Section 2 2Naming Chemicals We need to be

Section 2. 2—Naming Chemicals (We need to be able to name the chemicals in the antacids!) Objectives • Explain and practice nomenclature rules in naming chemicals
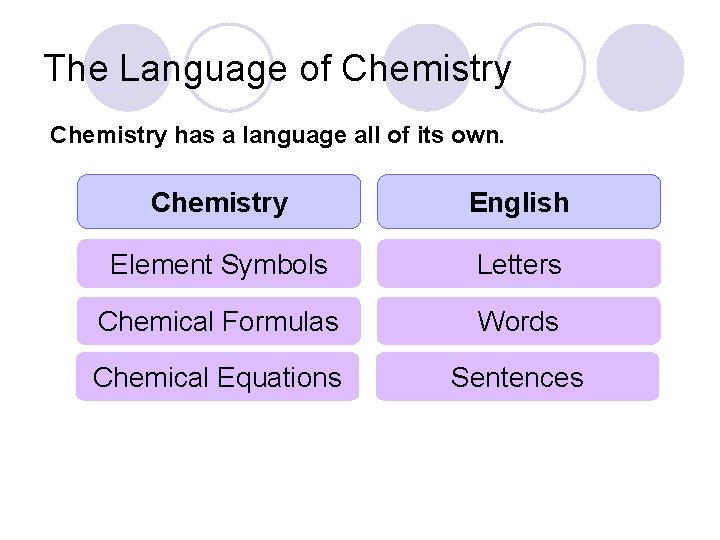
The Language of Chemistry has a language all of its own. Chemistry English Element Symbols Letters Chemical Formulas Words Chemical Equations Sentences

Symbols & Formulas • Each element symbol starts with a capital letter. If the element symbol is more than one letter, ONLY the first one is capitalized. • Na not NA represents sodium. • CO is 2 elements; Co is one. • Subscripts (numbers written below the text line) tell how many atoms of each element are in the compound. • Na 2 SO 4 contains. .
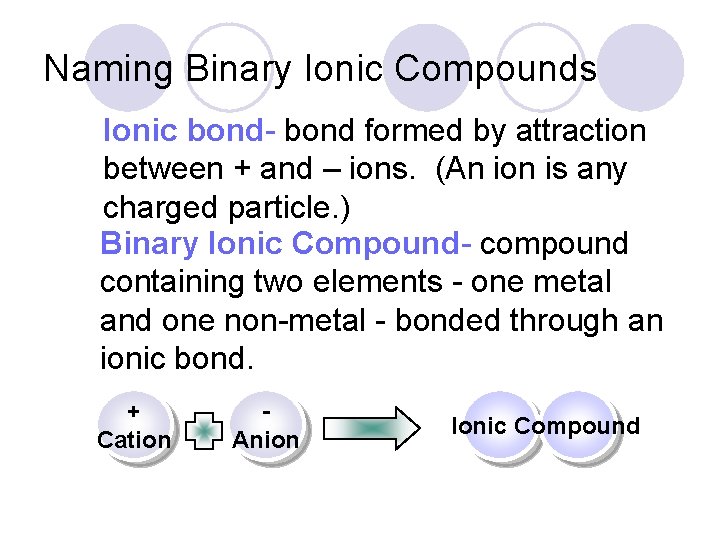
Naming Binary Ionic Compounds Ionic bond- bond formed by attraction between + and – ions. (An ion is any charged particle. ) Binary Ionic Compound- compound containing two elements - one metal and one non-metal - bonded through an ionic bond. + Cation Anion Ionic Compound
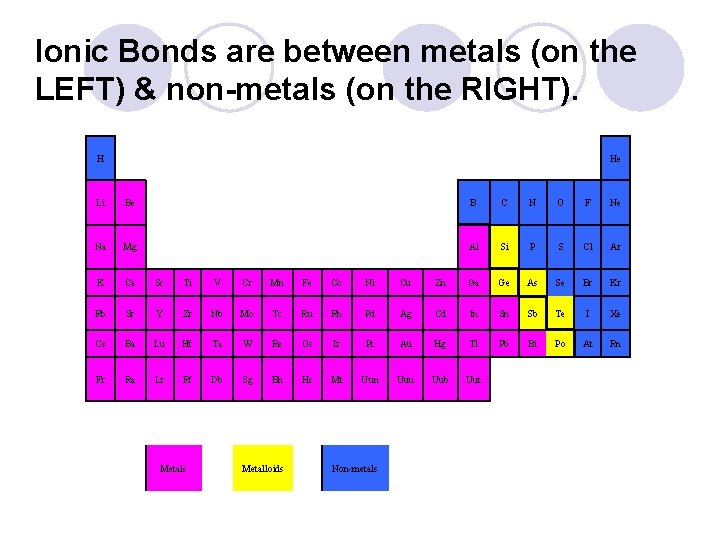
Ionic Bonds are between metals (on the LEFT) & non-metals (on the RIGHT). H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba Lu Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Lr Rf Db Sg Bh Hs Mt Uun Uuu Uub Uut Metals Metalloids Non-metals

