SECOND LAW OF THERMODYNAMICS Prepared by T Karthikeyan

SECOND LAW OF THERMODYNAMICS Prepared by T. Karthikeyan Assistant Professor Department of Mechanical Engineering, Saveetha Engineering College, Chennai

LIMITATIONS OF FIRST LAW • Ex 1: Coffee-Atmosphere • The reverse process, the hot coffee getting hotter in the cooler room as a result of heat transfer from the room • This process never takes place, still doing so will not violate first law. • Since first law is: Amount of energy lost by surrounds = gained by the coffee

LIMITATIONS OF FIRST LAW-Contd. . • First law places no restriction on the direction of a process. • Ex 2: Paddle wheel-Falling mass • The reverse process, raising the mass by transferring heat from the fluid to the paddle wheel, does not occur in nature. • Although, doing so would not violate the first law.

• It is clear from these arguments that processes proceed in a certain direction and not in the reverse direction. • The first law places no restriction on the direction of a process, but satisfying the first law does not ensure that the process can actually occur. • This inadequacy of the first law to identify whether a process can take place is remedied by introducing another general principle, the second law of thermodynamics. Process occur in a certain direction and not in the reverse direction

SECOND LAW • It is known that the ball (or) water possesses more potential energy at higher level than at lower level. • This suggests that these spontaneous processes proceed by decrease of energy. (ie. , from higher level to lower level). • The state of lowest energy corresponds to the state of maximum stability. • Therefore, the natural tendency of all systems in this universe to go from instability to stability supports the fact that any process which involves a decrease in energy should have an inherent tendency to take place (not the other way).
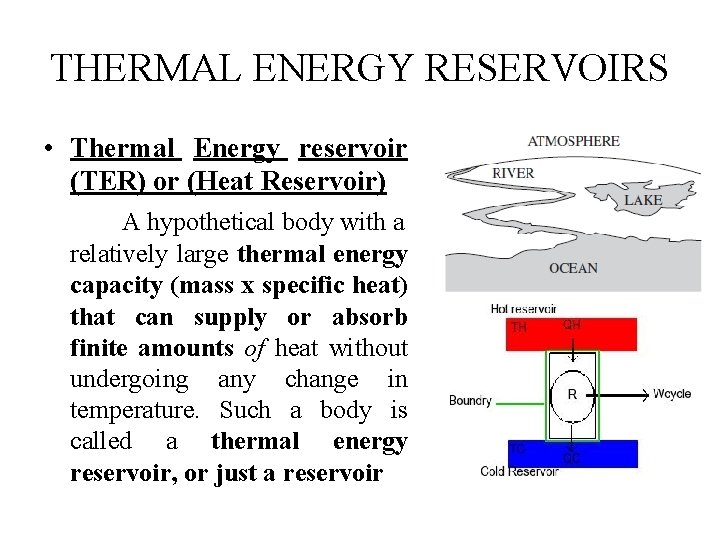
THERMAL ENERGY RESERVOIRS • Thermal Energy reservoir (TER) or (Heat Reservoir) A hypothetical body with a relatively large thermal energy capacity (mass x specific heat) that can supply or absorb finite amounts of heat without undergoing any change in temperature. Such a body is called a thermal energy reservoir, or just a reservoir

• In practice, large bodies of water such as oceans, lakes, and rivers as well as the atmospheric air can be modeled accurately as thermal energy reservoirs because of their large thermal energy storage capabilities or thermal masses. • The atmosphere, for example, does not warm up as a result of heat losses from residential buildings in winter. • Likewise, megajoules of waste energy dumped in large rivers by power plants do not cause any significant change in water temperature.

