SCREENING FOR CERVICAL CANCER THE OBSTETRICS GYNECOLOGICAL SOCIETY

SCREENING FOR CERVICAL CANCER THE OBSTETRICS & GYNECOLOGICAL SOCIETY OF BHOPAL & AMPOGS RESEARCH PUBLIC WELFARE SOCIETY
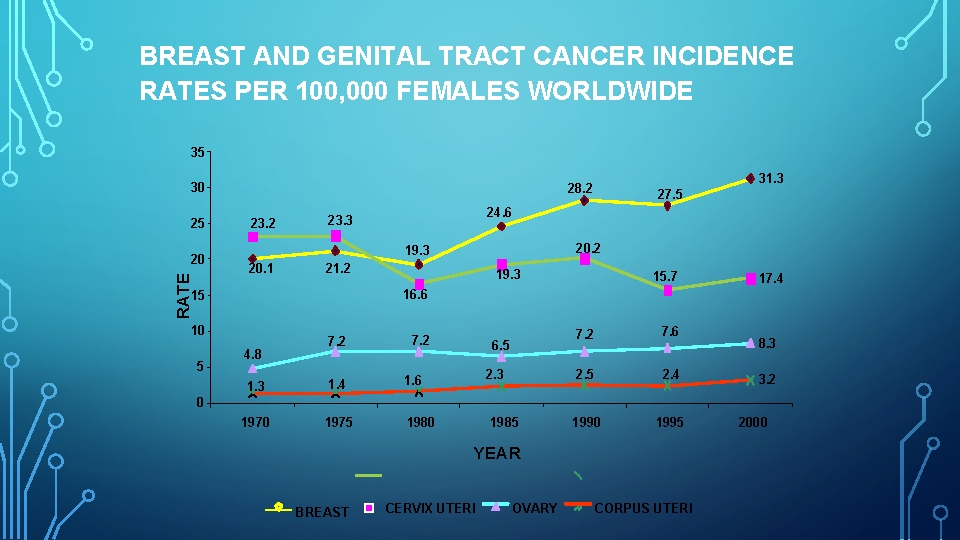
BREAST AND GENITAL TRACT CANCER INCIDENCE RATES PER 100, 000 FEMALES WORLDWIDE 35 30 25 RATE 20 23. 2 20. 2 19. 3 20. 1 27. 5 24. 6 23. 3 21. 2 19. 3 15. 7 17. 4 16. 6 15 10 5 31. 3 28. 2 4. 8 7. 2 1. 3 1. 4 1. 6 1970 1975 1980 6. 5 2. 3 7. 2 7. 6 2. 5 2. 4 1990 1995 8. 3 3. 2 0 1985 YEAR BREAST CERVIX UTERI OVARY CORPUS UTERI 2000
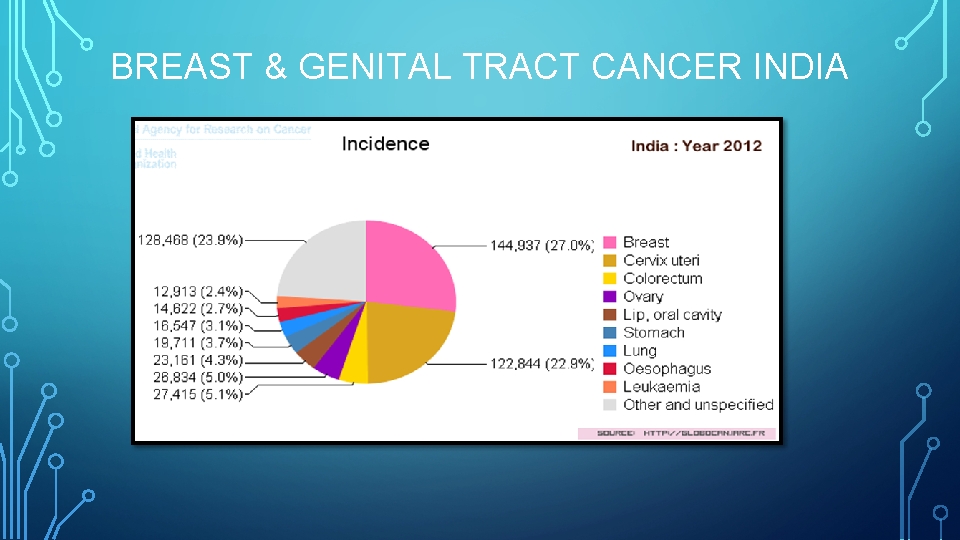
BREAST & GENITAL TRACT CANCER INDIA

CERVICAL CANCER INCIDENCE • • Cervical cancer is the fourth common cancer in women worldwide. • Cervical cancer is also preventable if precancerous lesions are detected in the early stages of development. Cervical cancer is also potentially curable with early detection and appropriate treatment. >80% women with cervical cancer live in low/middle income countries. > one fifth of these live in India. Cervical cancer is usually an asymptomatic slow growing cancer and can be detected easily in its early precancerous stages. If it is not detected at these early stages, there is an increased risk of progression of the disease to cervical cancer.

WHO NEEDS TO BE SCREENED • All sexually active/married women of 30 -60 years of age should be screened for cervical cancer except in the following situations: • • Menstruation Pregnancy Less than 6 weeks of delivery Had hysterectomy done • Screening to be done every 5 years
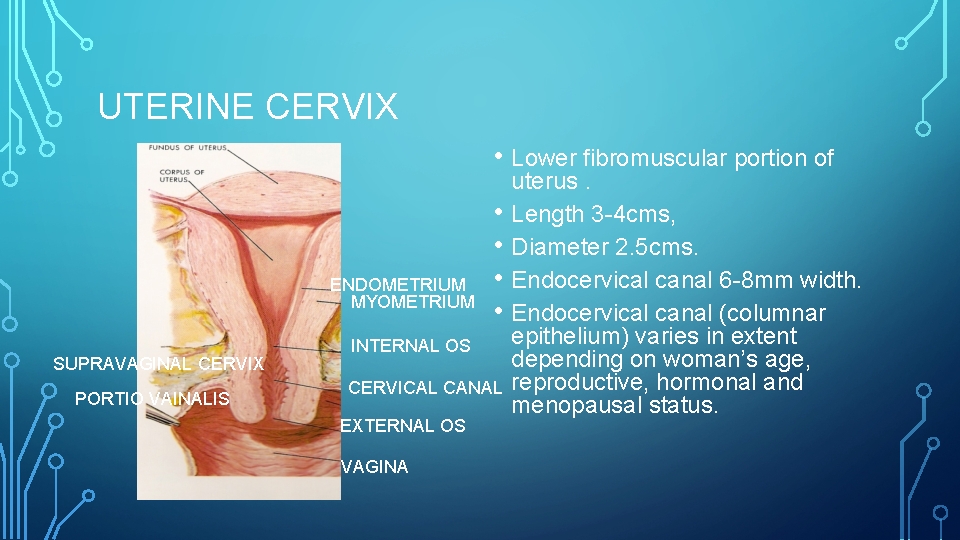
UTERINE CERVIX • Lower fibromuscular portion of ENDOMETRIUM MYOMETRIUM SUPRAVAGINAL CERVIX PORTIO VAINALIS • • INTERNAL OS CERVICAL CANAL EXTERNAL OS VAGINA uterus. Length 3 -4 cms, Diameter 2. 5 cms. Endocervical canal 6 -8 mm width. Endocervical canal (columnar epithelium) varies in extent depending on woman’s age, reproductive, hormonal and menopausal status.

