SecondMessenger Gated Ion Channels Tom Mast Membrane Biophysics
Second-Messenger Gated Ion Channels Tom Mast Membrane Biophysics 10/5/07

What is a Second Messenger? An intracellular signal produced in response to a stimulus usually when a ligand binds a receptor ex: cyclic nucleotides (c. AMP or c. GMP) calcium inositol 1, 4, 5 triphosphate (IP 3) diacylglycerol (DAG)
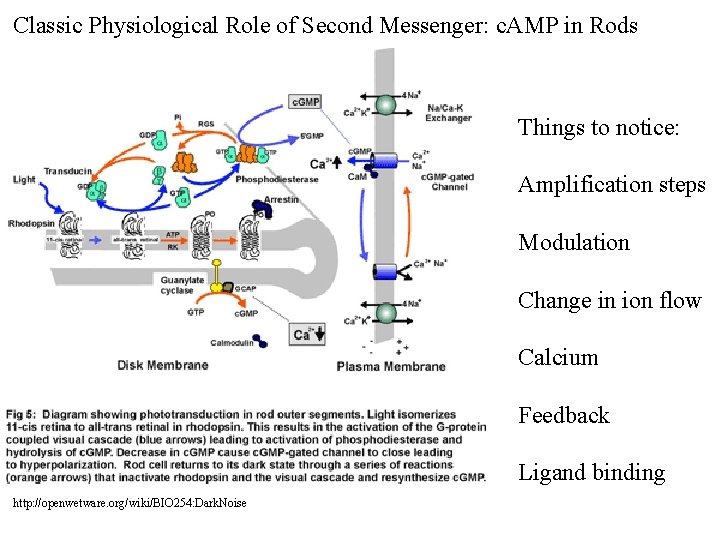
Classic Physiological Role of Second Messenger: c. AMP in Rods Things to notice: Amplification steps Modulation Change in ion flow Calcium Feedback Ligand binding http: //openwetware. org/wiki/BIO 254: Dark. Noise

Second Messenger Channel Topics Ion species flux- specific or not, calcium Structure- subunit architecture pore and selectivity filter conformational changes tetramerization Ligand-binding- what is the ligand conformation/ shape changes kinetics

Which Channels? Cyclic gated nucleotide Channel A 2 Transient Receptor Potential C 2 Inositol 1, 4, 5 Triphosphate 1