Identifying & Naming Binary Ionic l To identify: look for ¡ 2 elements (“binary”) ¡A metal & a non-metal (“ionic”) l To name these compounds: ¡Write the name of the metal (the cation) ¡Write the name of the non-metal (the anion) with the suffix “-ide” ¡The subscripts in the formula do not matter when naming this type of compound.

Example #1 Na. Cl

Example #1 “Sodium” Cation Na. Cl Anion “Chlorine” becomes “Chloride” Sodium Chloride
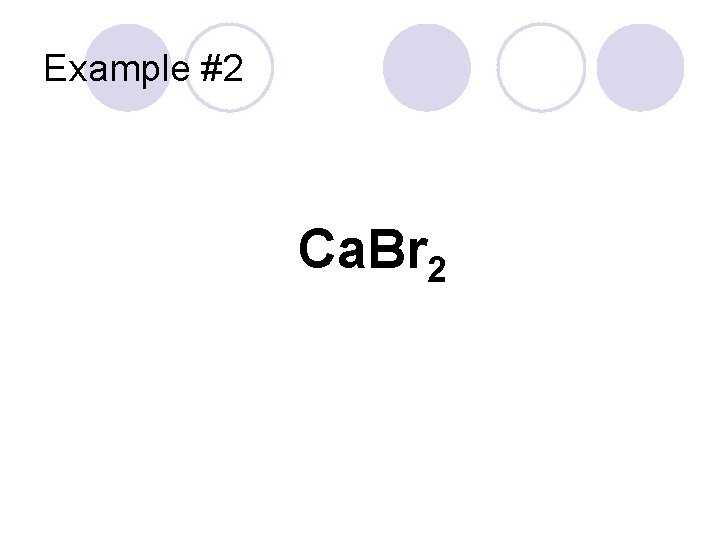
Example #2 Ca. Br 2
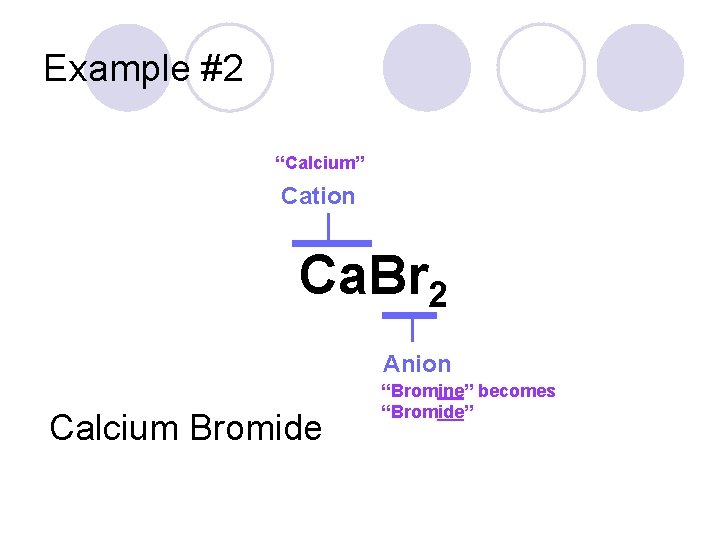
Example #2 “Calcium” Cation Ca. Br 2 Anion Calcium Bromide “Bromine” becomes “Bromide”
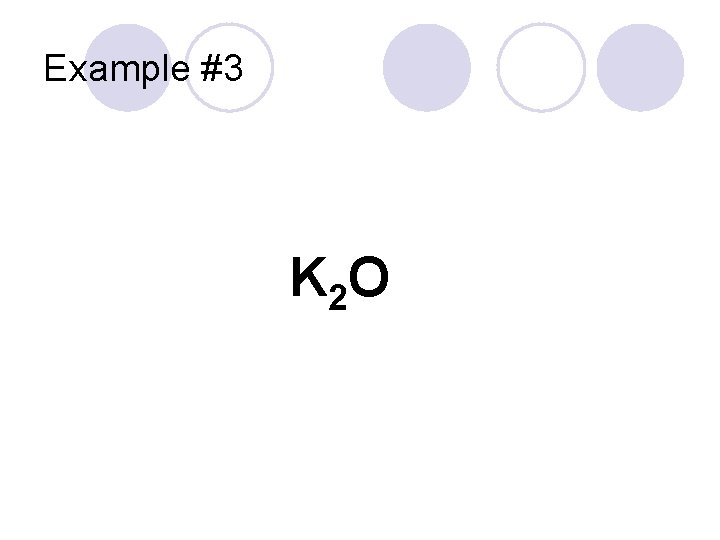
Example #3 K 2 O
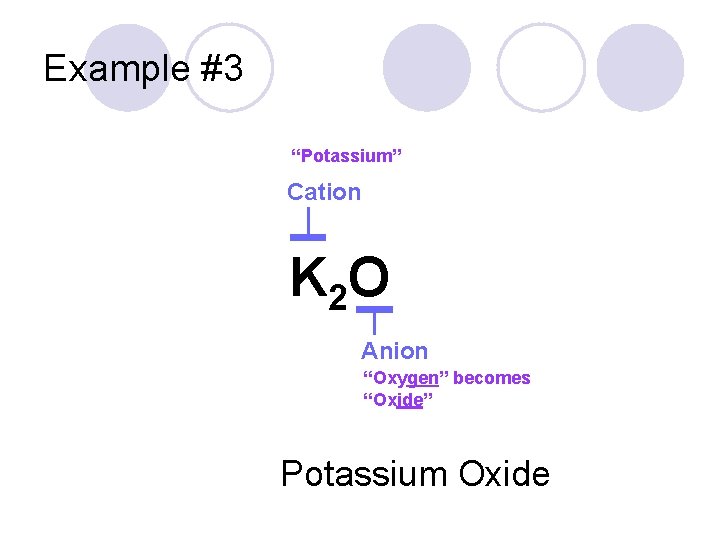
Example #3 “Potassium” Cation K 2 O Anion “Oxygen” becomes “Oxide” Potassium Oxide

