Seawater and Ocean Chemistry Seawater Chemistry n n














- Slides: 23

Seawater and Ocean Chemistry

Seawater Chemistry n n n Water Seawater Salts in seawater

Water n n Composition Properties

Water is … n a chemical compound (H 2 O) comprising two atoms of hydrogen and one atom of oxygen, in liquid state. . .

Ques The fusion of how many deuterium atoms would create tion: a carbon atom? nitrogen atom? oxygen atom?

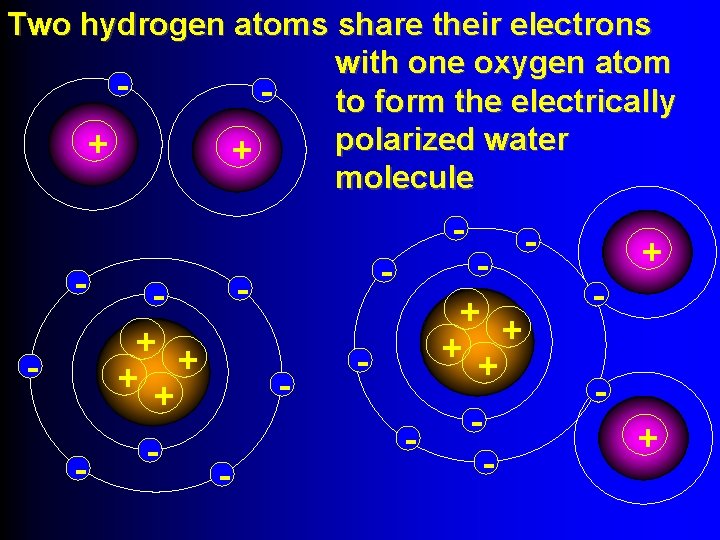
Two hydrogen atoms share their electrons with one oxygen atom to form the electrically polarized water + + molecule - - + + + + - - + +

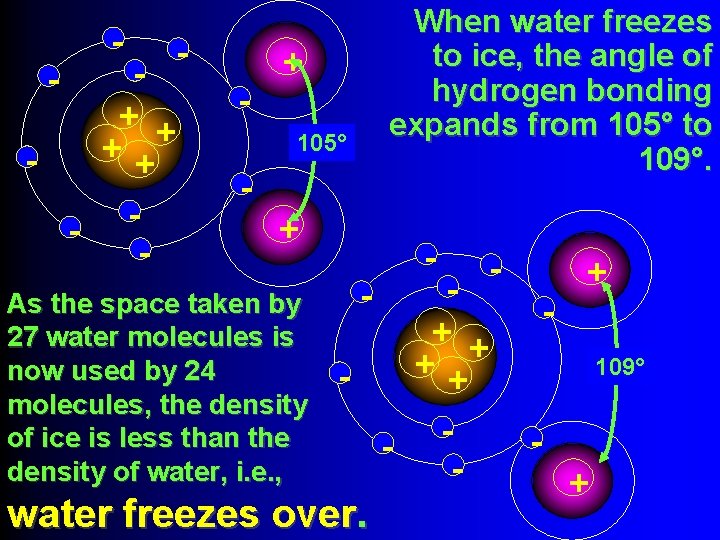
- - + + ++ - - When water freezes to ice, the angle of hydrogen bonding expands from 105° to 109°. + 105° + As the space taken by 27 water molecules is now used by 24 molecules, the density of ice is less than the density of water, i. e. , - - + + ++ - water freezes over. - + 109° +

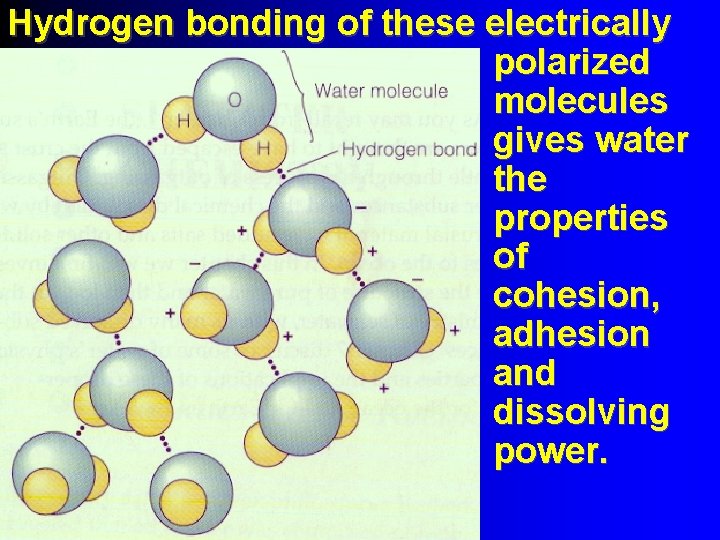
Hydrogen bonding of these electrically polarized molecules gives water the properties of cohesion, adhesion and dissolving power.