• The air in a room, for example, can be treated as a reservoir in the analysis of the heat dissipation from a TV set in the room, since the amount of heat transfer from the TV set to the room air is not large enough to have a noticeable effect on the room air temperature. HEAT
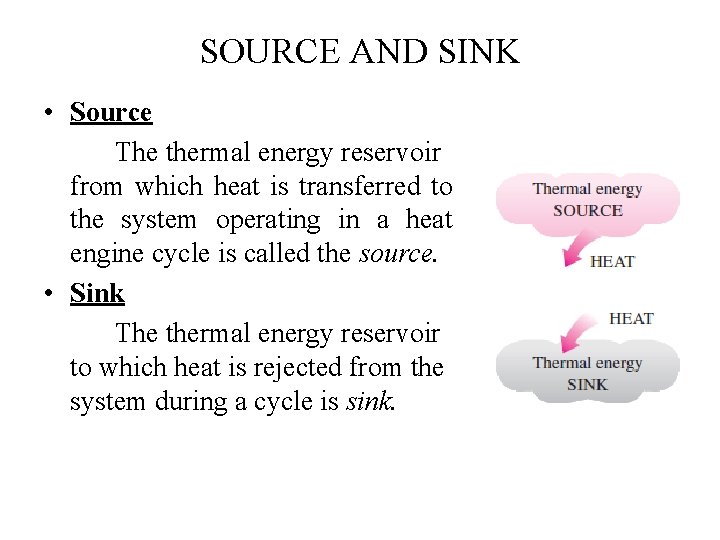
SOURCE AND SINK • Source The thermal energy reservoir from which heat is transferred to the system operating in a heat engine cycle is called the source. • Sink The thermal energy reservoir to which heat is rejected from the system during a cycle is sink.