Let’s Practice Ca. F 2 Example: Write the name for the following compounds Na 3 P Na. Cl Sr. Br 2

Let’s Practice Example: Write the name for the following compounds Ca. F 2 Calcium fluoride Na 3 P Sodium phosphide Na. Cl Sodium chloride Sr. Br 2 Strontium bromide

Naming Polyatomic Ionic Compounds Polyatomic Ion- a group of atoms (i. e. MORE THAN 1 ATOM) that are bonded together and have an overall charge Polyatomic Ionic Compoundcompound containing at least one polyatomic ion + Cation Polyatomic Anion Polyatomic Ionic Compound
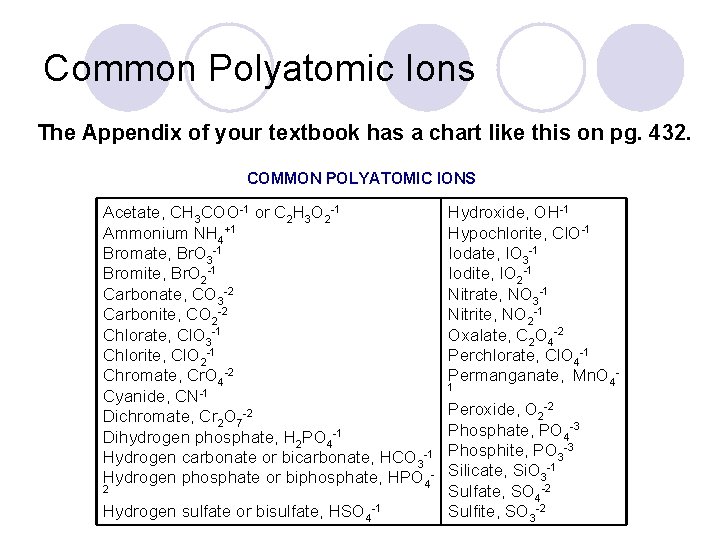
Common Polyatomic Ions The Appendix of your textbook has a chart like this on pg. 432. COMMON POLYATOMIC IONS Acetate, CH 3 COO-1 or C 2 H 3 O 2 -1 Ammonium NH 4+1 Bromate, Br. O 3 -1 Bromite, Br. O 2 -1 Carbonate, CO 3 -2 Carbonite, CO 2 -2 Chlorate, Cl. O 3 -1 Chlorite, Cl. O 2 -1 Chromate, Cr. O 4 -2 Cyanide, CN-1 Dichromate, Cr 2 O 7 -2 Dihydrogen phosphate, H 2 PO 4 -1 Hydrogen carbonate or bicarbonate, HCO 3 -1 Hydrogen phosphate or biphosphate, HPO 42 Hydrogen sulfate or bisulfate, HSO 4 -1 Hydroxide, OH-1 Hypochlorite, Cl. O-1 Iodate, IO 3 -1 Iodite, IO 2 -1 Nitrate, NO 3 -1 Nitrite, NO 2 -1 Oxalate, C 2 O 4 -2 Perchlorate, Cl. O 4 -1 Permanganate, Mn. O 41 Peroxide, O 2 -2 Phosphate, PO 4 -3 Phosphite, PO 3 -3 Silicate, Si. O 3 -1 Sulfate, SO 4 -2 Sulfite, SO 3 -2

Help Identifying Polyatomic Ions l The only cation (front-half) polyatomic ion is “NH 4+” l All other polyatomic ions are anions (backhalf) l The subscript within the polyatomic ion is important (it must ALWAYS match exactly with the on your ion list) l If there are parenthesis, the polyatomic ion is inside (ignore the number outside)
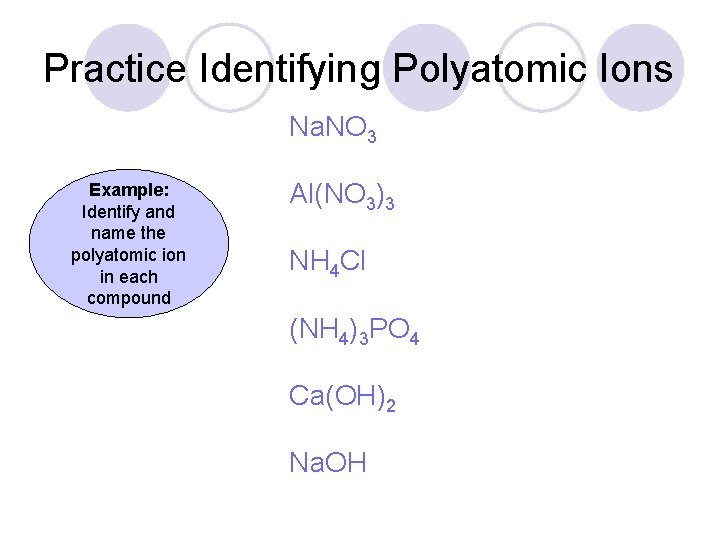
Practice Identifying Polyatomic Ions Na. NO 3 Example: Identify and name the polyatomic ion in each compound Al(NO 3)3 NH 4 Cl (NH 4)3 PO 4 Ca(OH)2 Na. OH
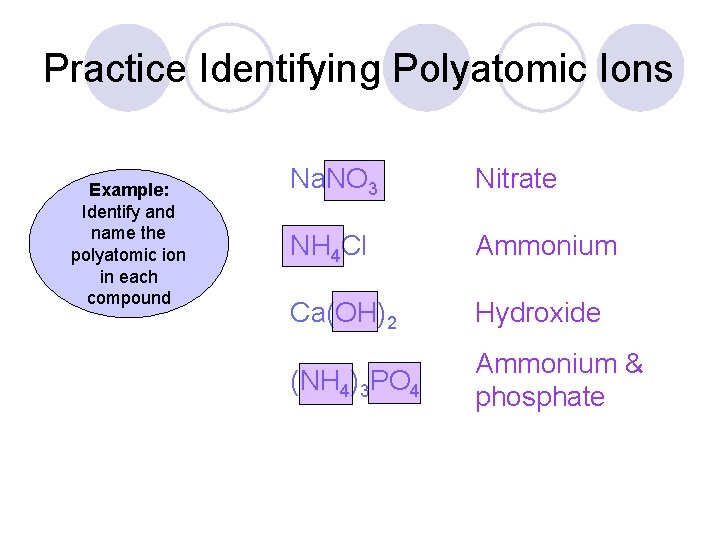
Practice Identifying Polyatomic Ions Example: Identify and name the polyatomic ion in each compound Na. NO 3 Nitrate NH 4 Cl Ammonium Ca(OH)2 Hydroxide (NH 4)3 PO 4 Ammonium & phosphate

