Second Level Clinical Pharmacy Students By Prof Dr


Second Level Clinical Pharmacy Students By Prof. Dr. Osama Abd El-Azeem Soliman, Ph. D. Professor of Pharmaceutics, Faculty of Pharmacy, Mansoura University

Microencapsulation Lecture (4) B- Complex Coacervation As acacia contains only free carboxylic groups, it always carries a negative charge.

At acidic p. H, gelatin in aqueous solution will acquire a positive charge, which attracts protons from the water and help bind water to gelatin molecules.

Any factor that influences; 1. Hydration, or 2. Charge, such as; 1. solvent addition, 2. p. H change, 3. temperature change, or 4. electrolyte addition

will affect the amount of water bound, which in turn will affect the: 1. solubility or 2. dispersibility of the colloid.

Complex coacervation involves the uses of more than one colloid. 1. gelatin and (+ ve charge) 2. Acacia (- ve charge) in water are the most frequently used, The coacervation is accomplished mainly by charge utilization of the colloids carrying opposite charges.

Two mechanisms have been proposed for the formation of microcapsules: 1. Individual coacervate droplets may be attracted to and coalesce around core particles immiscible in the system and/or, 2. A single coacervate droplet may encompass one or more core particles.

The ratio is 1: 1 of the two colloids The concentration is (10 % of each) about 20% of both.

Coacervation is occured by: 1. p. H adjustment. (charge) 2. dilution if necessary. (hydration) and promoted by decreasing of temperature.

The coacervate droplets will deposit on the core particles, serve as nuclei, until the coacervate finally surrounds the core material completely.

If only one particle is enclosed in coacervate, a mononuclear microcapsule is obtained; while, if more than one particle is enclosed, a multinuclear microcapsules are formed.

• When the system cooled, the coating is start to gelate (congealing). • This is fixed by adding a suitable hardening agent as formaldhyde, • Elevation of the p. H to 9 by alkali, neutralize the excess acid. • And gradual rising of the temperature to 500 C to complete the curing of the capsule walls.

Finally, after washing of residual hardening agent, the microcapsules may be recovered by: 1. Filtration, 2. Centrifugation or 3. Decantation

it may be converted into a dry powder by removal of residual water using a; 1. spray drying 2. freeze drying or 3. fluidized bed drying.

The Following are typical steps of a complex coacervation process for microencapsulation

2 -Coacervation - Using non aqueous vehicles. Many drugs are moderate to very watersoluble, So, Phase separation of the polymer may by induced by different methods such as; 1. Temperature change, 2. Addition of incompatible polymer, 3. Solvent alteration.

Examples of Polymers used for microencapsulation using non-aqueous vehicles include; Cellulose acetate phthalate (CAP), Hydroxypropyl ethylcellulose phthalate, Carboxy methylcellulose, Cellulose nitrate, Polystyrene, Acrylate.

A- Coacervation induced by temperature change: The process was used for heat-stable drugs. It involves; • dissolve ethyl cellulose (Polymer) in cyclohexane (solvent) at 80 -850 C. • Disperse the drug in the system.

• Gradually cool the solution so that the polymer separates as a liquid coacervate and encloses particles of core material that are dispersed by vigorous agitation in the system.

• The coat may be hardened by lowering the temperature while maintaining vigorous agitation to prevent coalescence of microcapsules. • When the system reaches 20 to 250 C, the microcapsules can be filtered off from cyclohexane and dried.

B- Incompatible polymer addition Coacervation can be accomplished by utilizing the incompatibility of dissimilar polymer existing in a common solvent. Examples: -

• Ethyl cellulose (polymer 1) was dissolved • in cyclohexane (solvent) at 80 -850 C in • presence of core particles of aspirin (drug) and • low molecular Polyethylene as an incompatible polymer (polymer 2), • using vigorous agitation.

The system was then cooled, ethyl cellulose separated out encapsulating the core material. This example combined the use of temperature and incompatible polymer addition.

Exampe 2 Ethyl cellulose (polymer 1) is dissolved in toluene to yield a polymer concentration of 2% by weight. Methylene blue hydrochloride being insoluble in toluene, is dispersed with stirring in the polymer solution at a ratio of 4 parts methylene blue hydrochloride to 1 part ethylcellulose.

