Seawater Chemical Properties Phases of Substances 2 The

Seawater Chemical Properties
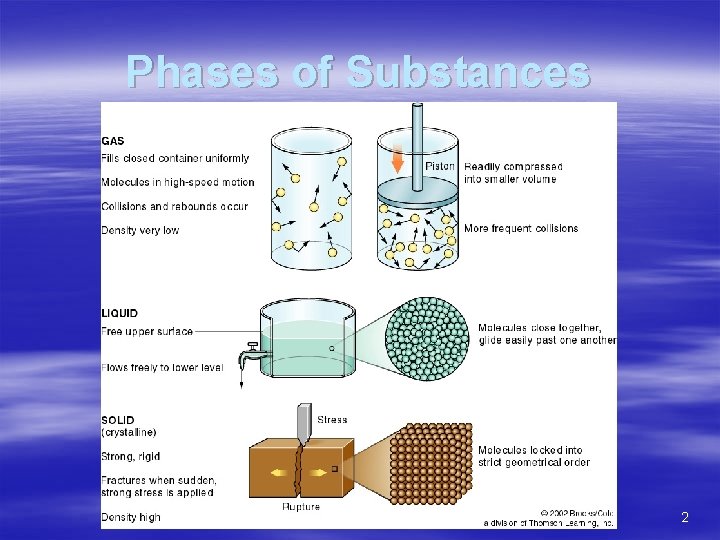
Phases of Substances 2
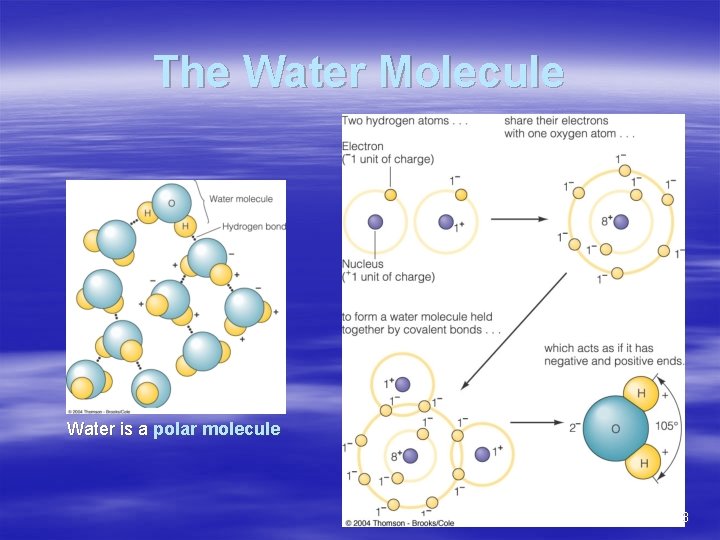
The Water Molecule Water is a polar molecule 3

Properties of Water Cohesion - the ability of water molecules to stick to each other, creating surface tension. Adhesion – the tendency of water molecules to stick to other substances Thus, water can act as a solvent 4

Properties of Water Heat is energy produced by the random vibrations of atoms or molecules. Temperature is an object’s response to input or removal of heat. Freezing point of water 0º C Boiling point of water 100º C 5
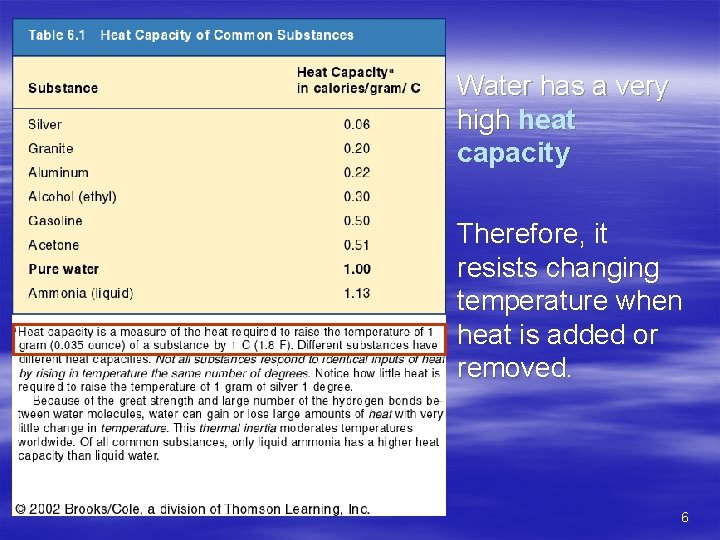
Water has a very high heat capacity Therefore, it resists changing temperature when heat is added or removed. 6