Identifying & Naming Polyatomic Ionic l These compounds have: ¡More than 2 capital letters (none starting with H) ¡Usually contain at least 1 metal & 1 non-metal l To name these compounds: ¡Write the name of the cation (the metal element name or “Ammonium” for “NH 4”) ¡If the anion is a polyatomic ion, write the polyatomic ion’s name (from the look-up sheet) ¡If the anion is a single non-metal element, write its name with the suffix “-ide”
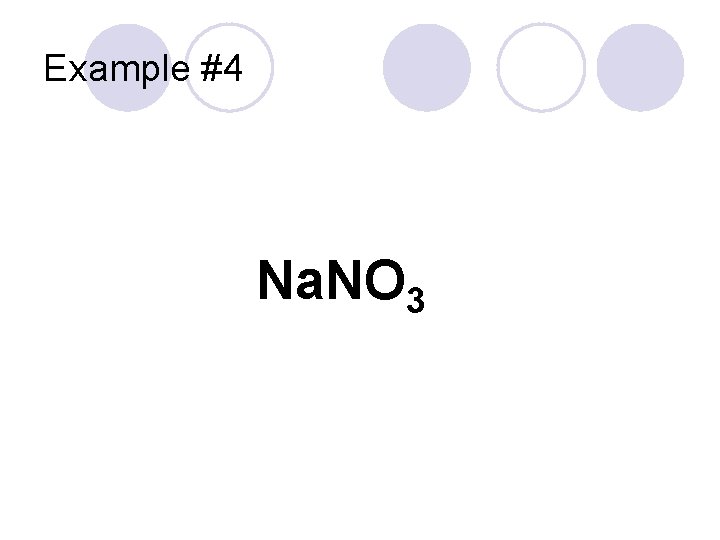
Example #4 Na. NO 3