UTERINE CERVIX • Stroma of cervix is composed of dense, fibromuscular tissue through which vascular, lymphatic, nerve supplies to cervix pass and form a complexus. • Ectocervix is relatively insensitive, while endocervix has extensive sensory nerve endings, both sympathetic and parasympathetic.
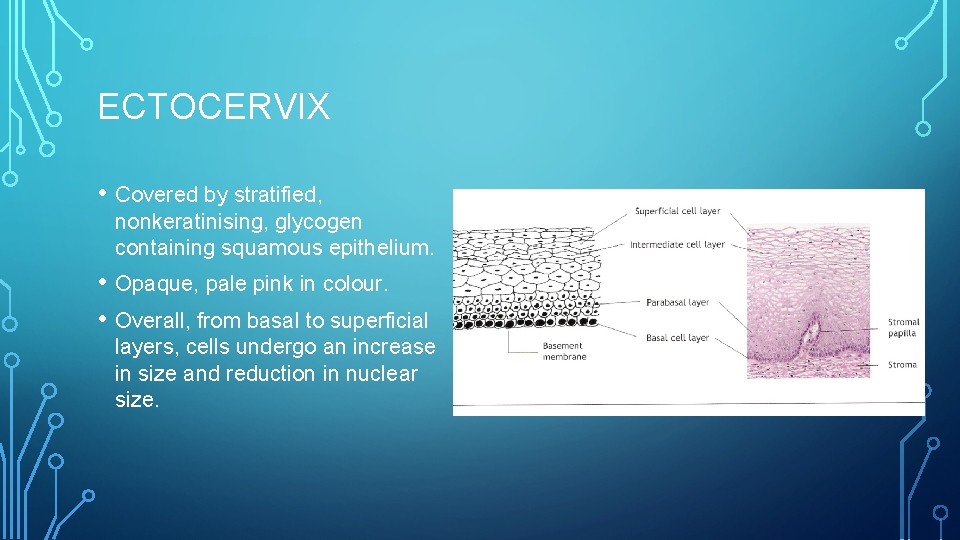
ECTOCERVIX • Covered by stratified, nonkeratinising, glycogen containing squamous epithelium. • Opaque, pale pink in colour. • Overall, from basal to superficial layers, cells undergo an increase in size and reduction in nuclear size.