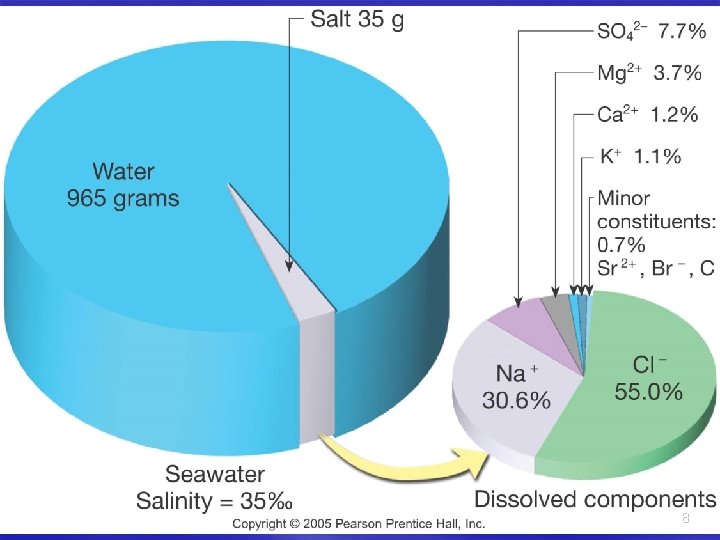
Salinity is the total amount of solid material dissolved in water Typically expressed as 0/00 or parts per thousand (ppt) 35 0/00 is world average 7
8

Salinity § Salt = cation + anion § Na. Cl = Na+ + Cl– Na+ has a positive charge (cation) – Cl- has a negative charge (anion) – SO 42 -, Mg 2+, HCO 3 -, Ca 2+, and K+ – Also present are trace elements 9
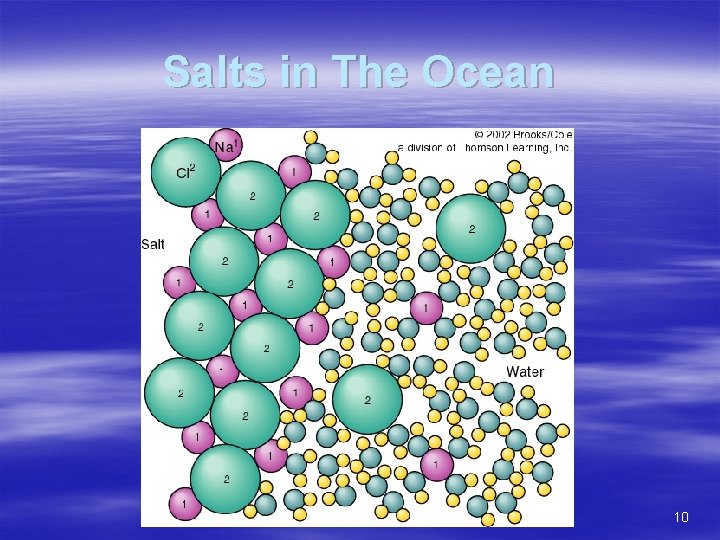
Salts in The Ocean 10
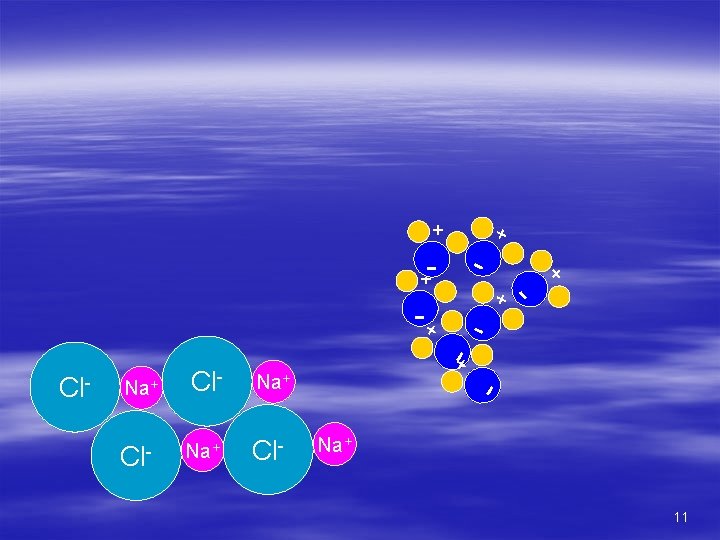