Example #4 “Sodium” Cation Na. NO 3 Polyatomic Anion “Nitrate” Sodium Nitrate
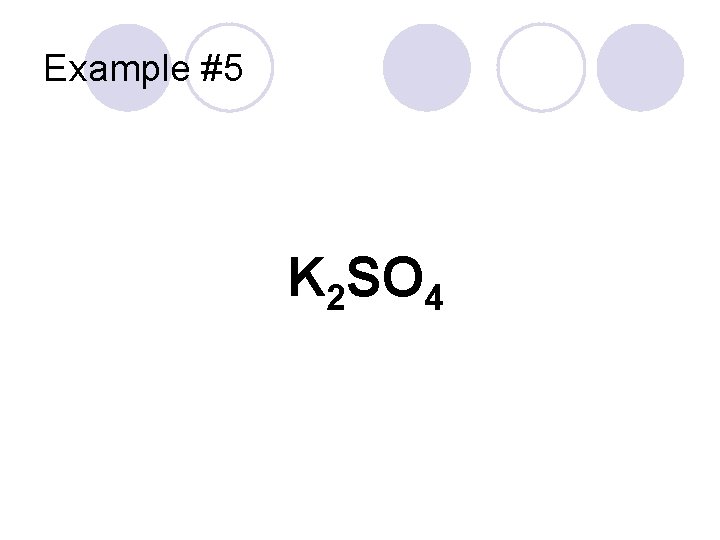
Example #5 K 2 SO 4
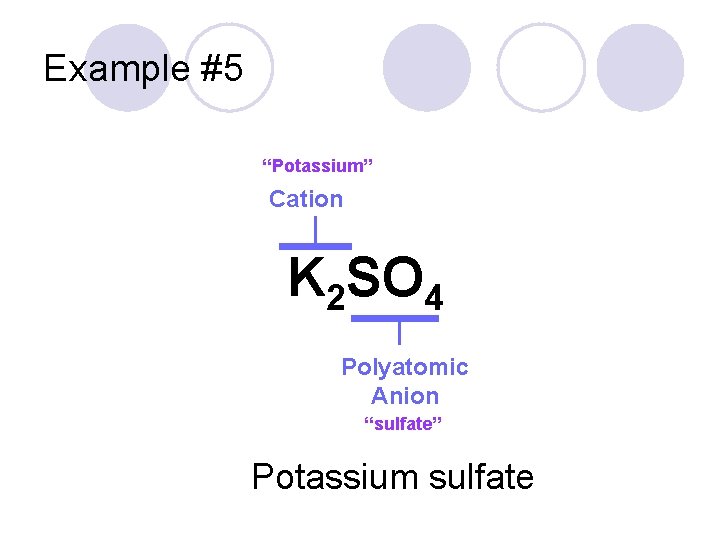
Example #5 “Potassium” Cation K 2 SO 4 Polyatomic Anion “sulfate” Potassium sulfate
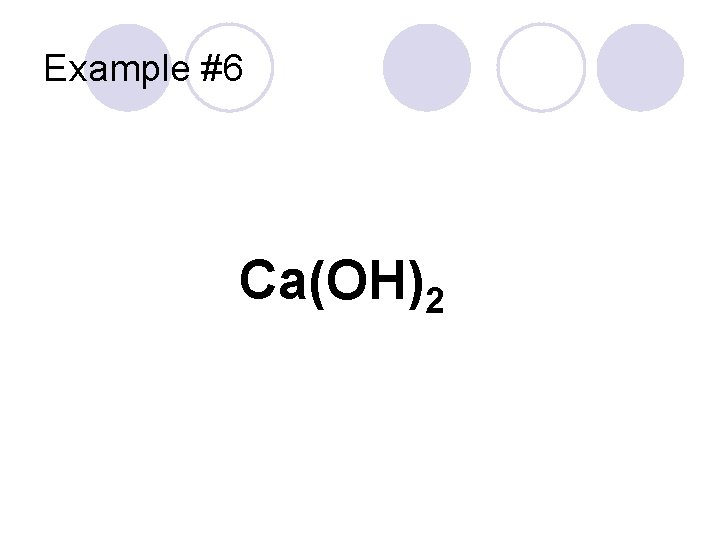
Example #6 Ca(OH)2