ECTOCERVIX • Intermediate and superficial cells • • • contain abundant glycogen in their cytoplasm. Maturation of squamous epithelium dependant on estrogen. Terminal capillaries of subepithelial tissue arise to form capillary loops, just below and indenting the epithelium. It is these capillary loops that are seen on colposcopy
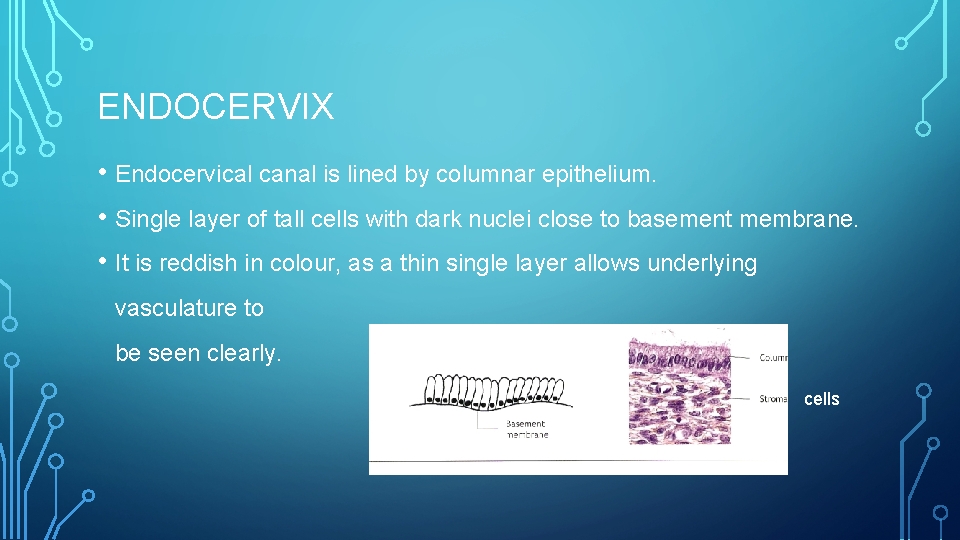
ENDOCERVIX • Endocervical canal is lined by columnar epithelium. • Single layer of tall cells with dark nuclei close to basement membrane. • It is reddish in colour, as a thin single layer allows underlying vasculature to be seen clearly. cells
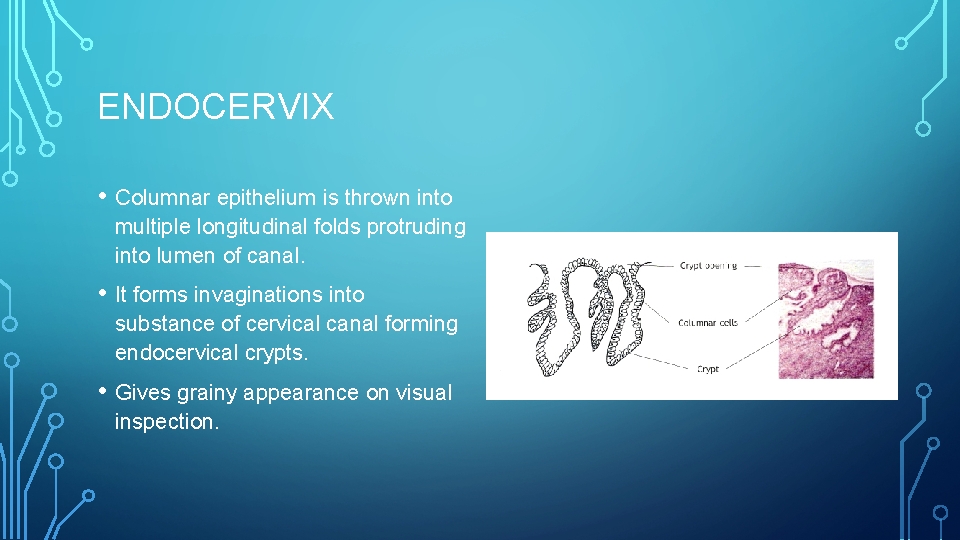
ENDOCERVIX • Columnar epithelium is thrown into multiple longitudinal folds protruding into lumen of canal. • It forms invaginations into substance of cervical canal forming endocervical crypts. • Gives grainy appearance on visual inspection.
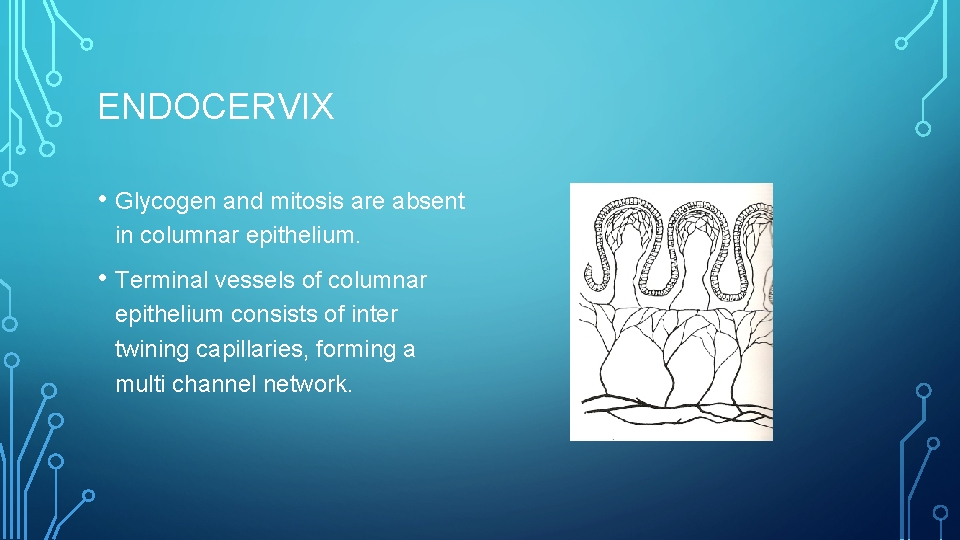
ENDOCERVIX • Glycogen and mitosis are absent in columnar epithelium. • Terminal vessels of columnar epithelium consists of inter twining capillaries, forming a multi channel network.
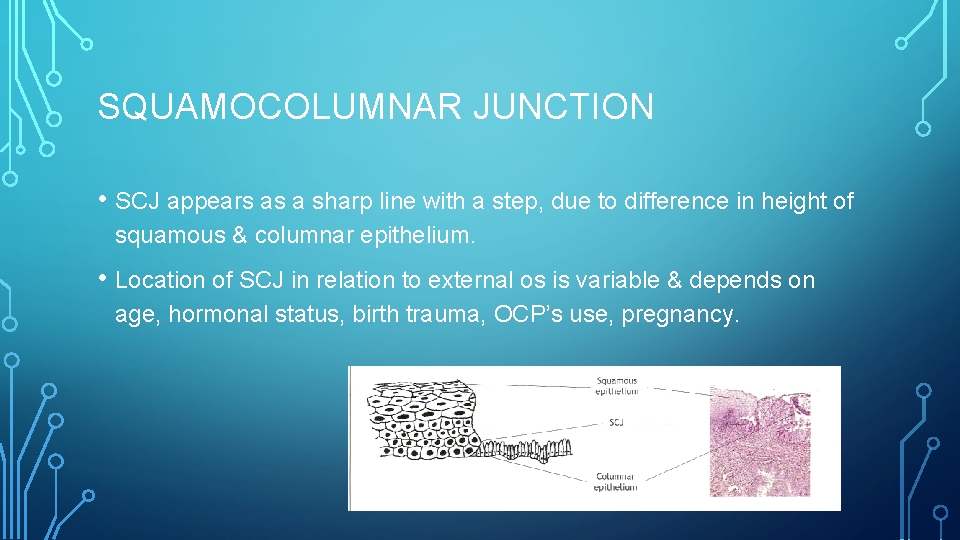
SQUAMOCOLUMNAR JUNCTION • SCJ appears as a sharp line with a step, due to difference in height of squamous & columnar epithelium. • Location of SCJ in relation to external os is variable & depends on age, hormonal status, birth trauma, OCP’s use, pregnancy.
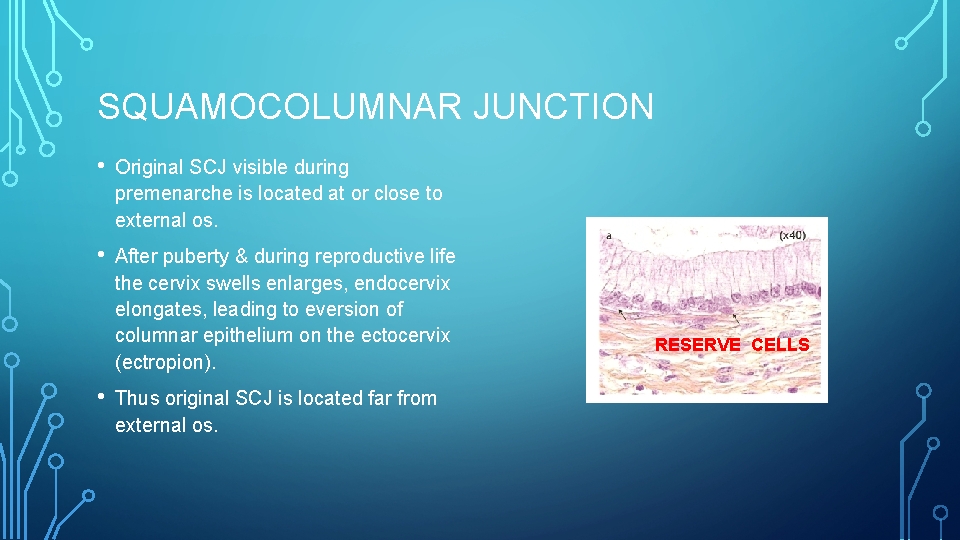
SQUAMOCOLUMNAR JUNCTION • Original SCJ visible during premenarche is located at or close to external os. • After puberty & during reproductive life the cervix swells enlarges, endocervix elongates, leading to eversion of columnar epithelium on the ectocervix (ectropion). • Thus original SCJ is located far from external os. RESERVE CELLS
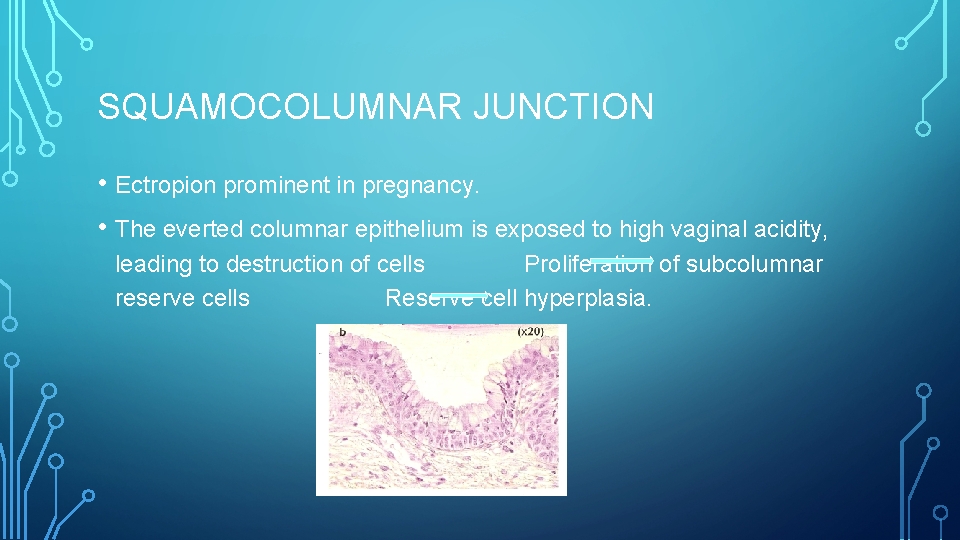
SQUAMOCOLUMNAR JUNCTION • Ectropion prominent in pregnancy. • The everted columnar epithelium is exposed to high vaginal acidity, leading to destruction of cells Proliferation of subcolumnar reserve cells Reserve cell hyperplasia.

