SDSPAGE Sodium Dodecyl Sulfate Polyacrylamid Gel Electrophoresis Timothy









- Slides: 13

SDS-PAGE Sodium Dodecyl Sulfate Polyacrylamid Gel Electrophoresis Timothy G. Standish, Ph. D. © 1999 Timothy G. Standish

Sodium Dodecyl Sulfate � SDS is a common ingredient in detergents � Other names for SDS include lauryl sulfate and sodium lauryl sulfate � As a detergent SDS destroys protein secondary, tertiary and quaternary structure � This makes proteins rod shaped � SDS also sticks to proteins in a ratio of approximately 1. 4 g of SDS for each gram of protein � Negative charge on the sulfate groups of SDS mask any charge on the protein © 1999 Timothy G. Standish

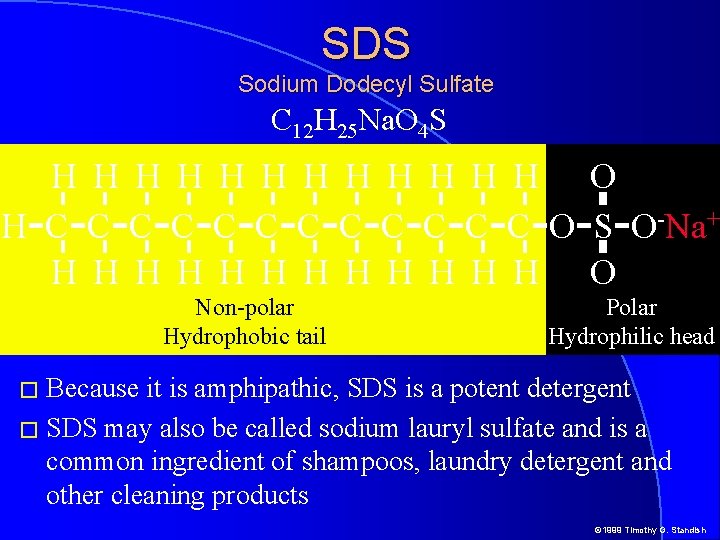
SDS Sodium Dodecyl Sulfate C 12 H 25 Na. O 4 S H H H O H-C-C-C-C-C-C-O-S-O-Na+ H H H O Non-polar Hydrophobic tail Polar Hydrophilic head � Because it is amphipathic, SDS is a potent detergent � SDS may also be called sodium lauryl sulfate and is a common ingredient of shampoos, laundry detergent and other cleaning products © 1999 Timothy G. Standish

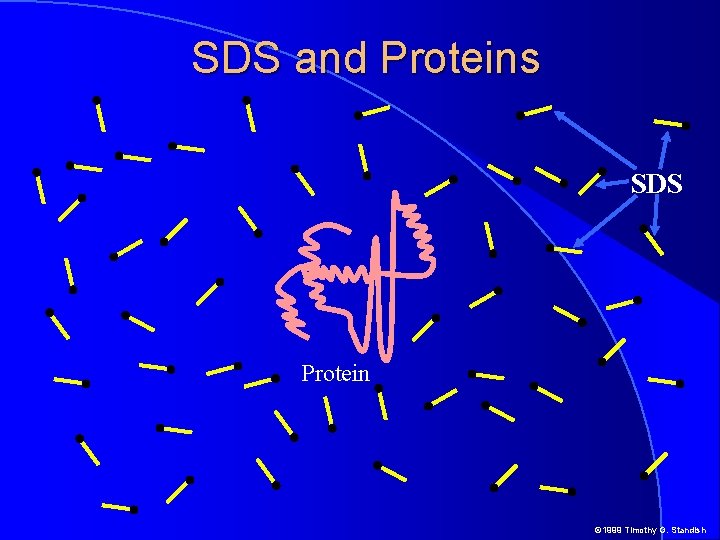
SDS and Proteins SDS Protein © 1999 Timothy G. Standish

SDS and Proteins � SDS nonpolar chains arrange themselves on proteins and destroy secondary tertiary and quarternary structrure � Thus shape is no longer an issue as the protein SDS complex becomes rod shaped � In aqueous solutions, SDS polarizes releasing Na+ and retaining a negative charge on the sulfate head � So much SDS binds to proteins that the negative charge on the SDS drowns out any net charge on protein side chains � In the presence of SDS all proteins have uniform shape and charge per unit length © 1999 Timothy G. Standish