Phase separation is accomplished by slowly adding liquid polybutadiene (polymer 2) in sufficient quantity to yield a ratio of 25 parts polybutadiene to one part ethylcellulose. M. B. H : ethylcellulose : polybutadiene 4 1 (2%) 25 The polybutadiene being quite soluble in toluene and incompatible with ethylcellulose.

Ethylcellulose coating is solidified by adding a non-solvent for the coating polymer ethyl cellulose such as hexane. Also, the Polybutadiene, being soluble in hexane, is washed from the mixture by decantation and additional hexane.

C- Coacervation induced by solvent alteration (Non-Solvent addition). A liquid that is a non-solvent for a given polymer can be added to a solution of the polymer to induce phase separation. The resulting immiscible, liquid Polymer can be utilized to affect microencapsulation of an immiscible core material as illustrated in the following example.

• A 5% w/v methyl ketone (solvent 1) solution of cellulose acetate butyrate (polymer) is prepared, • And in it, micronzed methyl scopolamine hydrobormide (drug) is dispersed with stirring. • A core material to coating material ratio (methyl scopolamine hydrobromide to cellulose acetate butyrate) of about 2: 1 is used.

• • Heat to 550 C, Add isopropyl ether (solvent 2), • The system is slowly cooled to room temperature, and the microencapsulated particles are separated by centrifugation, washed with isopropyl ether, and dried in vacuum.

II- Interfacial Polycondensation It involves the reaction of various monomers at the interface between two immiscible liquid phases to form a film of polymer that encapsulates the dispersed phase.
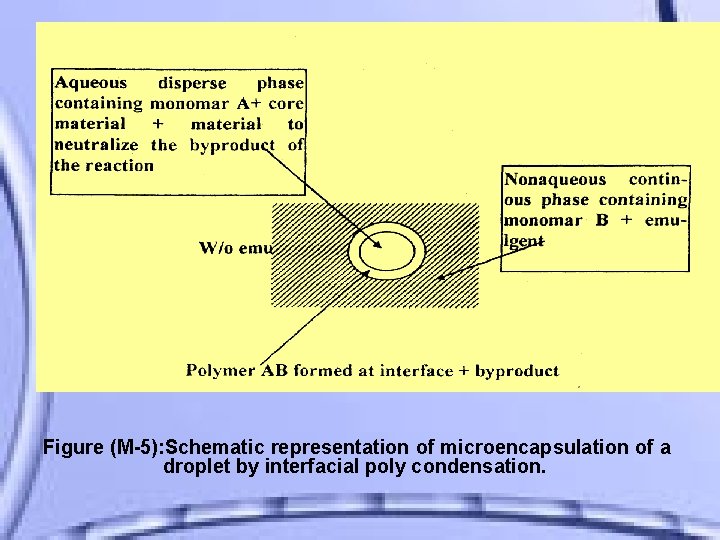
Figure (M-5): Schematic representation of microencapsulation of a droplet by interfacial poly condensation.

Usually two reactive monomers are employed, one dissolved in the aqueous disperse phase containing a solution or dispersion of the core material, and the other monomer dissolved after emulsification step in the non aqueous continuous phase.

• The monomers diffuse together and rapidly polymerize at the interface to form a thin coating, and the byproduct of the reaction is neutralized by adding material such as an alkaline buffer.

The degree of polymerization can be controlled by: - 1. 2. 3. 4. the reactivity of the monomers chosen, their concentrations, the composition of the phases, the temperature of the system.

This method is Not exploited, because of: - 1. Toxicity problems associated with un-reacted monomers, the polymer, or other constituents of the system. 2. Excessive drug degradation caused by reaction with monomers. 3. The high permeability of the coat formed to low molecular weight species. 4. The fragility of the microcapsules formed. 5. The lack of biodegradability of the product.