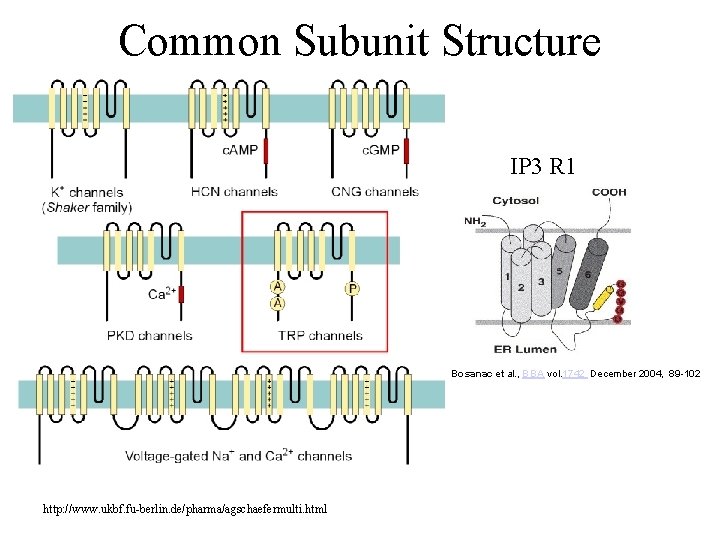
Common Subunit Structure IP 3 R 1 Bosanac et al. , BBA vol. 1742 December 2004, 89 -102 http: //www. ukbf. fu-berlin. de/pharma/agschaefermulti. html

Common Pore Region Channel Pore and Selectivity Sequence Bovine Rod CNGC RKYVYSLYWSTLTLTTIG. . ETPPPV Catfish Olfactory CGNC FCYVYCFYWSTLTLTTIG. . EMPPPV Bacterial K KSA TYPRALWWSVETATTVGYGDLY. PY Shaker K SIPDAFWWAVVTMTTVGYGDMT. PV Mammalian IP 3 R 1 LLMCIVTVLSHGLRSGGGVGDVLRK Mammalian TRPC 2 FNETFQFLFWTMFGMEEHTVVDMP Other regions within the channels are similar (ie S 4) Original channel may have been a 1 TMD Ca++ Strong et al. , Mol. Bio. Evol. 1993 (10) 221 -242 Due to these relatively non-selective pore regions these channels flux cations mainly Na+ and Ca++

TRPC 2 and IP 3 R 1 Signaling C. Badland
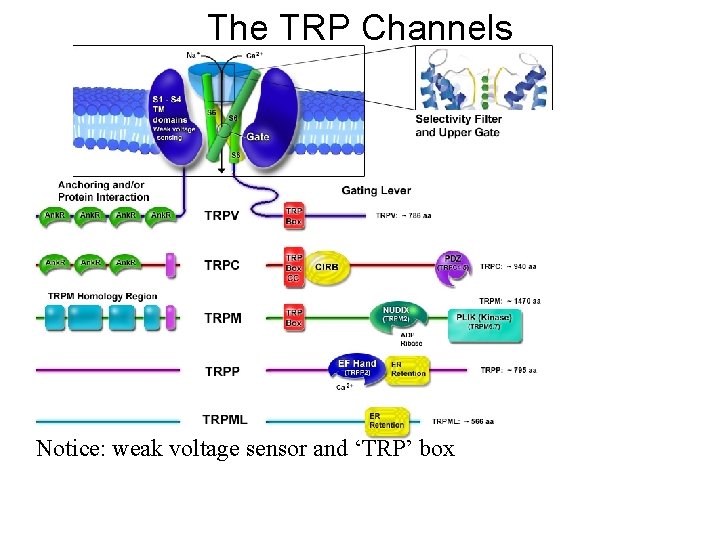
The TRP Channels Notice: weak voltage sensor and ‘TRP’ box