SQUAMOCOLUMNAR JUNCTION Immature metastatic squamous epithelium. Normal glycogen containing mature squamous epithelium. Immature squamous epithelium
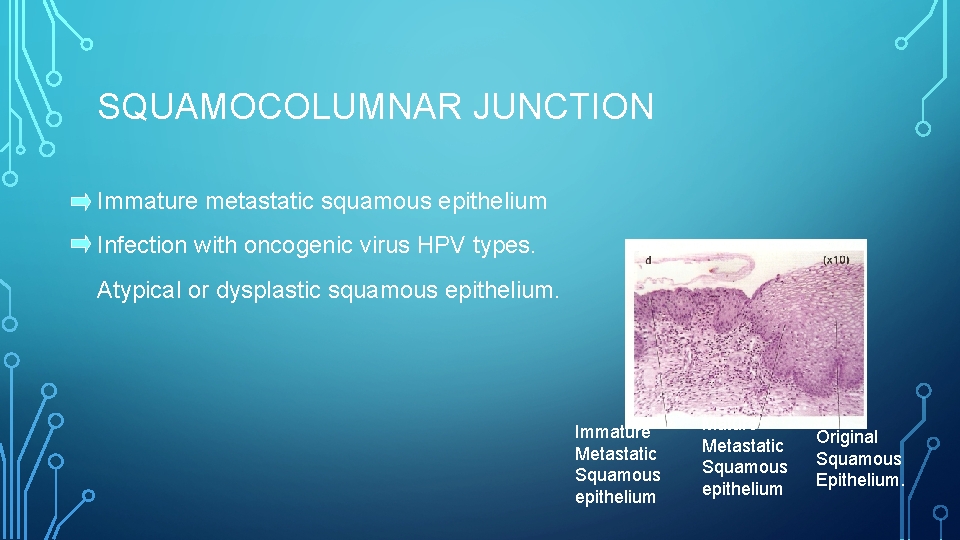
SQUAMOCOLUMNAR JUNCTION Immature metastatic squamous epithelium Infection with oncogenic virus HPV types. Atypical or dysplastic squamous epithelium. Immature Metastatic Squamous epithelium Mature Metastatic Squamous epithelium Original Squamous Epithelium.

TRANSFORMATION ZONE • Region of the cervix where columnar epithelium has been replaced and/or is being replaced by new metastatic squamous epithelium. • Its the area of cervix, bound by original SCJ at distally & metastatic SCJ proximally. • In premenopausal women the TZ is fully located on ectocervix. • After menopause cervix shrinks & TZ may move partially or fully into cervical canal.

TRANSFORMATION ZONE • • Normal TZ is composed of immature &/or mature squamous metaplasia along with intervening areas or islands of columnar epithelium with no signs of cervical carcinogenesis. Abnormal or atypical TZ evidence of cervical carcinogenesis such as dysplastic change. crypt TZ

HARALD ZUR HAUSEN
HUMAN PAPILLOMAVIRUS (HPV) • Double stranded circular DNA • • • molecule. Long known to cause warts Found in many cancers too Over 100 types identified Most benign, but 15 -20 can cause cancers Very common • • 20, 000 current cases in US 6, 200, 000 new cases annually 80% of women have HPV by age 50 50% of college students are infected