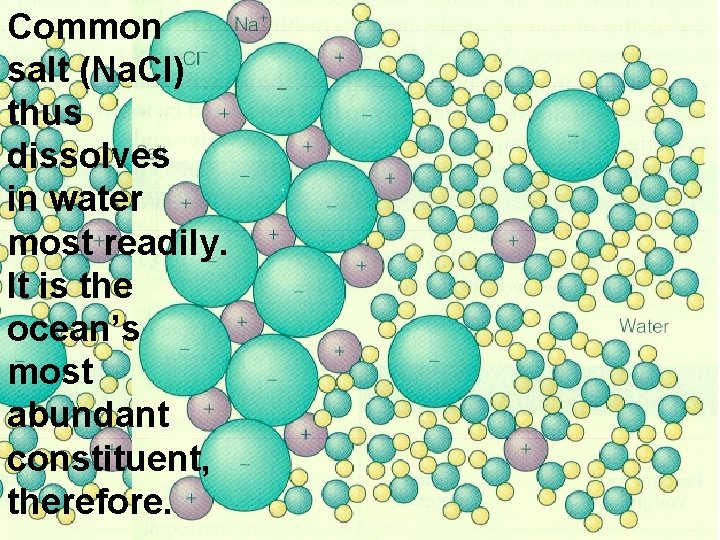
Common salt (Na. Cl) thus dissolves in water most readily. It is the ocean’s most abundant constituent, therefore.

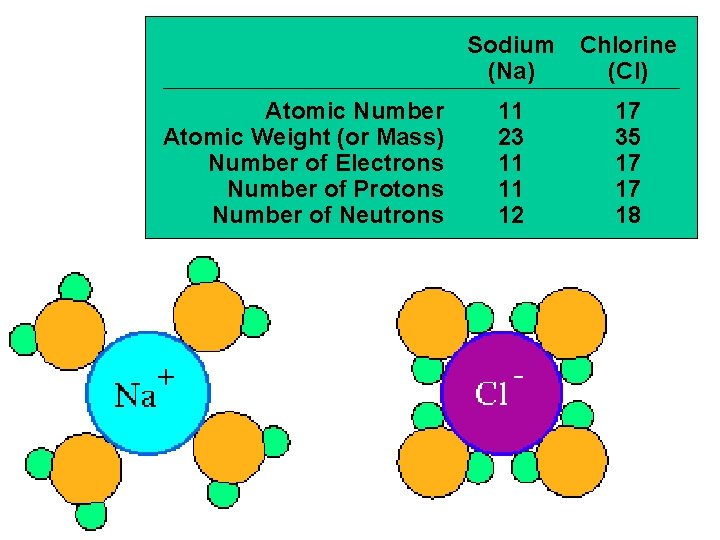
Atomic Number Atomic Weight (or Mass) Number of Electrons Number of Protons Number of Neutrons Sodium (Na) Chlorine (Cl) 11 23 11 11 12 17 35 17 17 18


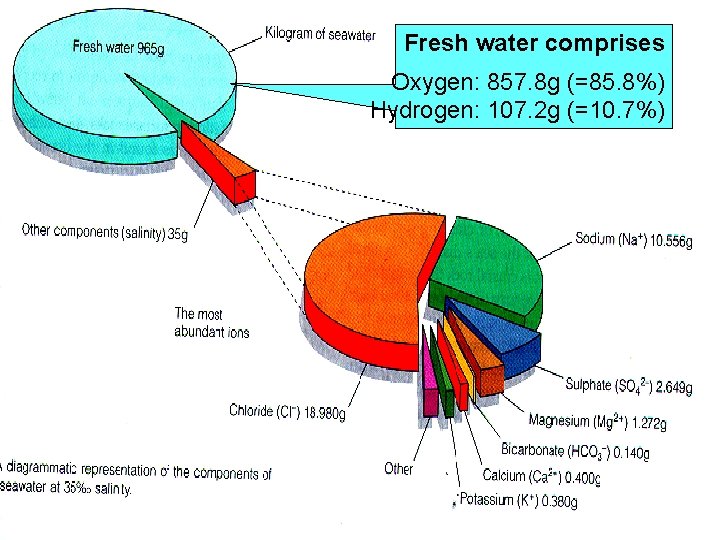
Fresh water comprises Oxygen: 857. 8 g (=85. 8%) Hydrogen: 107. 2 g (=10. 7%)