Paper 1: TRPC 2 Lucas P, Ukhanov K, Leinders-Zufall T, Zufall F A Diacylglycerol-Gated Cation Channel in Vomeronasal Neuron Dendrites Is Impaired in TRPC 2 Mutant Mice Neuron. 2003 Oct 30; 40(3): 551 -61. Primary Question: Are DAG-induced currents present in VNO neurons?
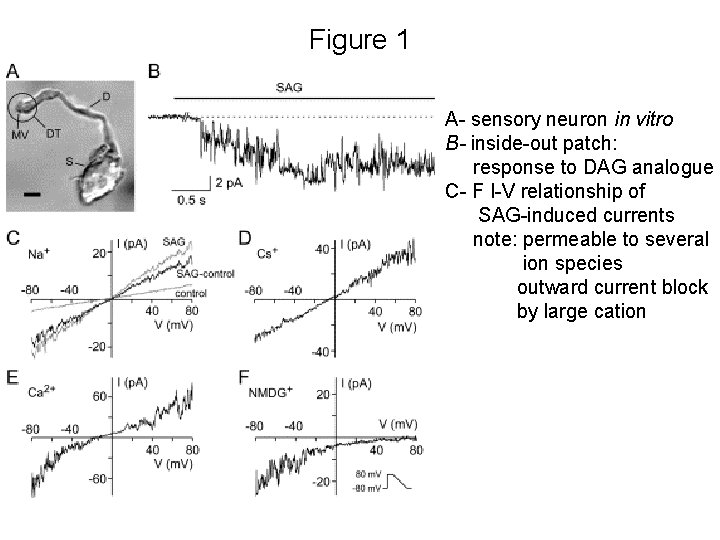
Figure 1 A- sensory neuron in vitro B- inside-out patch: response to DAG analogue C- F I-V relationship of SAG-induced currents note: permeable to several ion species outward current block by large cation
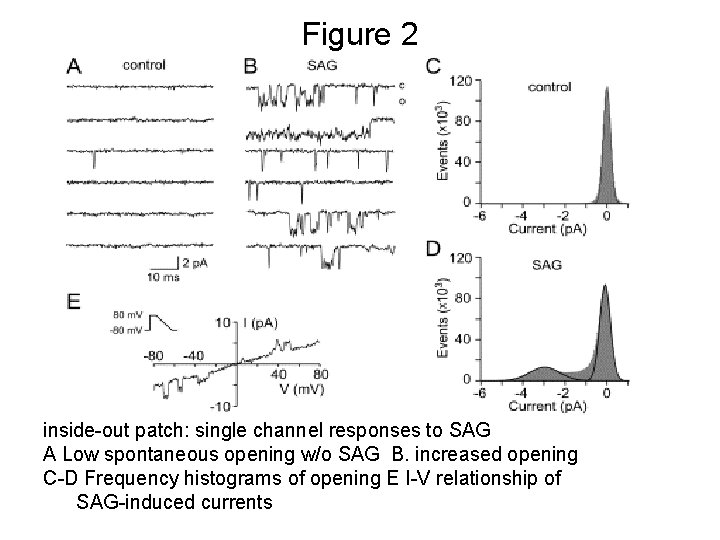
Figure 2 inside-out patch: single channel responses to SAG A Low spontaneous opening w/o SAG B. increased opening C-D Frequency histograms of opening E I-V relationship of SAG-induced currents
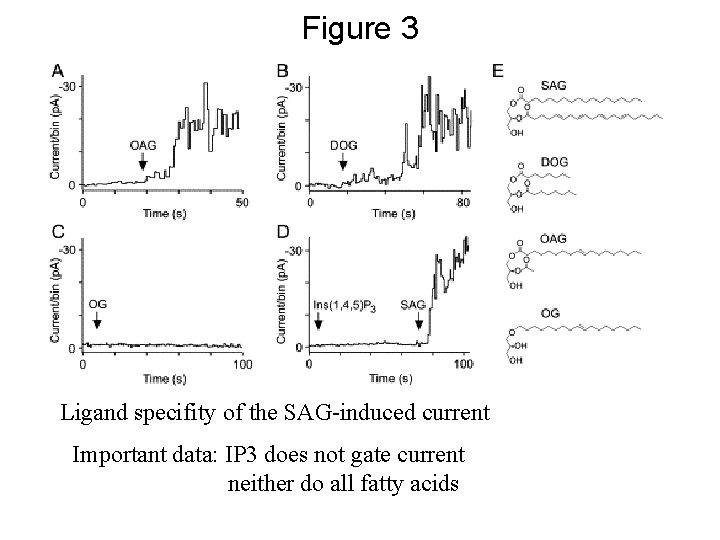
Figure 3 Ligand specifity of the SAG-induced current Important data: IP 3 does not gate current neither do all fatty acids
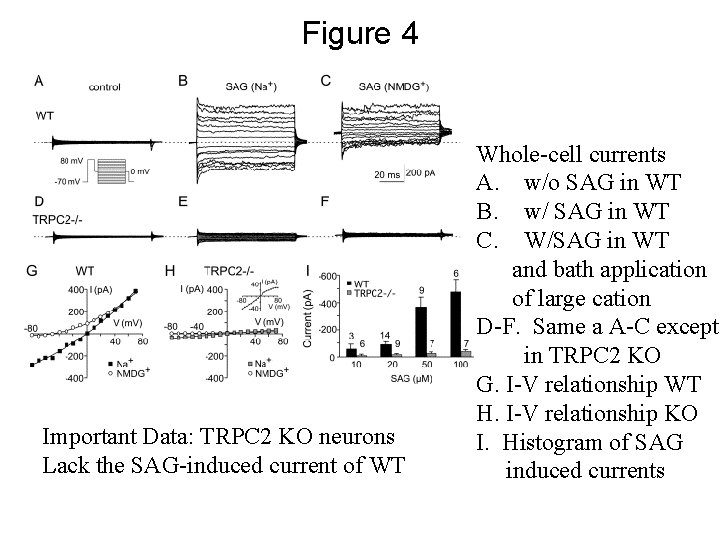
