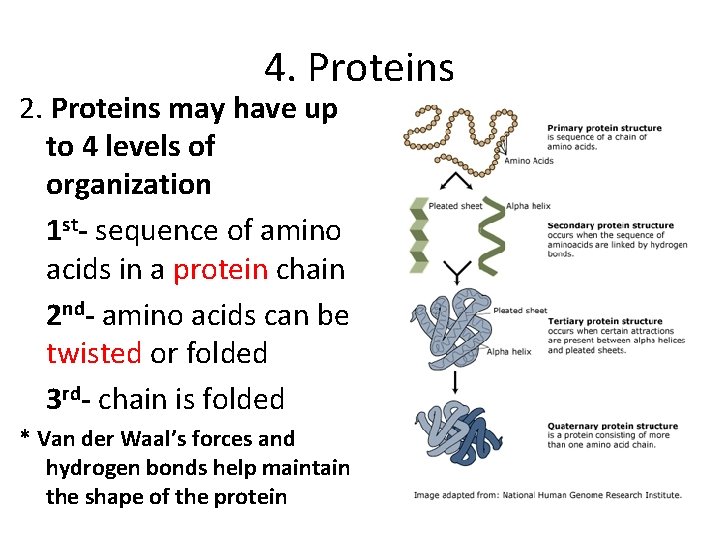
4. Proteins 2. Proteins may have up to 4 levels of organization 1 st- sequence of amino acids in a protein chain 2 nd- amino acids can be twisted or folded 3 rd- chain is folded * Van der Waal’s forces and hydrogen bonds help maintain the shape of the protein

III. Enzymes- biological catalysts that cells use to speed up chemical reactions within a cell ü Enzymes speed up reactions by lowering the activation energy ü Activation Energy- the amount of energy needed to initiate a chemical reaction ü Most enzymes work best at a certain p. H and certain temperatures

Enzymes ü Enzymes play essential roles in i. iii. iv. Regulating chemical pathways Making materials that cells need Releasing energy Transferring information
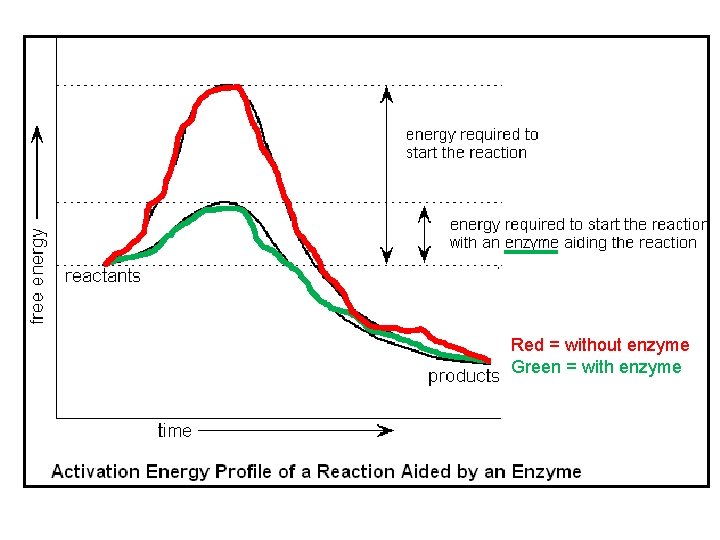
Red = without enzyme Green = with enzyme
- Slides: 22