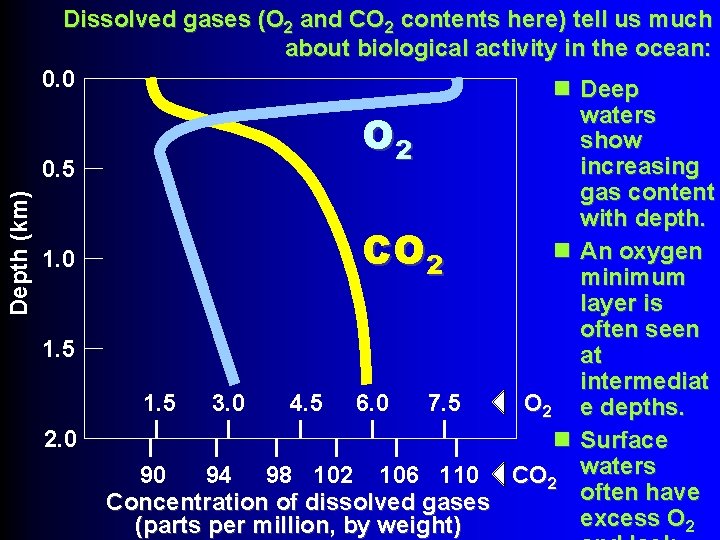
Dissolved gases (O 2 and CO 2 contents here) tell us much about biological activity in the ocean: 0. 0 n Deep waters show 2 increasing 0. 5 gas content with depth. n An oxygen 1. 0 2 minimum layer is often seen 1. 5 at intermediat 1. 5 3. 0 4. 5 6. 0 7. 5 O 2 e depths. 2. 0 n Surface 90 94 98 102 106 110 CO 2 waters often have Concentration of dissolved gases excess O 2 (parts per million, by weight) Depth (km) O CO

Most of carbon from the Earth’s atmosphere eventually ended up in the ocean % of gas, by Dissolved Gas volumeseawat in atmosph ere er Nitrogen (N 2) 78. 08% 48% Oxygen (O 2) 20. 95% 36% Carbon 0. 035% 15% dioxide (CO )

Major constituents of Seawater at 3. 5% Salinity Constituent Water: Oxygen (O) Hydrogen (H) 85. 8% 10. 7% The most abundant ions Chloride (Cl-) Sodium (Na+) Sulfate (SO 42 -) Magnesium (Mg 2+) Calcium (Ca 2+) Potassium (K+) Bicarbonate (HCO 3 -) 1. 9% 1. 1% 0. 3% 0. 1% 0. 04% 0. 01% only 2% of Cl in seawater could have come from land sources only 20% of sulfur in seawater could have come from land sources

Constant Proportions and Conservative Constituents 1. Some constituent salts in seawater occur in constant proportions, e. g. , Dissolved Salts in Seawater (‰) Chlorine in Seawater (‰) = 1. 80655 Thus, for average seawater with Cl = 19. 2‰, salinity = 1. 80655 × 19. 2‰ = 34. 7‰ 2. The constituents of seawater that occur in constant proportion (i. e. , change very slowly) are known as conservative constituents or elements.

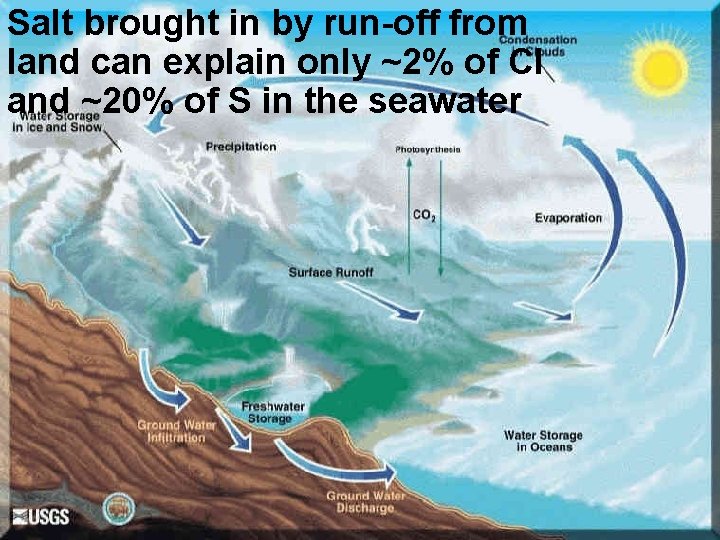
Salt brought in by run-off from land can explain only ~2% of Cl and ~20% of S in the seawater