HPV & CERVICAL CANCER • HPV recognized as the underlying cause of cervical cancer since 1996 • NIH Consensus Conference on Cervical Cancer, 1996 • World Health Organization/European Research Organization on Genital Infection and Neoplasia, 1996
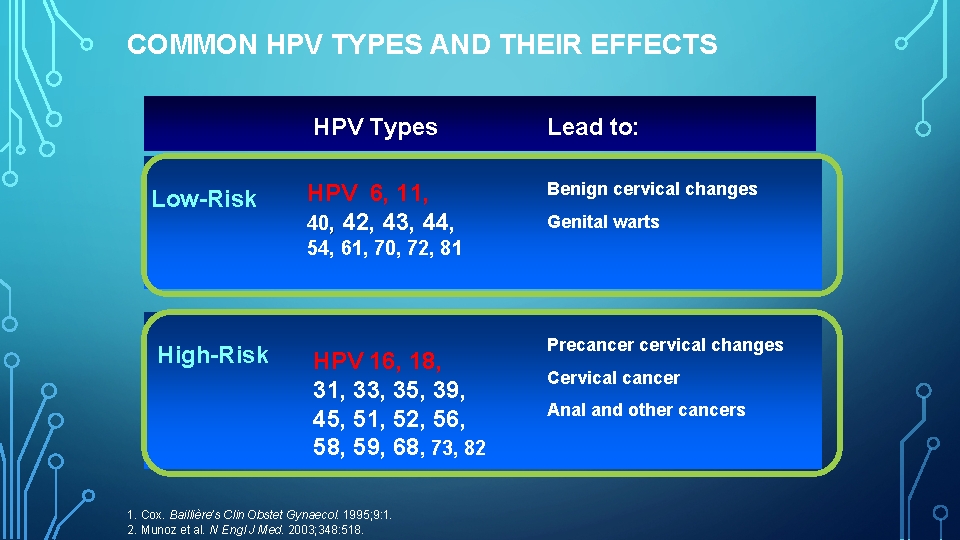
COMMON HPV TYPES AND THEIR EFFECTS HPV Types Low-Risk HPV 6, 11, 40, 42, 43, 44, Lead to: Benign cervical changes Genital warts 54, 61, 70, 72, 81 High-Risk HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, 82 1. Cox. Baillière’s Clin Obstet Gynaecol. 1995; 9: 1. 2. Munoz et al. N Engl J Med. 2003; 348: 518. Precancer cervical changes Cervical cancer Anal and other cancers
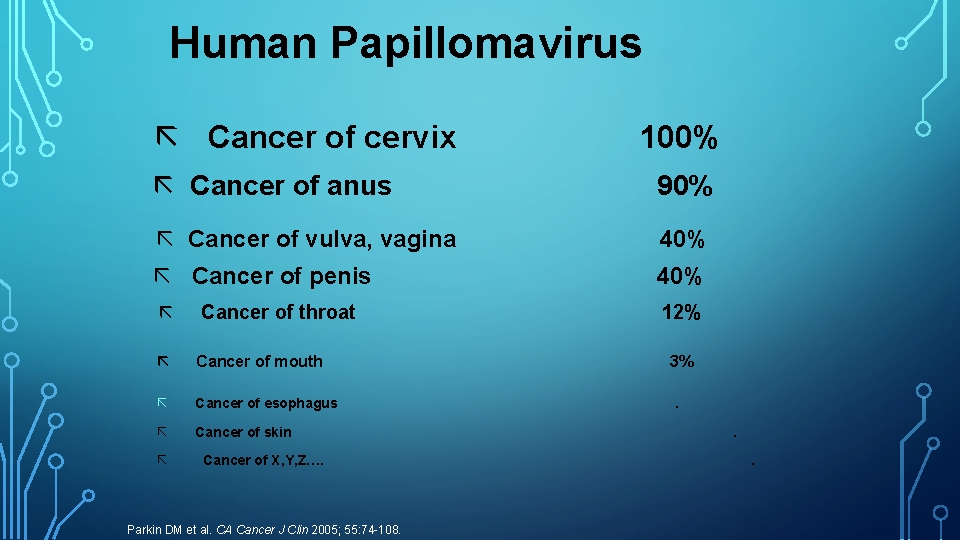
Human Papillomavirus ã Cancer of cervix 100% ã Cancer of anus 90% ã Cancer of vulva, vagina 40% ã Cancer of penis 40% ã 12% Cancer of throat ã Cancer of mouth ã Cancer of esophagus ã Cancer of skin ã Cancer of X, Y, Z…. Parkin DM et al. CA Cancer J Clin 2005; 55: 74 -108. 3%. . .