Polyamide e. g. Nylon 6 -10 microcapsules The reaction between an acid dichloride and a compound containing reactive hydrogen atom (-NH, - OH, - SH), is the bases for such interfacial polymerization.
![H 2 N(CH 2)6 -NH 2 + Cl-CO-(CH 2)8 CO-Cl [HN(CH 2)6 -NHCO(CH 2)-CO-] H 2 N(CH 2)6 -NH 2 + Cl-CO-(CH 2)8 CO-Cl [HN(CH 2)6 -NHCO(CH 2)-CO-]](http://slidetodoc.com/presentation_image_h2/96a437383919573ba1f3de67ff5a0ca7/image-38.jpg)
H 2 N(CH 2)6 -NH 2 + Cl-CO-(CH 2)8 CO-Cl [HN(CH 2)6 -NHCO(CH 2)-CO-] + HCl 1, 6 -hexamethylenediamine + Sebacoyl Chloride Nylon 6, 10 + Hcl As an example of this type of reaction, a poly (hexamethylene sebacamide) polymer was formed at the interface between a solution of 1, 6 -hexamethylene diamine in water and sebacoyl chloride in a water-immiscible solvent.

An inorganic base such as sodium bicarbonate or sodium hydroxide in the aqueous phase was used to neutralize the hydrogen chloride formed in the condensation reaction. The polyamide polymer formed is called nylon.

The water - in - oil (w/o) emulsion formed requires the addition of a suitable emulgent (span 85 ) as stabilizer. The following figure M-5 shows a diagrammatic representation of the process, which is often Polymerization. referred as interfacial
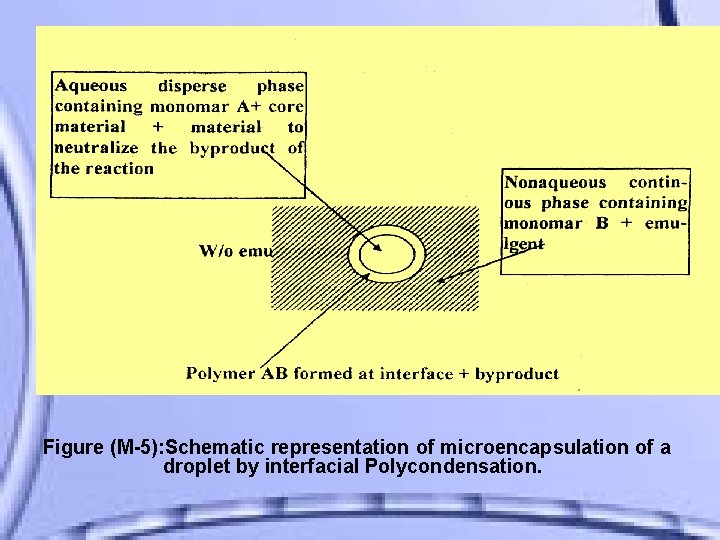
Figure (M-5): Schematic representation of microencapsulation of a droplet by interfacial Polycondensation.

Table (1): Principle monomer combinations for the microencapsulation by interfacial poly condensation. Aqueous phase monomer A Nonaqueous phase monomer B Polymer AB wall material formed 1 - polyamide e. g. -1, 6 -hexamethylene diamine - Piperazine - L-lysine polybasic acid halide i. e. - Sebacoyl chloride 2 - Polyphenol e. g. 2, 2 -bis (4 hydroxyphenyl) propane polybasic acid halide - Sebacoyl chloride Polyester polyphenyl ester. 3 - Polyamide e. g. 1, 6 -hexamethyelene diamine Bischloroformate 2, 2 dichlorodiethyl ether polyurethanpe polyurethane Polyamide. Nylon 6 -10 -Terphthaloyl chloride Polyterephthalamide poly(terephthaloyl-L-lysine


Limitations and considerations for interfacial polycondensation 1. Low-molecular-weight ions such as urea, creatinine, glucose, and salicylic acid readily permeate the Nylon wall formed.

2. The bicarbonate buffer is included to neutralize the hydrogen liberated in reaction, otherwise the encapsulated material. chloride polymerization damage the

• Organic liquids such as chloroform, cyclohexane, and CCl 4 are not readily denature proteins and in suitable combination can be matched to the density of the microcapsules, so as to reduce their rate of sedimentation.

• The partition coefficient of the diamine between the aqueous and nonaqueous phase is very important and is influenced by the composition of the latter. • Sebacoyl chloride is almost insoluble in water, while 1, 6 -hexamethylene diamine soluble in both, so, the wall will be near the organic phase.

• Emulsification of the aqueous and nonaqueous phases is aided by inclusion of a suitable nonionic emulgent such as 1% sorbitan trioleate, HLB 1. 8 (span 85), • The emulsion formed is cooled by immersion in an ice bath to avoid the backward reaction.

- Slides: 49