SOURCE AND SINK • A reservoir that supplies energy in the form of heat is called a source. • A reservoir that absorbs energy in the form of heat is called a sink. • Thermal energy reservoirs are often referred to as heat reservoirs since they supply or absorb energy in the form of heat.
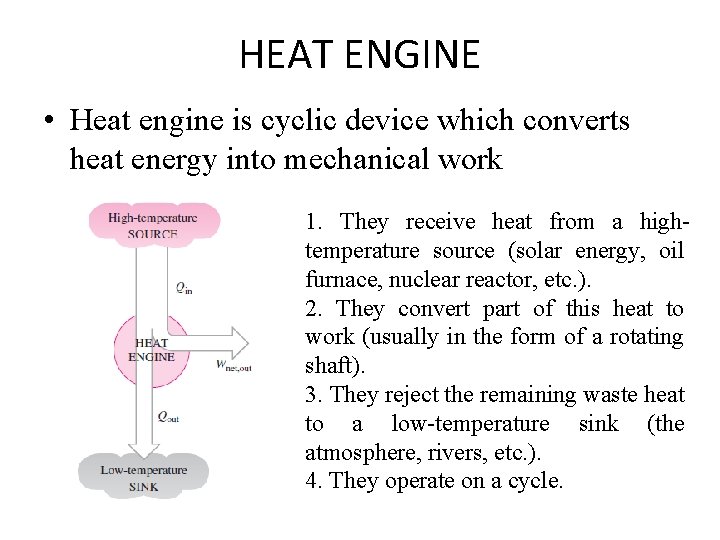
HEAT ENGINE • Heat engine is cyclic device which converts heat energy into mechanical work 1. They receive heat from a hightemperature source (solar energy, oil furnace, nuclear reactor, etc. ). 2. They convert part of this heat to work (usually in the form of a rotating shaft). 3. They reject the remaining waste heat to a low-temperature sink (the atmosphere, rivers, etc. ). 4. They operate on a cycle.
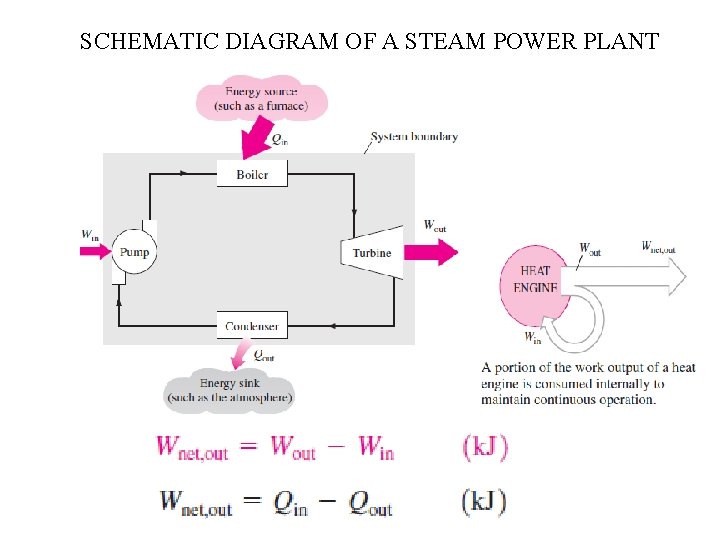
SCHEMATIC DIAGRAM OF A STEAM POWER PLANT
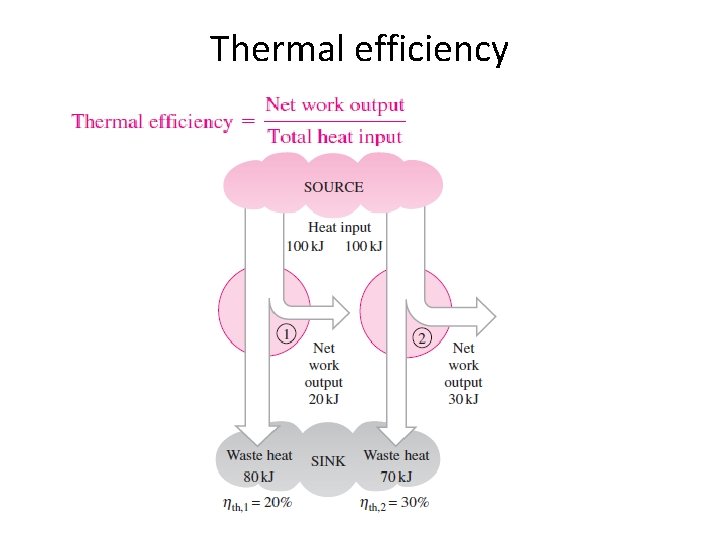
Thermal efficiency
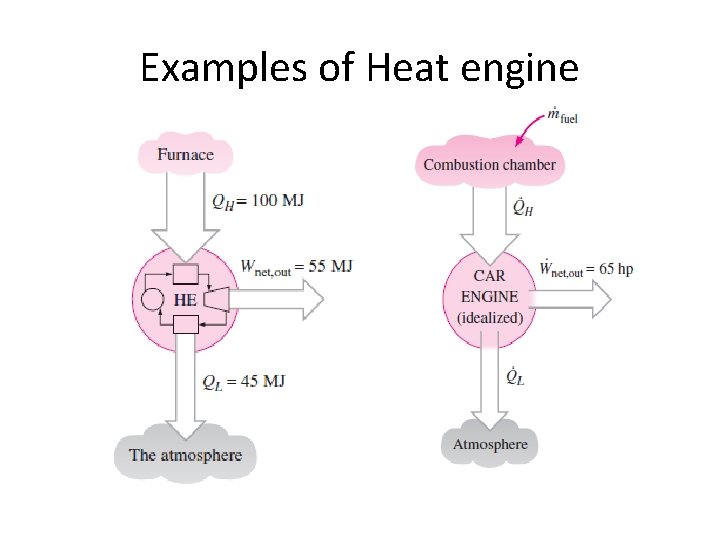
Examples of Heat engine
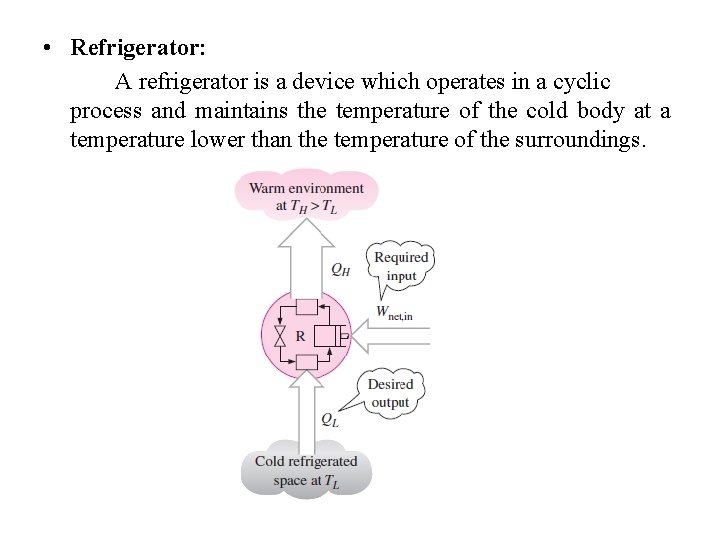
• Refrigerator: A refrigerator is a device which operates in a cyclic process and maintains the temperature of the cold body at a temperature lower than the temperature of the surroundings.