Figure 4 Important Data: TRPC 2 KO neurons Lack the SAG-induced current of WT Whole-cell currents A. w/o SAG in WT B. w/ SAG in WT C. W/SAG in WT and bath application of large cation D-F. Same a A-C except in TRPC 2 KO G. I-V relationship WT H. I-V relationship KO I. Histogram of SAG induced currents
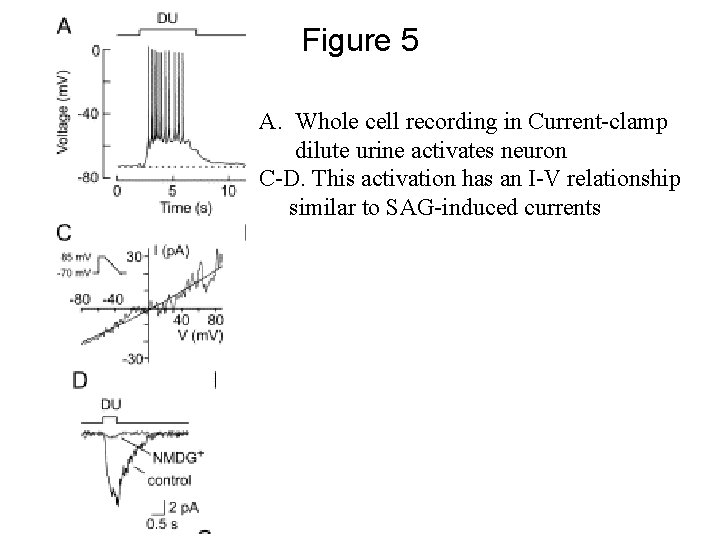
Figure 5 A. Whole cell recording in Current-clamp dilute urine activates neuron C-D. This activation has an I-V relationship similar to SAG-induced currents
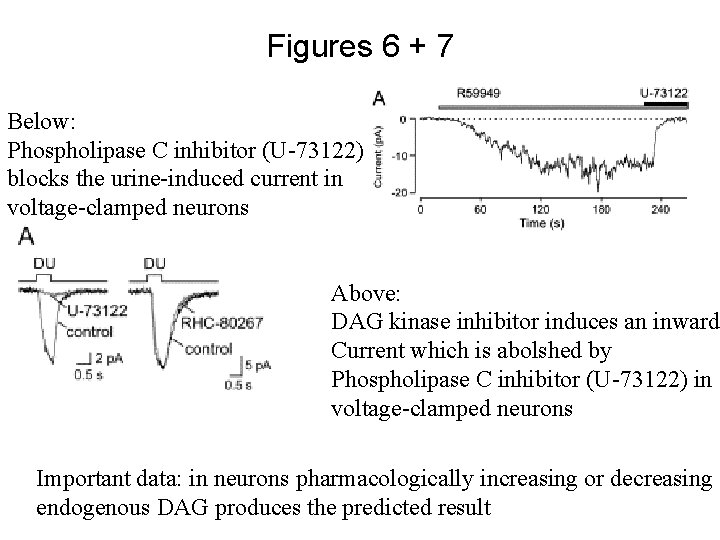
Figures 6 + 7 Below: Phospholipase C inhibitor (U-73122) blocks the urine-induced current in voltage-clamped neurons Above: DAG kinase inhibitor induces an inward Current which is abolshed by Phospholipase C inhibitor (U-73122) in voltage-clamped neurons Important data: in neurons pharmacologically increasing or decreasing endogenous DAG produces the predicted result