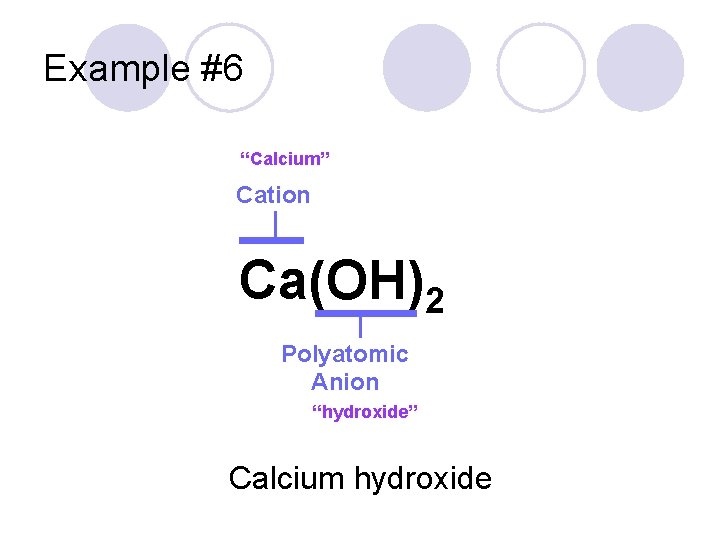
Example #6 “Calcium” Cation Ca(OH)2 Polyatomic Anion “hydroxide” Calcium hydroxide
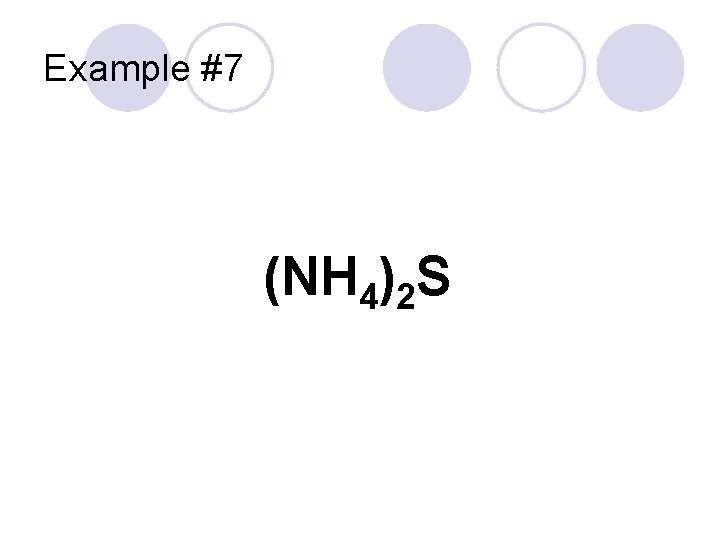
Example #7 (NH 4)2 S
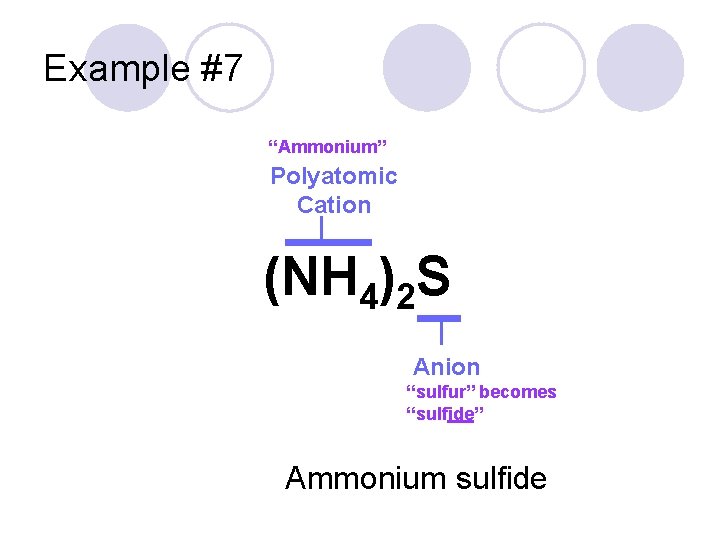
Example #7 “Ammonium” Polyatomic Cation (NH 4)2 S Anion “sulfur” becomes “sulfide” Ammonium sulfide