• The efficiency of a refrigerator is expressed in terms of the coefficient of performance (COP), denoted by COPR. The objective of a refrigerator is to remove heat (QL) from the refrigerated space. To accomplish this objective, it requires a work input of Wnet, in. Wnet=QH-QL
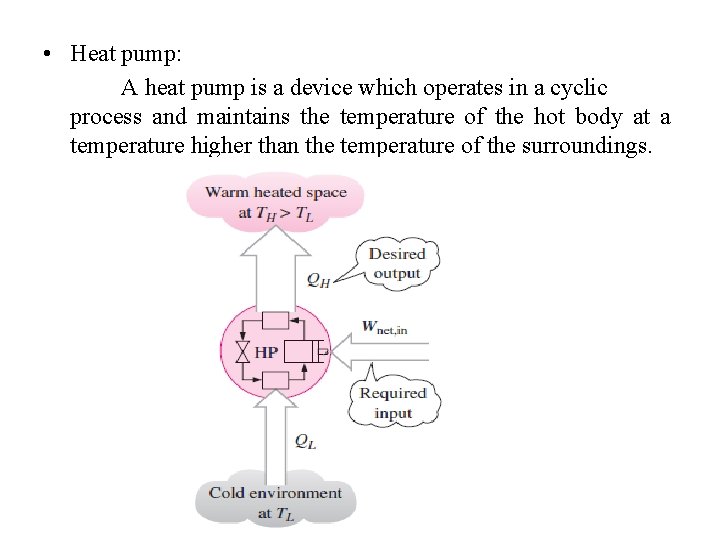
• Heat pump: A heat pump is a device which operates in a cyclic process and maintains the temperature of the hot body at a temperature higher than the temperature of the surroundings.

• The measure of performance of a heat pump is also expressed in terms of the coefficient of performance COPHP, defined as Wnet=QH-QL

STATEMENTS OF SECOND LAW OF THERMODYNAMICS KELVIN-PLANCK’S Statement: It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy from a single thermal reservoir into an equivalent amount of work. Also, no actual engine working on a cyclic process can convert whole of the heat supplied to it into mechanical work.
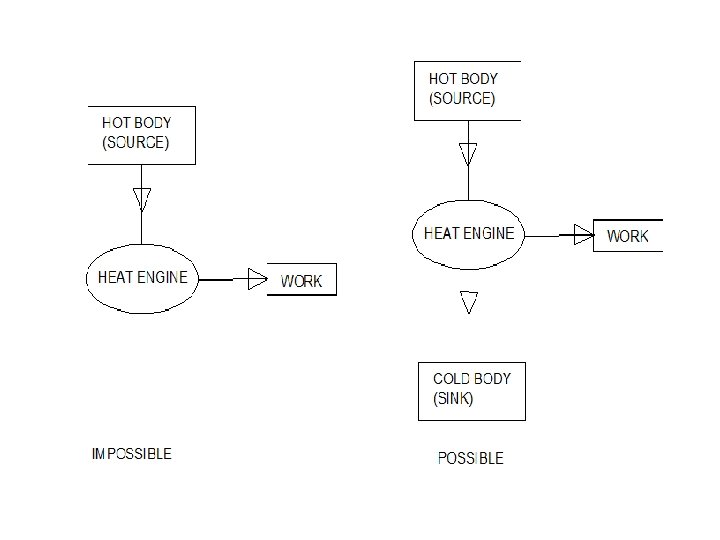