Conclusions A urine induced current is non-selective for external cations It is dependent on PLC It is abolished by gene-targeted deletion of TRPC 2 It closely resembles that of a SAG-induced current

Paper 2: IP 3 R 1 Hamada K, Terauchi A, Mikoshiba K. Three-dimensional rearrangements within inositol 1, 4, 5 trisphosphate receptor by calcium. J Biol Chem. 2003 Dec 26; 278(52): 52881 -9. Primary Question: How do the allosteric factors Ca++ and IP 3 effect conformational changes in the channel?
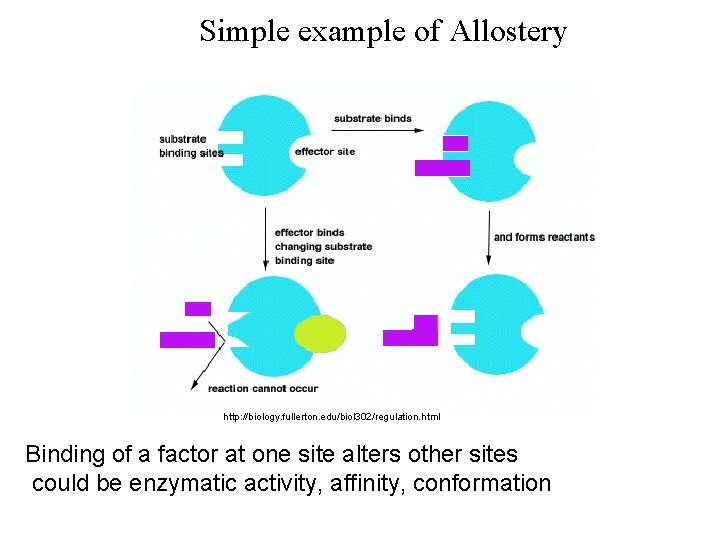
Simple example of Allostery http: //biology. fullerton. edu/biol 302/regulation. html Binding of a factor at one site alters other sites could be enzymatic activity, affinity, conformation

Figure 1 A. Incubation of IP 3 R 1 with a lysine-protease results in different fragment patterns dependent on Ca++ concentration B. Also dependent on C-term of cytoplasmic domain which is involved in tetramerization C. Location of epitopes used in western analysis
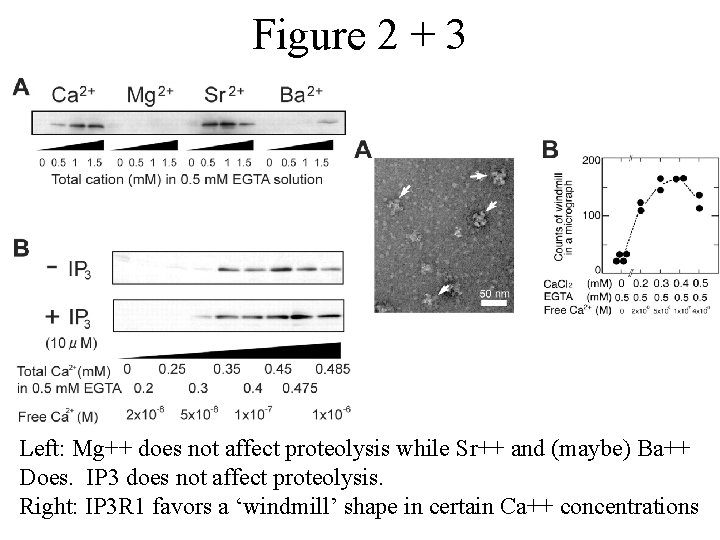