PATHOGENESIS l There are 3 steps necessary for cervical cancer development 1. HPV infection 2. Progression to cervical intraepithelial neoplasia (CIN) 3. Invasion HPV infection must persist for more than one year Normal Cervix HPV Infection HPV Clearance Infected Cervix Progression Invasion CIN Regression Cancer
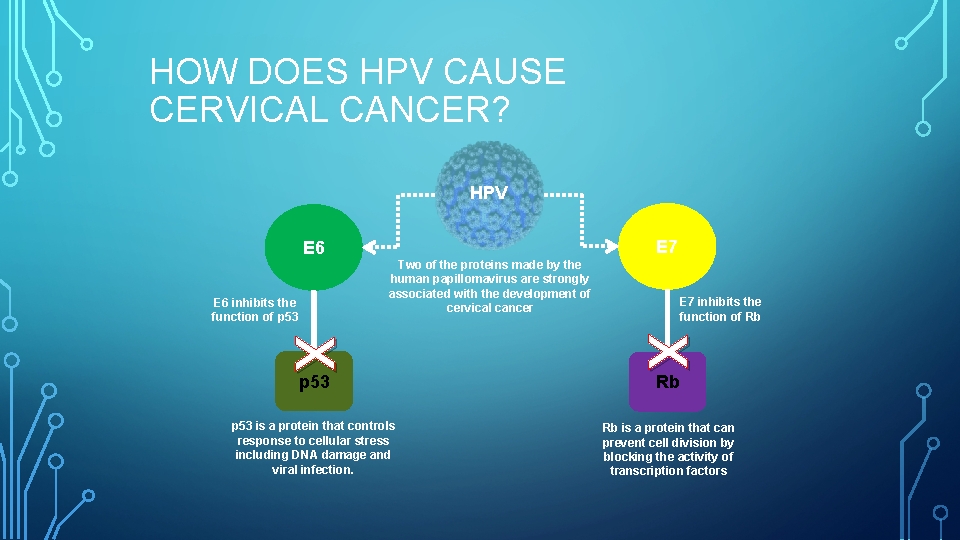
HOW DOES HPV CAUSE CERVICAL CANCER? HPV E 7 E 6 Two of the proteins made by the human papillomavirus are strongly associated with the development of cervical cancer E 6 inhibits the function of p 53 E 7 inhibits the function of Rb X X p 53 is a protein that controls response to cellular stress including DNA damage and viral infection. Rb is a protein that can prevent cell division by blocking the activity of transcription factors p 53 Rb
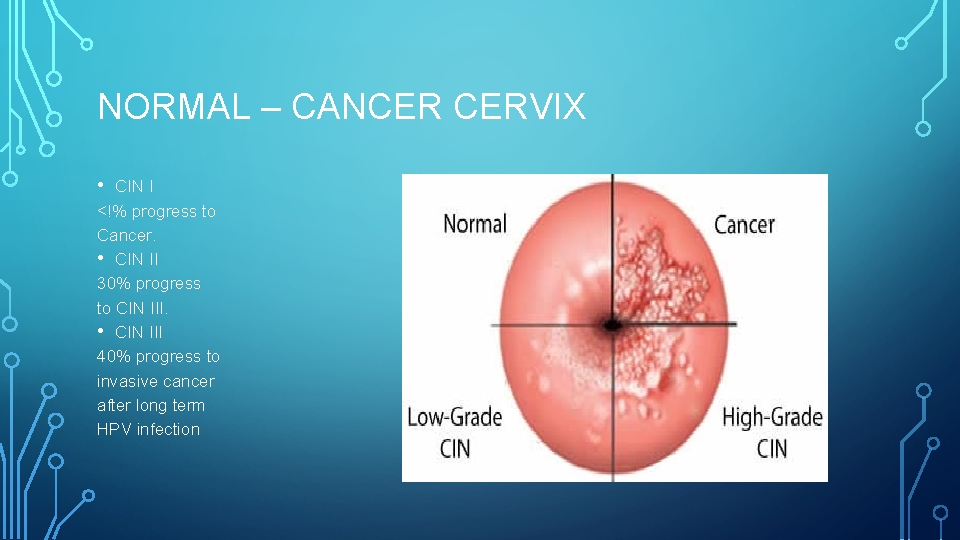
NORMAL – CANCER CERVIX • CIN I <!% progress to Cancer. • CIN II 30% progress to CIN III. • CIN III 40% progress to invasive cancer after long term HPV infection
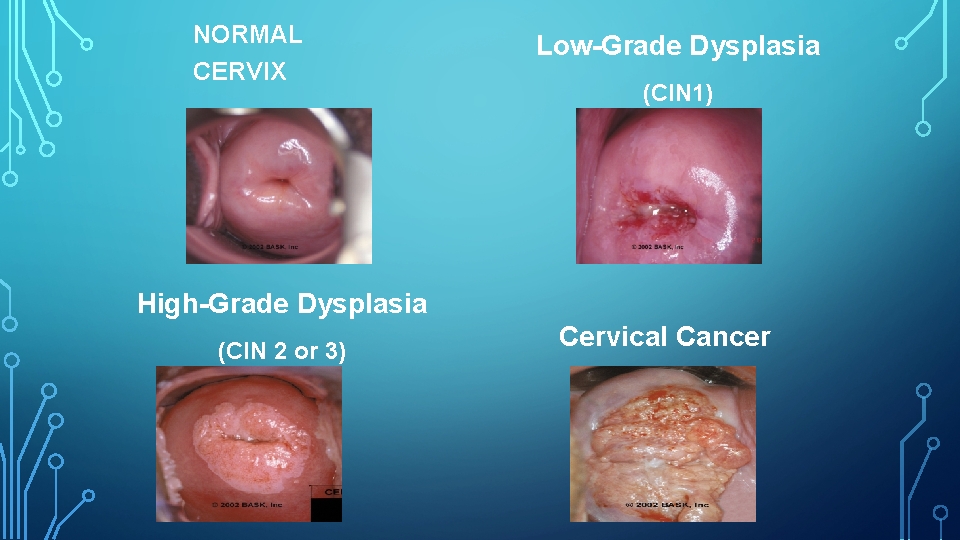
NORMAL CERVIX Low-Grade Dysplasia (CIN 1) High-Grade Dysplasia (CIN 2 or 3) Cervical Cancer
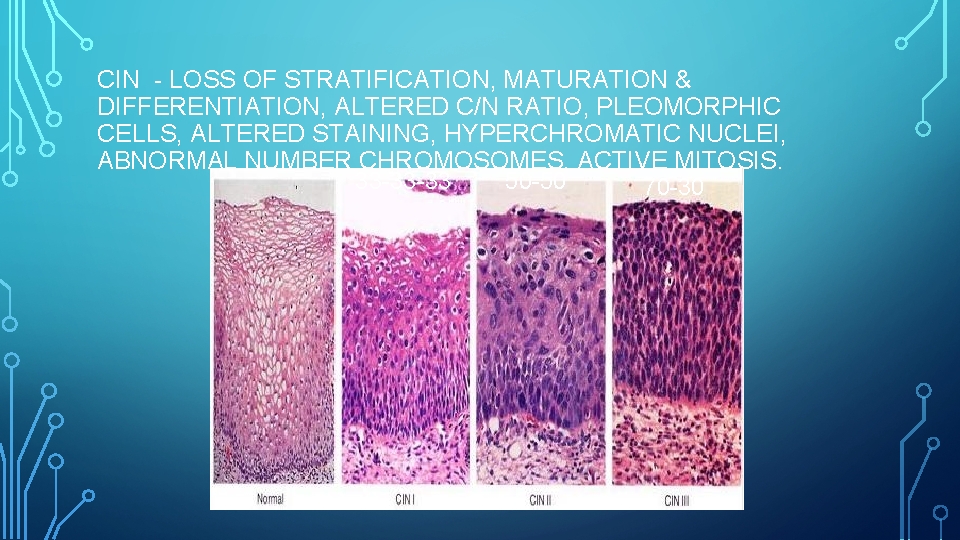
CIN - LOSS OF STRATIFICATION, MATURATION & DIFFERENTIATION, ALTERED C/N RATIO, PLEOMORPHIC CELLS, ALTERED STAINING, HYPERCHROMATIC NUCLEI, ABNORMAL NUMBER CHROMOSOMES, ACTIVE MITOSIS. 33 -33 -33 50 -50 70 -30

DIAGNOSIS OF CIN • Symptomless, excessive discharge, postcoital bleeding. • Pap’s smear - >21 yrs or sexually active and 3 yearly thereafter. • HPV test – for >30 yrs, after Pap’s. • Colposcopy - if Pap’s or HPV test positive, look for acetowhite areas, mosaic, punctation or abnormal vessels.
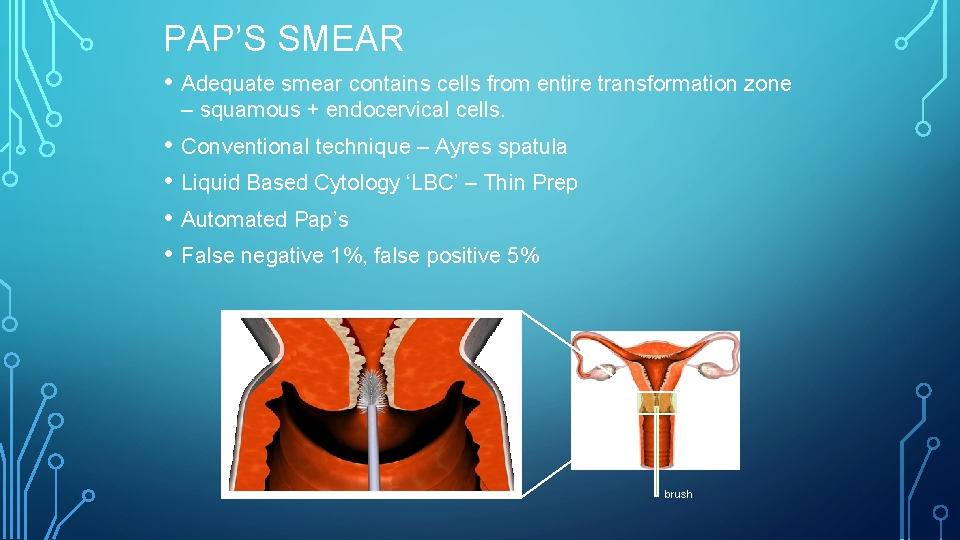
PAP’S SMEAR • Adequate smear contains cells from entire transformation zone – squamous + endocervical cells. • Conventional technique – Ayres spatula • Liquid Based Cytology ‘LBC’ – Thin Prep • Automated Pap’s • False negative 1%, false positive 5% brush