Let’s Practice Ca(NO 3)2 Example: Write the name for the following compounds Na 3 PO 4 NH 4 Cl. O K 2 CO 3
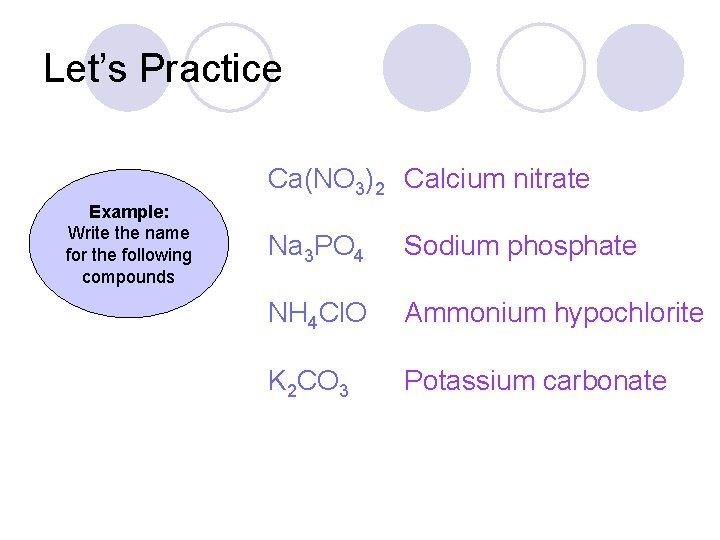
Let’s Practice Ca(NO 3)2 Calcium nitrate Example: Write the name for the following compounds Na 3 PO 4 Sodium phosphate NH 4 Cl. O Ammonium hypochlorite K 2 CO 3 Potassium carbonate