Figure 2 + 3 Left: Mg++ does not affect proteolysis while Sr++ and (maybe) Ba++ Does. IP 3 does not affect proteolysis. Right: IP 3 R 1 favors a ‘windmill’ shape in certain Ca++ concentrations
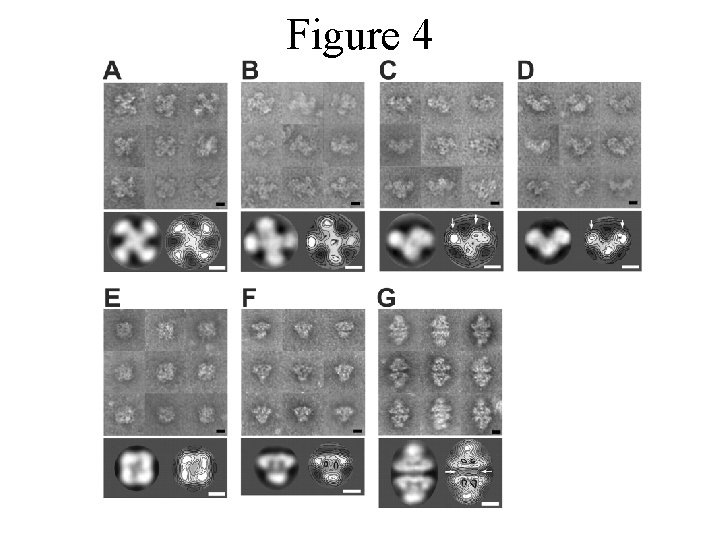
Figure 4
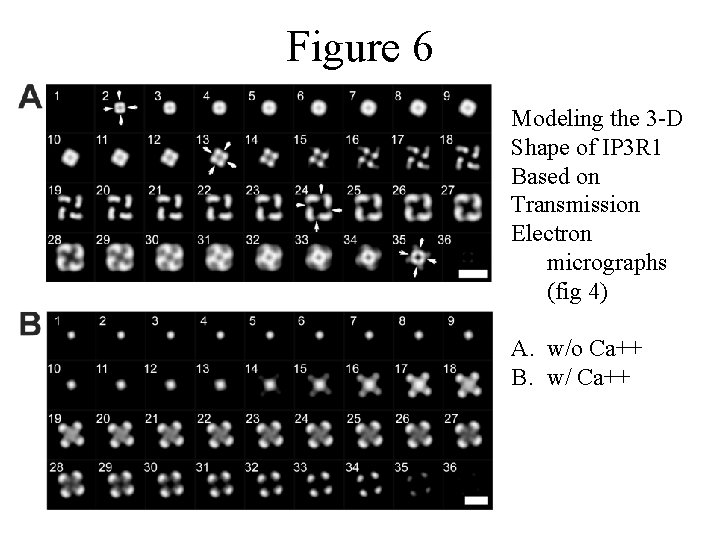
Figure 6 Modeling the 3 -D Shape of IP 3 R 1 Based on Transmission Electron micrographs (fig 4) A. w/o Ca++ B. w/ Ca++
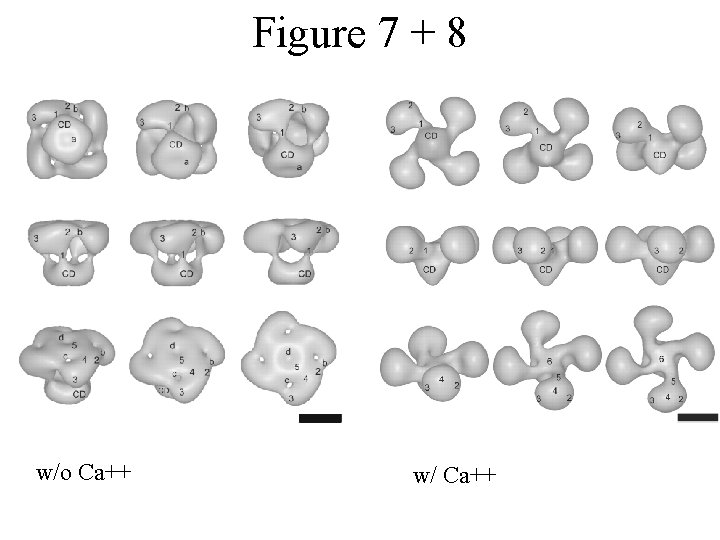
Figure 7 + 8 w/o Ca++ w/ Ca++

Conclusions IP 3 R 1 is sensitive to Ca++ Channel-wide conformational changes are due to Ca++ binding and not IP 3 Tetramerization may play a role in the conformational changes

Paper 3: CNGA 2 Nache V, Schulz E, Zimmer T, Kusch J, Biskup C, Koopmann R, Hagen V, Benndorf K Activation of olfactory-type cyclic nucleotide-gated channels is highly cooperative J Physiol. 2005 Nov 15; 569(Pt 1): 91 -102 Primary Question: What is the allosteric model for c. GMP binding to CNGA 2?
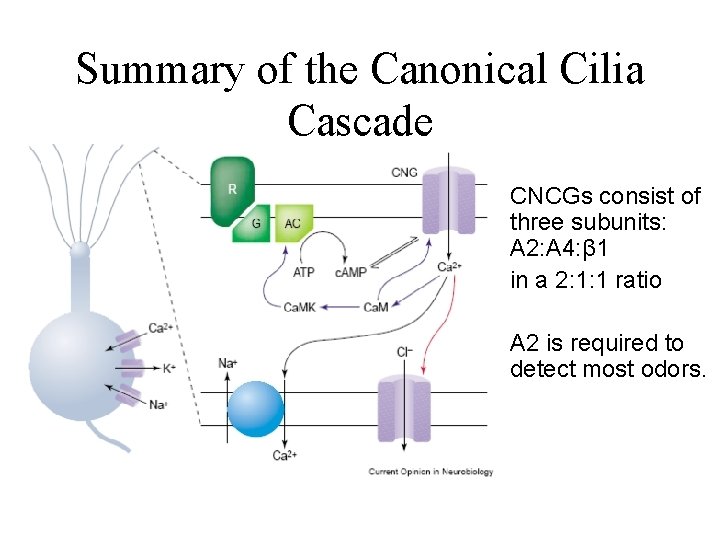