PAPS SMEAR Normal squamous cells Dysplastic cells Sensitivity – True Positive Specificity – True Negative
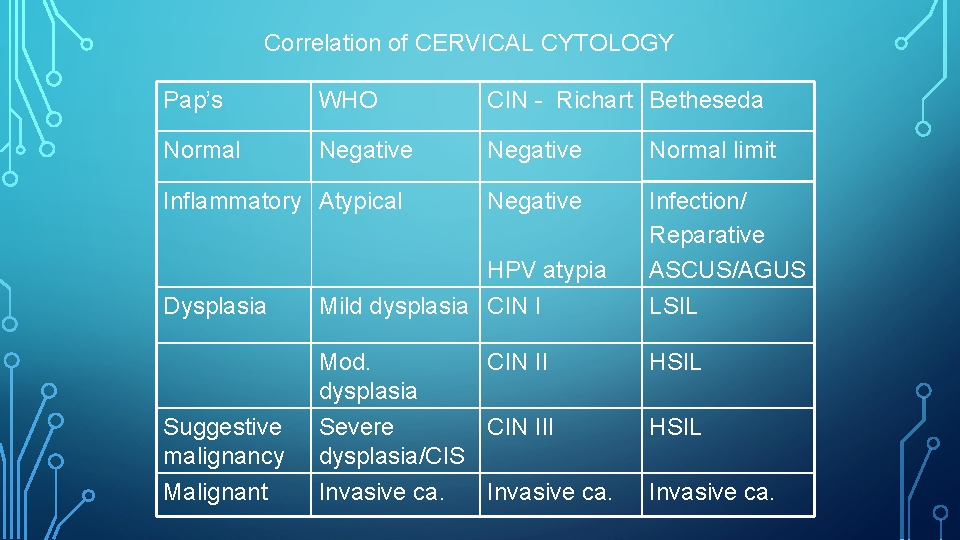
Correlation of CERVICAL CYTOLOGY Pap’s WHO CIN - Richart Betheseda Normal Negative Normal limit Negative Infection/ Reparative ASCUS/AGUS Inflammatory Atypical HPV atypia Dysplasia Mild dysplasia CIN I LSIL HSIL Suggestive malignancy Mod. CIN II dysplasia Severe CIN III dysplasia/CIS Malignant Invasive ca. HSIL
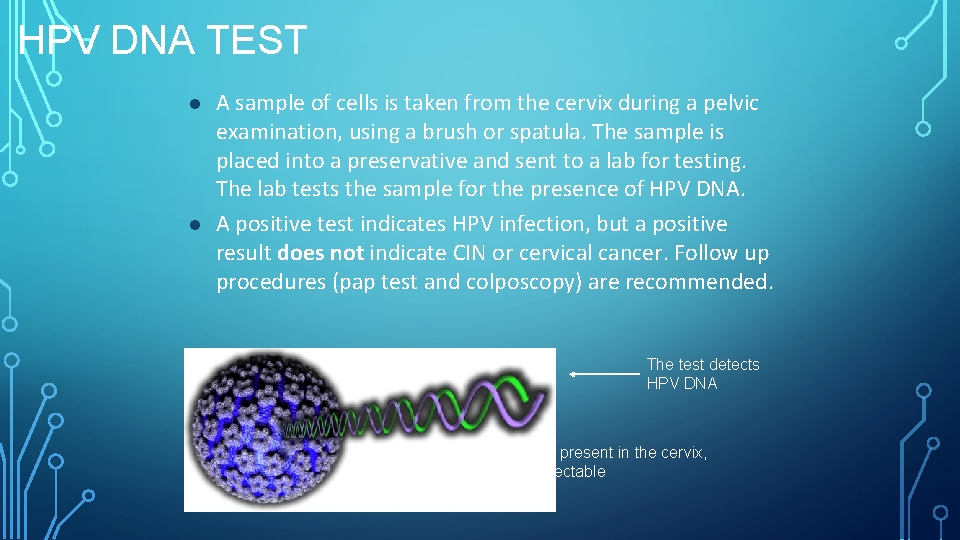
HPV DNA TEST l l A sample of cells is taken from the cervix during a pelvic examination, using a brush or spatula. The sample is placed into a preservative and sent to a lab for testing. The lab tests the sample for the presence of HPV DNA. A positive test indicates HPV infection, but a positive result does not indicate CIN or cervical cancer. Follow up procedures (pap test and colposcopy) are recommended. The test detects HPV DNA If an HPV infection is present in the cervix, HPV DNA will be detectable

HPV TESTS • Digene HPV DNA Hybrid Capture 2 test. It tests for 13 high risk and 5 low risk types of HPV*. • HPV genotyping • HPV Oncotect: HPV E 6, E 7 m. RNA assay– identifies presence of HPV E 6, E 7 m. RNA (viral gene expression) in intact epithelial cells. ** • Onco. E 6 TM cervical test – Arbor Vita.

COLPOSCOPE • • Introduced by Hans Hinselmann in 1925. • Study of surface epithelium of cervix and underlying tissue stroma along with its vascular network. Optical method for visualizing lower female genital tract under bright illumination and magnification.

INDICATIONS • Evaluation of women with abnormal Pap’s smear, positive VIA or VILI, • • • to localize or select punch biopsy site. Evaluation of women with normal smear but suspicious cervix, post coital bleeding or leucoplakia. Persistent vaginal discharge. Subclinical HPV infection. Conservative Rx of CIN Follow up after Rx of CIN III or invasive cancer.