Is the ocean becoming progressively saltier with age? No! The ocean is in chemical equilibrium. The proportion and amounts of dissolved solids remain constant. This is known as the “steady state ocean. ” What goes in = what goes out 12

Salts going in must equal salts going out! § Salts come from: – Anions § Volcanic gasses – Cations § Minerals in rocks § Salts removed by: – Sea spray – Evaporites – Biologic processes § fecal pellets, shells – Magma at M. O. R. 13

Important Factors: Decrease Salinity Increase Salinity Precipitation Evaporation Run-off Freezing 14

The Hydrologic Cycle 15

Residence time 16

Salinity calculation S 0/00 = 1. 80655 x chlorinity in 0/00 Also use salinometer © S. Leyva 2004 17

Physical Factors That Affect O 2 and CO 2 Levels

Physical Factors Water Temperature Cold water holds more gas in solution than warm 19

Physical Factors Partial pressure of the gasses in the atmosphere The higher the pressure of these gasses in the atmosphere, the more gas is held in solution Deeper waters = more pressure = more gas in solution 20

Biological Factors Photosynthesis Consumes CO 2, produces O 2 + water Respiration Consumes O 2, produces CO 2 + water Decomposition (Organic matter decay) Consumes O 2, produces CO 2 + water Organic matter decays; releases CO 2 = biological pump 21
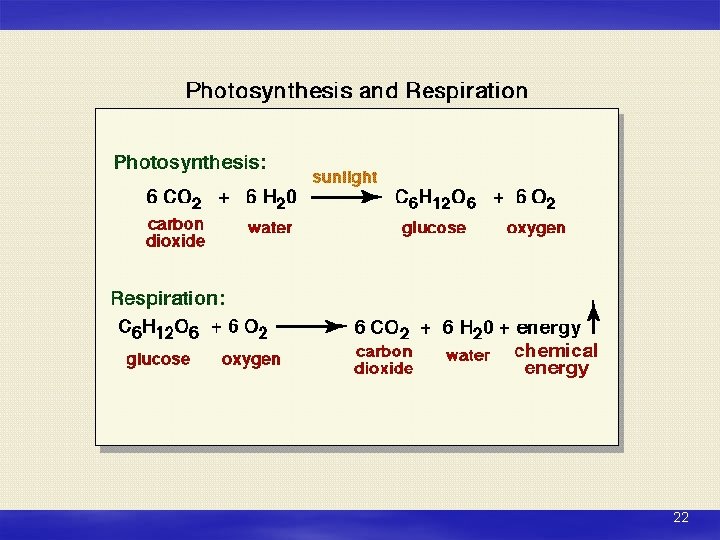
22

Where do the gasses come from? 1. From the atmosphere 2. Breaking waves / surf (aeration) 3. Volcanic eruptions 4. Phytoplankton 23

O 2 and CO 2 Levels in the Sea Photosynthesis ALWAYS occurs in the photic zone Respiration mostly occurs in the photic zone Decomposition occurs at the sea bed Therefore, O 2 levels should be high near the surface, and low near the sea floor, and the opposite for CO 2 levels 24

O 2 and CO 2 Levels in the Sea Lots of photosynthesis = lots of O 2 Time of day Highest at mid-afternoon, lowest just before dawn Season High in spring and summer, low in winter 25