Summary of the Canonical Cilia Cascade CNCGs consist of three subunits: A 2: A 4: β 1 in a 2: 1: 1 ratio A 2 is required to detect most odors.
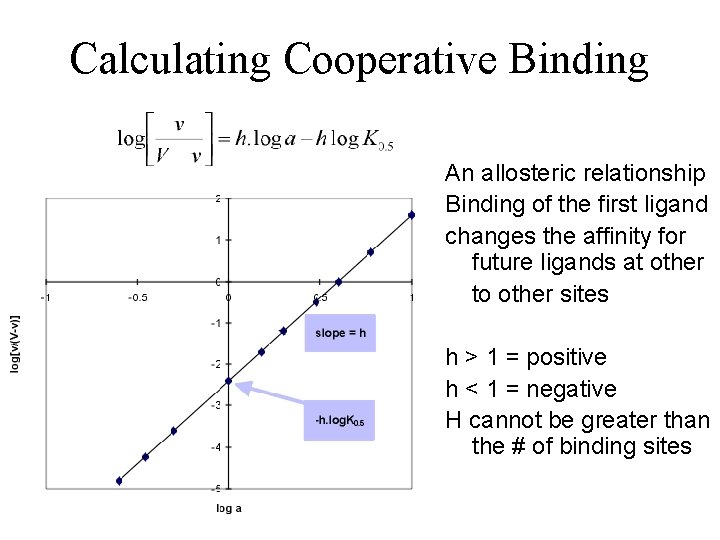
Calculating Cooperative Binding An allosteric relationship Binding of the first ligand changes the affinity for future ligands at other to other sites h > 1 = positive h < 1 = negative H cannot be greater than the # of binding sites
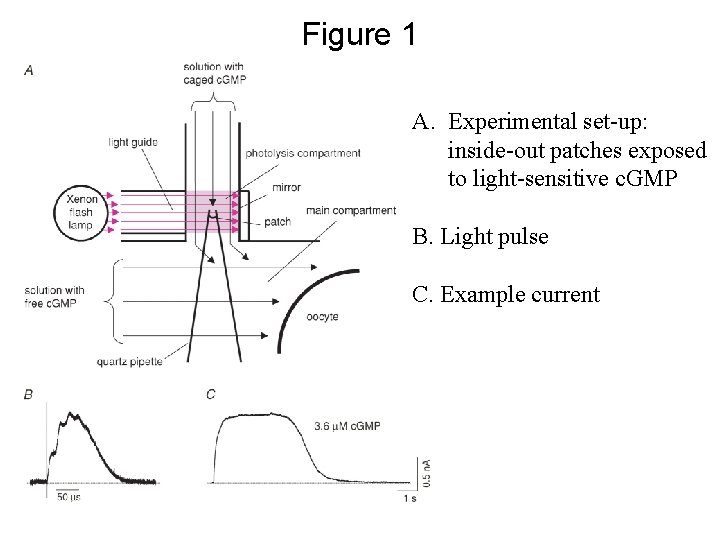
Figure 1 A. Experimental set-up: inside-out patches exposed to light-sensitive c. GMP B. Light pulse C. Example current
![Figure 2 A. Example of current used for calculations B-D [c. XMP]-response curves with Figure 2 A. Example of current used for calculations B-D [c. XMP]-response curves with](http://slidetodoc.com/presentation_image/80b6432bb05f9a03ea195b99dabf1efa/image-30.jpg)
Figure 2 A. Example of current used for calculations B-D [c. XMP]-response curves with calculated parameters