Why is seawater salty? n Hydrothermal activity on the seafloor can provide the – sources for excess Cl and S and – sinks to balance the continued enrichment in sulfates and magnesium by streams. n We should also note that, compositionally, seawater has remained remarkably uniform through the

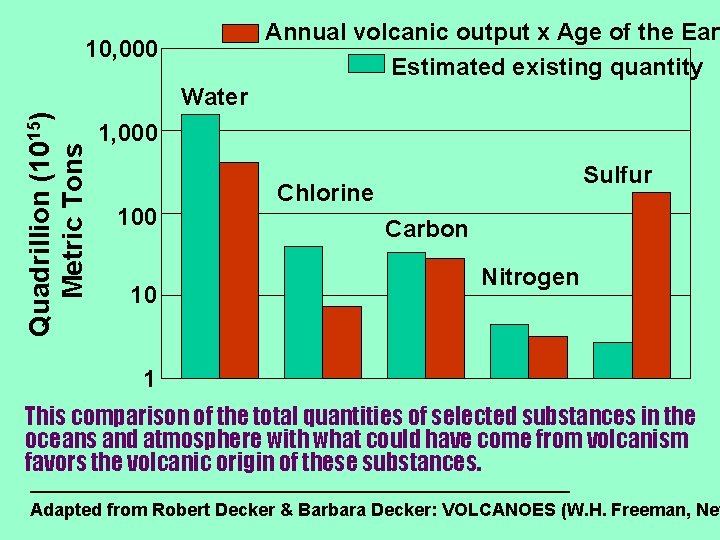
Annual volcanic output x Age of the Eart Estimated existing quantity 10, 000 Quadrillion (1015) Metric Tons Water 1, 000 10 Sulfur Chlorine Carbon Nitrogen 1 This comparison of the total quantities of selected substances in the oceans and atmosphere with what could have come from volcanism favors the volcanic origin of these substances. Adapted from Robert Decker & Barbara Decker: VOLCANOES (W. H. Freeman, New

The case for extraterrestrial origin of the oceans Deuterium n Comets are >40% water. Total Hydrogen ratio Comet Halley 0. 06 -0. 48 ppt n The deuterium/hydrogen ratio 0. 16 ppt of comets and oceans overlap. Earth’s Oceans n During the initial 2 Ga of its history, Earth may well have received 2 x 108 to 1 x 1017 metric tons of cometary matter by way of bombardment episodes. n Comets may well have contributed significantly, therefore, to the hydrospheric mass of 1. 4 -1. 7 x 1018 metric tons. Indeed, all this water could have been produced by either ~10% of the cometary mass or entirely by the asteroidal source if initial bombardment was of carbonaceous chondrites. Adapted from C. F. Chyba & C. Sagan in COMETS AND THE ORIGIN AND EVOLUTI (Ed: P. J. Thomas, C. F. Chyba & C. P. Mc. Kay; Springer-Verlag, New York, 1997).

A trio of frames from Polar's Visible Imaging System (VIS), taken 6 seconds apart last December 31 st, captures an object rapidly descending toward northern Europe. Because the camera's filter isolates emission from hydroxyl (OH) radicals, the incoming object must have

Suppose • annual influx from outer space is 50100 billion gallons of water vapor into the atmosphere, Compare this to the and that • this rate has been constant through geological history (~4. 5 billion years). This amounts to 225450 x 1018 gallons of water. total amount of water in the oceans = 1370 x 106 Km 3 (volume) � 109 m 3/Km 3 � 264. 2 gallons/m 3 = 362 � 1018 gallons of water

Therefore, there is no problem in accepting the extraterrestrial possibility, except that this requires • a much smaller ocean and • an appreciably weaker hydrological cycle in the past, than at the present. But there is no geological evidence to support this possibility.