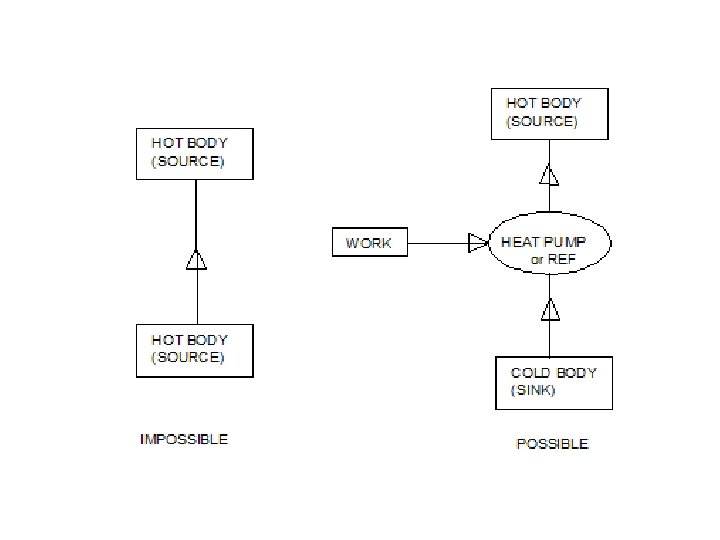
CLAUSIUS STATEMENT: It is impossible for a self acting machine, working in a cyclic process, to transfer heat from a body at a lower temperature to a body at higher temperature without the aid of an external agency.

PERPETUAL MOTION MACHINES • PMMK-1: A device that violates the first law of thermodynamics (by creating energy) is called a perpetual-motion machine of the first kind (PMMK 1) A perpetual motion machine of the first kind produces work without the input of energy. It thus violates the first law of thermodynamics: the law of conservation of energy.

• PMMK 2: A device that violates the second law of thermodynamics is called a perpetual-motion machine of the second kind (PMM 2). A perpetual motion machine of the second kind is a machine which spontaneously converts thermal energy into mechanical work. When thermal energy is equivalent to the work done, this does not violate the law of conservation of energy. However, it does violate the more subtle second law of thermodynamics.

CARNOT CYCLE • The Carnot cycle is a theoretical thermodynamic cycle proposed by Nicolas Léonard Sadi Carnot in 1824. • It gives us the maximum possible thermal efficiency of the system in case of heat engine and the maximum possible COP in case of Refrigeration and Heat pump.
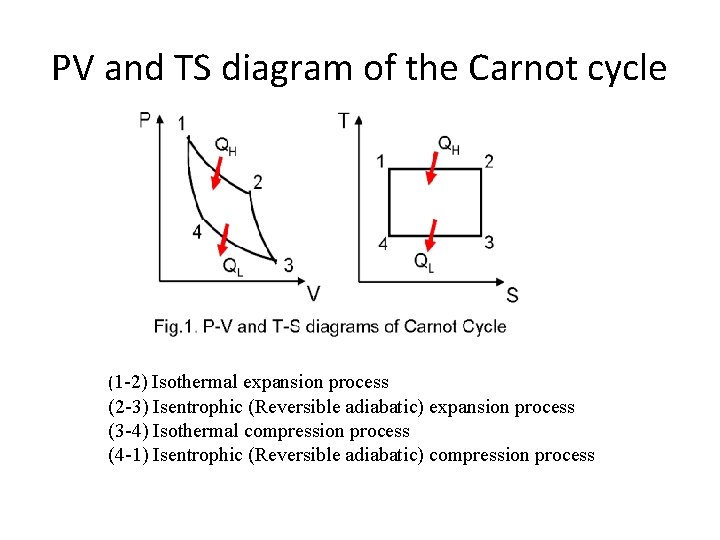
PV and TS diagram of the Carnot cycle (1 -2) Isothermal expansion process (2 -3) Isentrophic (Reversible adiabatic) expansion process (3 -4) Isothermal compression process (4 -1) Isentrophic (Reversible adiabatic) compression process

CLAUSIUS INEQUALITY • It states that whenever a closed system undergoes a cyclic process, the is less than zero for an irreversible cyclic process and equal to zero for reversible cyclic process.
- Slides: 27