Binary Covalent Compounds Covalent bond atoms share electrons Binary Covalent Compound compound made from two non-metals that share electrons Non metal Covalent compound

Identifying & Naming Binary Covalent l These compounds have: ¡ 2 elements (“binary”) ¡Both non-metals (“covalent”) l To name these compounds: ¡Write the name of the first element with a prefix indicating the number of atoms. One important exception: “Mono” is never used!! ¡Write the name of the second element with the prefix indicating the number of atoms (including “mono-”) and the suffix “ide”

The Appendix of your book (Page 433) has a prefix chart. P 2 O 5
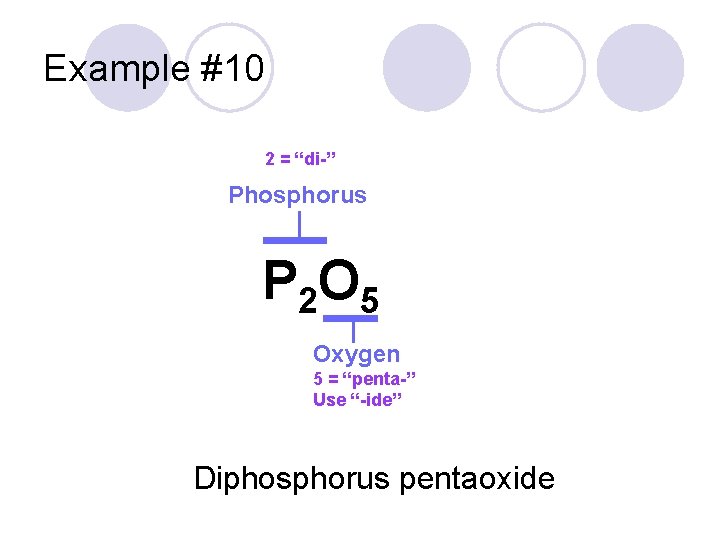
Example #10 2 = “di-” Phosphorus P 2 O 5 Oxygen 5 = “penta-” Use “-ide” Diphosphorus pentaoxide
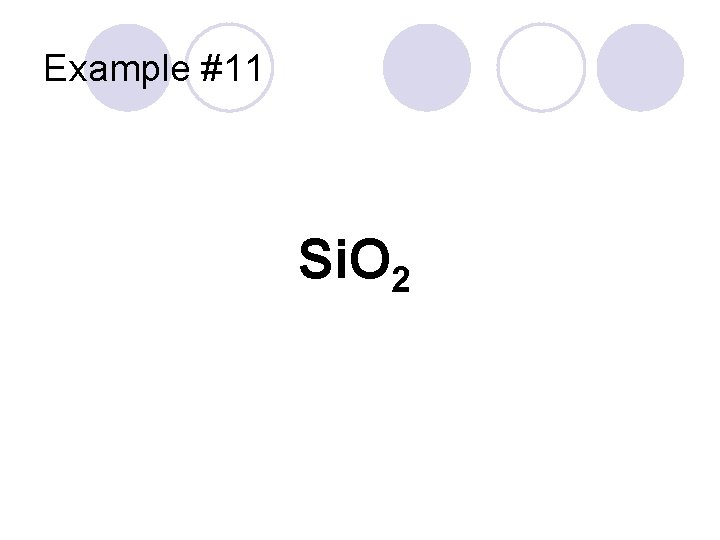
Example #11 Si. O 2

Example #11 Don’t use “mono-” on first element Silicon Si. O 2 Oxygen 2 = “di-” Use “-ide” Silicon dioxide

Let’s Practice CO 2 Example: Write the name for the following compounds N 2 O 4 P 4 O 10 CO

Let’s Practice Example: Write the name for the following compounds CO 2 Carbon dioxide N 2 O 4 Dinitrogen tetraoxide P 4 O 10 Tetraphosphorus decaoxide CO Carbon monoxide
- Slides: 38