Section 17 2 Factors Affecting Chemical Equilibrium Describe

Section 17. 2 Factors Affecting Chemical Equilibrium • Describe how various factors affect chemical equilibrium. • Explain how Le Châtelier’s principle applies to equilibrium systems. When changes are made to a system at equilibrium, the system shifts to a new equilibrium position.

Le Châtelier’s Principle • Le Châtelier’s Principle was proposed in 1888 and states that if stress is applied to a system at equilibrium, the system shifts in the direction that relieves the stress. • Stress is any kind of change in a system that upsets the equilibrium.

Le Châtelier’s Principle (cont. ) • Adjusting the concentrations of either the reactants or the products puts stress on a system in equilibrium. • Adding reactants increases the number of effective collisions between molecules and upsets the equilibrium.

Le Châtelier’s Principle (cont. ) • The equilibrium shifts to the right to produce more products. • Stress is relieved by shifting to the left, converting products to reactants.
• What dose the longer equilibrium arrow to the left or right mean? • It means that, one reaction occurs faster than the other temporarily. Once a new equilibrium position is established, the two reactions again occur at equal rates

Le Châtelier’s Principle (cont. ) • Changing the volume (and pressure) of an equilibrium system shifts the equilibrium only if the number of moles of gaseous reactants is different from the moles of gaseous products. • CO(g)+ 3 H 2(g)⇌ CH 4(g)+ H 2 O(g) • If the number of moles is the same on both sides of the balanced equation, changes in pressure and volume have no effect on the equilibrium.
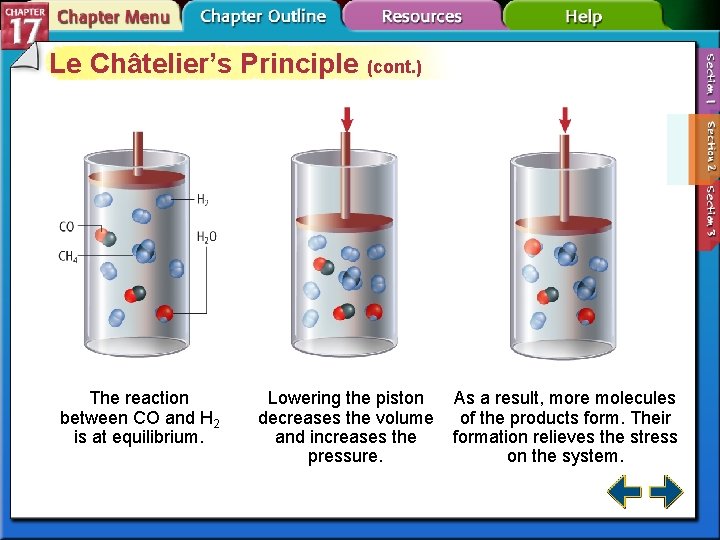
Le Châtelier’s Principle (cont. ) The reaction between CO and H 2 is at equilibrium. Lowering the piston decreases the volume and increases the pressure. As a result, more molecules of the products form. Their formation relieves the stress on the system.

Le Châtelier’s Principle (cont. ) • Changes in temperature alter the equilibrium position and the equilibrium constant. • If heat is added to an equilibrium system, the equilibrium shifts in the direction in which the heat is used up.

Le Châtelier’s Principle (cont. ) • Any change in temperature results in a change in Keq.

Le Châtelier’s Principle (cont. ) • A catalyzed reaction reaches equilibrium more quickly, but with no change in the amount of product formed.

Section 17. 2 Assessment Which does NOT result in a shift of the equilibrium to the right? A. removing products B. adding reactants C. increasing concentration of reactants A. A D. adding products B. B C. C D. D

Section 17. 2 Assessment Any change in ____ results in a change in Keq. A. temperature B. pressure C. volume D. concentration A. B. C. D. A B C D

A change in ____ alters both the equilibrium position and the equilibrium constant. A. pressure B. temperature C. volume D. density A. B. C. D. A B C D

Adding product to a chemical equilibrium: A. does nothing B. creates a stress and shifts the equilibrium to the right C. creates a stress and shifts the equilibrium to the left D. causes more product to form A. B. C. D. A B C D

Ethylene (C 2 H 4) reacts with hydrogen to form ethane (C 2 H 6). C 2 H 4(g) + H 2(g) ↔ C 2 H 6(g) + heat. How could you increase the amount of hydrogen in the system? A. Increase the heat. B. Decrease the heat. C. Increase the C 2 H 4. D. Decrease the C 2 H 6. A. B. C. D. A B C D

Which does NOT result in a shift of the equilibrium to the right? A. removing products B. adding reactants C. increasing concentration of reactants D. decreasing the concentration of reactants A. B. C. D. A B C D

• Q 1: why the addition of a catalyst does not change an equilibrium position. • A catalyst speeds up the forward and reverse reactions equally. • Q 2: Explain how the following stresses affect the equilibrium system. • PF 3(g) + F 2(g) ⇌ PF 5(g) + heat • If volume is decreased: The equilibrium shifts to the right. • Heat is increased : The equilibrium shifts to the left.

• Q 3: Explain how a system at equilibrium responds to a stress and list factors that can be stresses on an equilibrium system. • If possible, the equilibrium shifts in the direction that relieves the stress. changes in concentration, pressure (volume), and temperature

Q 4: Explain how decreasing the volume of the reaction vessel affects each equilibrium. a. 2 SO(g) + O 2(g) ⇌ 2 SO(g) b. H 2 (g) + Cl 2(g) ⇌ 2 HCl(g) a. The equilibrium shifts to the right. b. The stress has no effect on the equilibrium

Q 5: Decide whether higher or lower temperatures will produce more CH 3 CHO in the following equilibrium. C 2 H 2(g) + H 2 O(g) ⇌ CH 3 CHO(g) ∆H° = -151 k. J lower temperatures

Q 6: The table below shows the concentrations of Substances A and B in two reaction mixtures. A and B react according to the equation 2 A ⇌ B; K eq = 200. Are the two mixtures at different equilibrium positions? The two mixtures are at the same equilibrium position.
End of section 17. 2
- Slides: 22