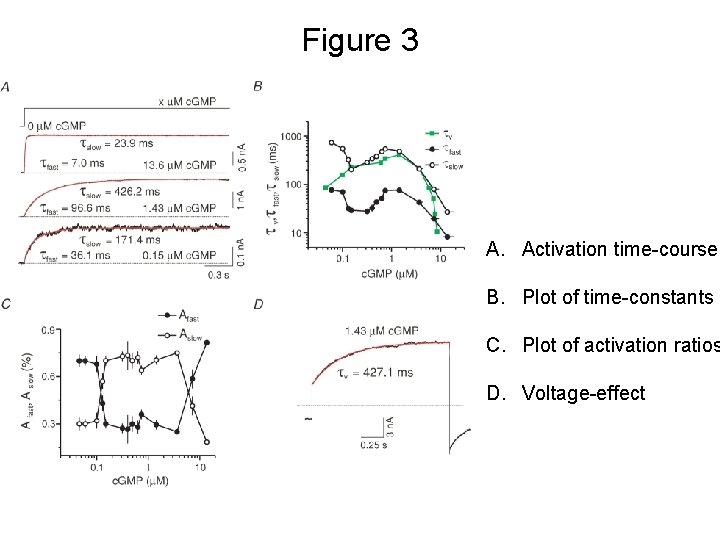
Figure 3 A. Activation time-courses B. Plot of time-constants C. Plot of activation ratios D. Voltage-effect
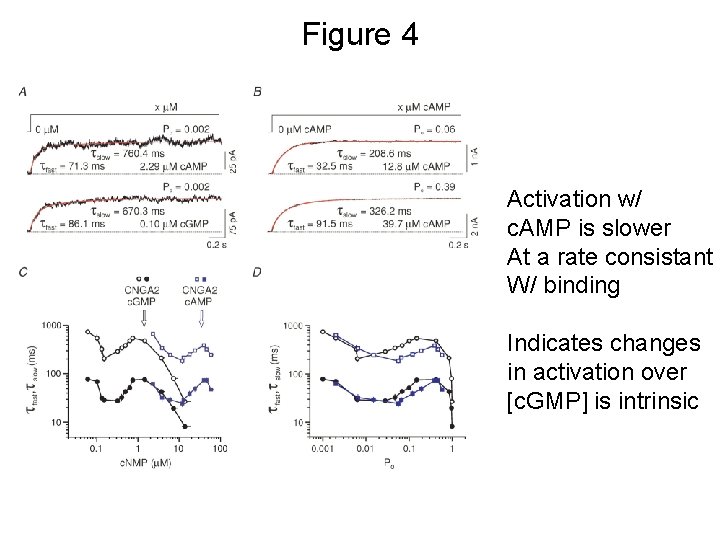
Figure 4 Activation w/ c. AMP is slower At a rate consistant W/ binding Indicates changes in activation over [c. GMP] is intrinsic
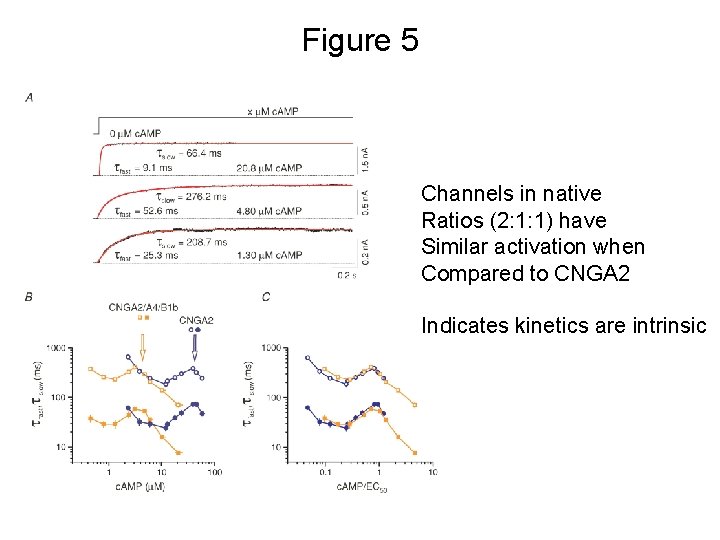
Figure 5 Channels in native Ratios (2: 1: 1) have Similar activation when Compared to CNGA 2 Indicates kinetics are intrinsic
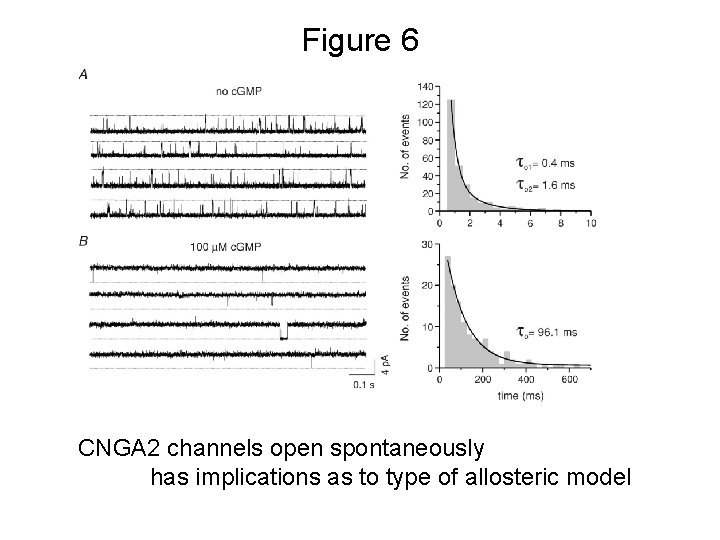
Figure 6 CNGA 2 channels open spontaneously has implications as to type of allosteric model
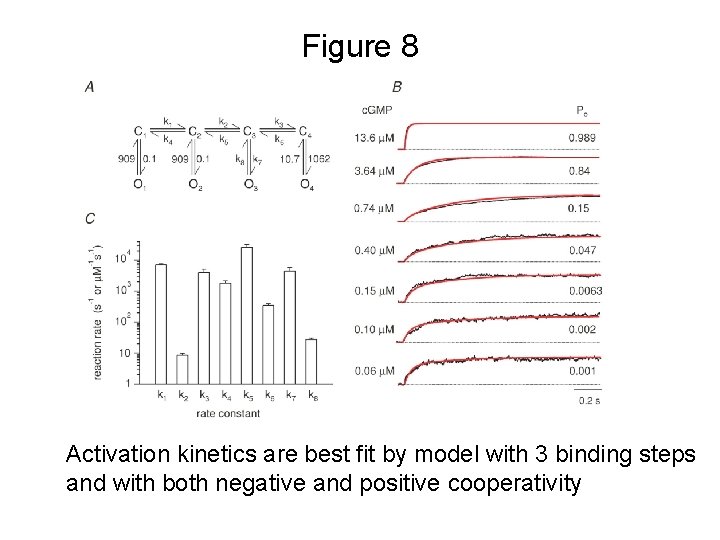
Figure 8 Activation kinetics are best fit by model with 3 binding steps and with both negative and positive cooperativity

Conclusions CNGA 2 channels have a greater affinity for c. GMP CNGA 2 channels display cooperative binding CNGA 2 and hetereomultimer channels are affected by Vm
- Slides: 36