Acidity Of Seawater An acid is a substance that releases a hydrogen ion in solution. A base is a substance that combines with a hydrogen ion in solution. Acidity or alkalinity is measured on the p. H scale. 26

Acidity Of Seawater p. H is measure of Hydrogen ions in water Higher p. H = more basic Lower p. H = more acidic More CO 2 = more acidic Expressed as numbers From 0 (acid) to 14 (base) Values for sea water are 7. 5 – 8. 4, 7. 8 is average 27

The p. H Scale 28 © 2002 Brooks/Cole, a division of Thomson Learning, Inc.

Acidity Of Seawater p. H related to CO 2 content Add CO 2 = more acidic Remove CO 2 = less acidic Respiration/organic matter decay = more CO 2 29
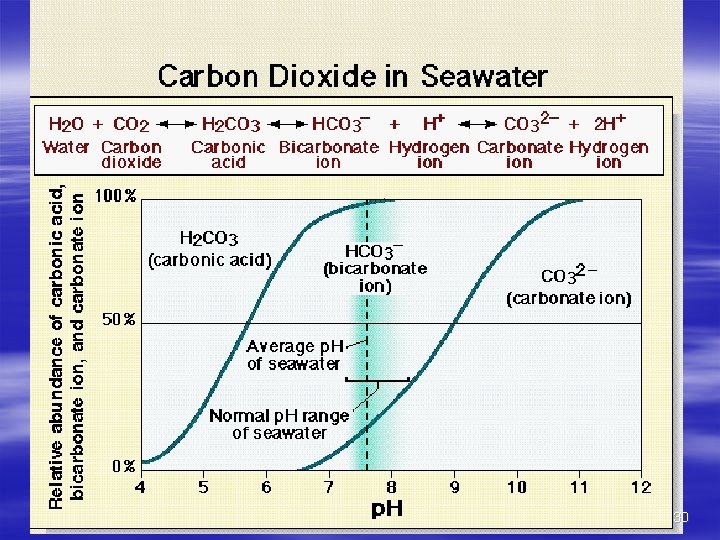
30

The carbonate buffering system: carbonic acid 31

Acidity Of Seawater Processes That Affect CO 2 Levels Increase CO 2 Decrease CO 2 § Partial pressure of CO 2 gas in the atmosphere § Animal respiration § Volcanic eruptions § Burning of fossil fuels, etc. § Photosynthesis, both on land in the sea § Consumption of CO 2 by corals, marine clams and snails 32
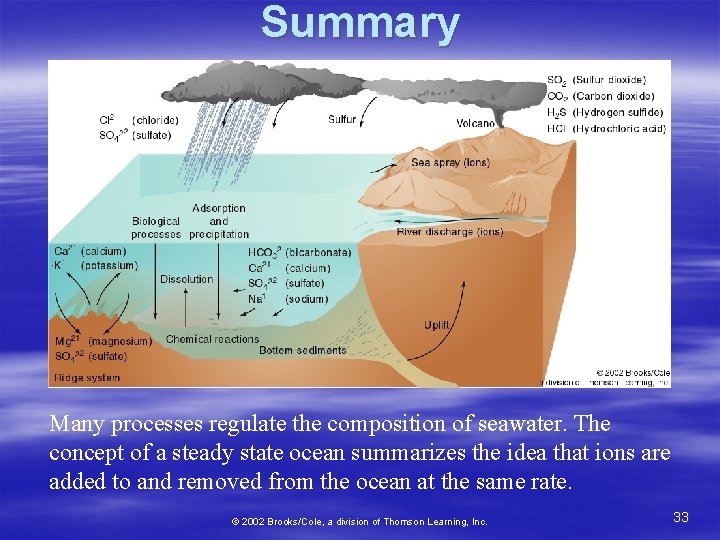
Summary Many processes regulate the composition of seawater. The concept of a steady state ocean summarizes the idea that ions are added to and removed from the ocean at the same rate. © 2002 Brooks/Cole, a division of Thomson Learning, Inc. 33

~ End ~
- Slides: 34