Polyacrylamide Gels � Polyacrilamide is a polymer made of acrylamide (C 3 H 5 NO) and bis-acrilamide (N, N’-methylenebis-acrylamide C 7 H 10 N 2 O 2) CH 2 CH O C NH 2 CH Acrylamide NH 2 CH 2 O C CH 2 CH NH 2 Acrylamide bis-Acrylamide © 1999 Timothy G. Standish

Polyacrylamide Gels � Acrylamide polymerizes in the presence of free radicals typically supplied by ammonium persulfate O C CH 2 CH O NH 2 CH SO 4 -. © 1999 Timothy G. Standish

Polyacrylamide Gels Acrylamide polymerizes in the presence of free radicals typically supplied by ammonium persulfate � TMED (N, N, N’-tetramethylenediamine) serves as a catalyst in the reaction l O C CH 2 CH O NH 2 CH 2 CH SO 4 -. © 1999 Timothy G. Standish

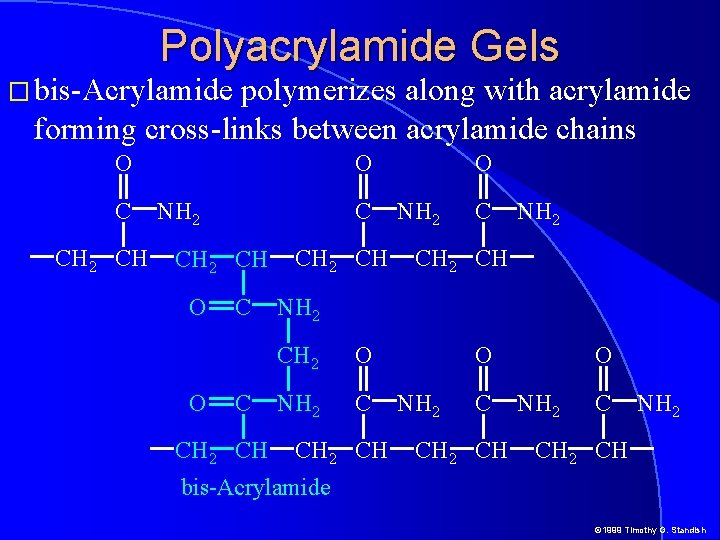
Polyacrylamide Gels � bis-Acrylamide polymerizes along with acrylamide forming cross-links between acrylamide chains O C CH 2 CH O NH 2 C CH 2 CH O O C C CH 2 CH O NH 2 CH NH 2 CH 2 O NH 2 C CH 2 CH O NH 2 CH bis-Acrylamide © 1999 Timothy G. Standish

Polyacrylamide Gels � bis-Acrylamide polymerizes along with acrylamide forming cross-links between acrylamide chains © 1999 Timothy G. Standish

� Pore Polyacrylamide Gels size in gels can be varied by varying the ratio of acrylamide to bis-acrylamide � Protein separations typically use a 29: 1 or 37. 5: 1 acrylamide to bis ratio Lots Littleofbis-acrylamide © 1999 Timothy G. Standish

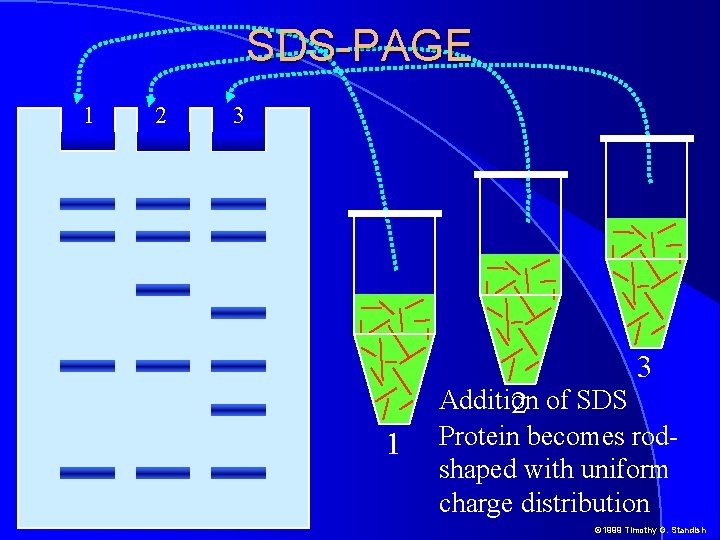
SDS-PAGE 1 2 3 3 1 Addition 2 of SDS Protein becomes rodshaped with uniform charge distribution © 1999 Timothy G. Standish

© 1999 Timothy G. Standish