COLPOSCOPE • Low power, stereoscopic, binocular, field microscope with powerful light source. • Magnification 6 x-40 x. • Other instruments. Bivalve speculum, sponge forceps, endocervical speculum & curette, punch biopsy forceps.
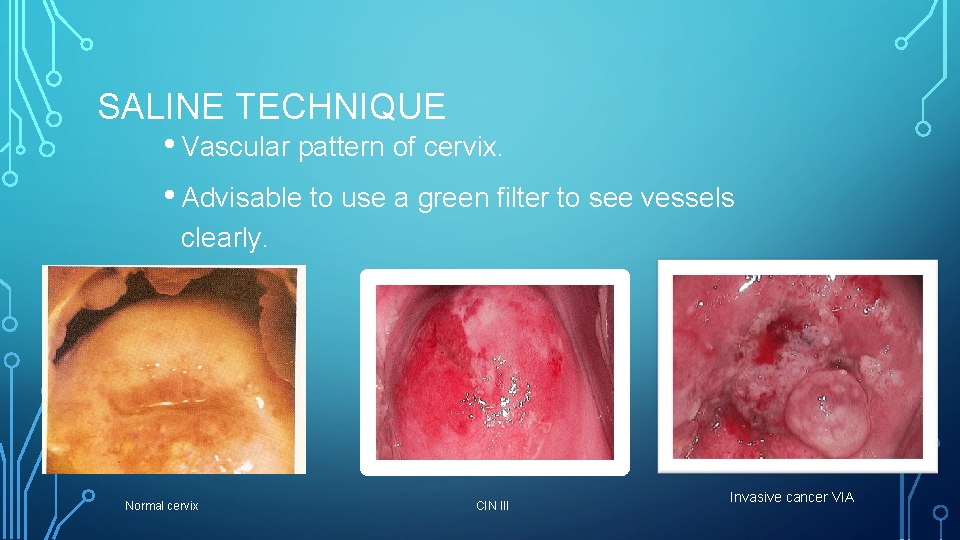
SALINE TECHNIQUE • Vascular pattern of cervix. • Advisable to use a green filter to see vessels clearly. Normal cervix CIN III Invasive cancer VIA
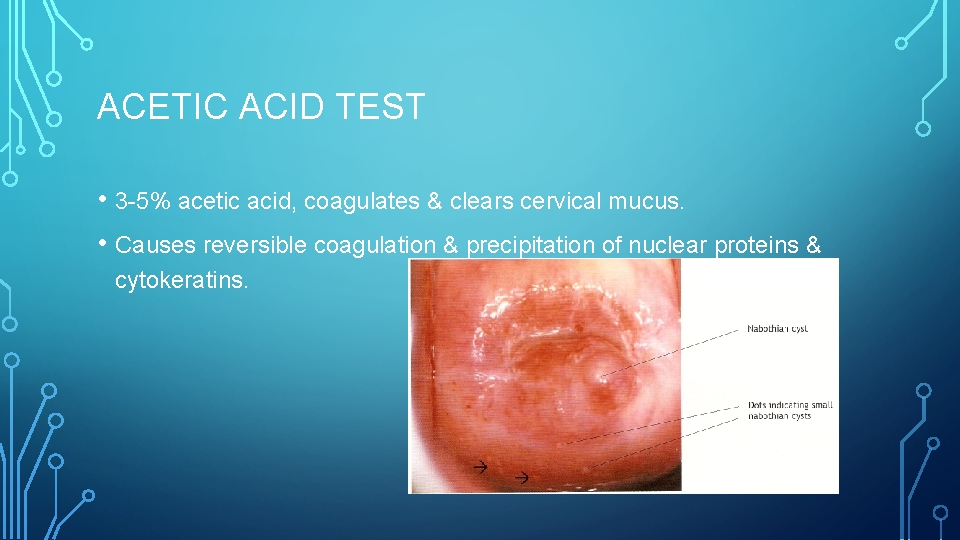
ACETIC ACID TEST • 3 -5% acetic acid, coagulates & clears cervical mucus. • Causes reversible coagulation & precipitation of nuclear proteins & cytokeratins.

ACETIC ACID TEST • Normal squamous epithelium, little coagulation occurs as superficial • epithelium is sparsely nucleated. Areas of CIN undergo maximal coagulation due to high content of nuclear protein & prevent light from passing through epithelium. Therefore subepithelial vessel pattern is obliterated & epithelium appears “acetowhite”.
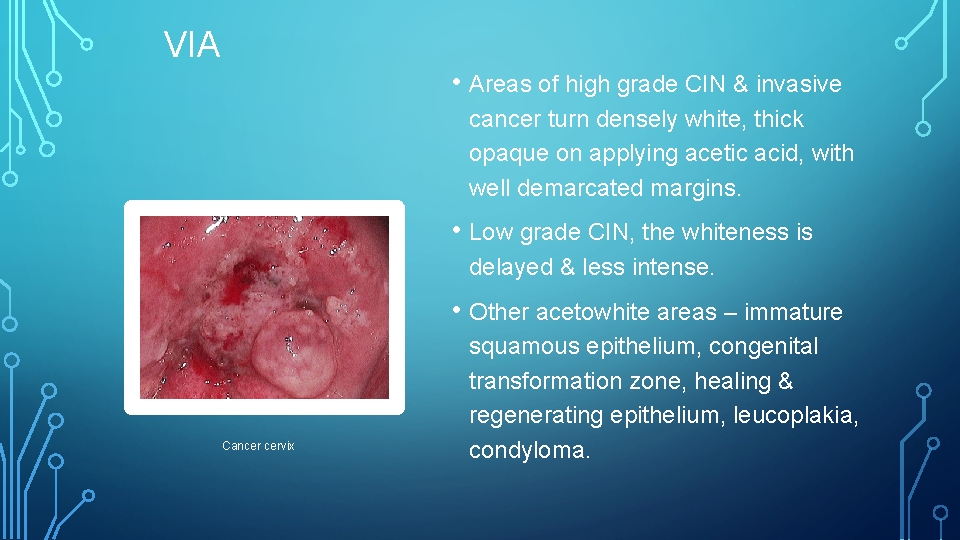
VIA • Areas of high grade CIN & invasive cancer turn densely white, thick opaque on applying acetic acid, with well demarcated margins. • Low grade CIN, the whiteness is delayed & less intense. • Other acetowhite areas – immature Cancer cervix squamous epithelium, congenital transformation zone, healing & regenerating epithelium, leucoplakia, condyloma.

VIA PROCEDURE • • Ask the woman is to lie down in a modified lithotomy position with leg rests or stirrups. • • • Introduce a sterile vaginal speculum and open blades of the speculum to view the cervix. • • Proceed to identify the transformation zone, the upper limit of which is formed by the SCJ. Observe the external genitalia and perineal region for any signs of excoriations, edema, vesicles, papules, sores, ulceration and warts. Look for any swelling in the inguinal/femoral region. Look for any vaginal discharge & observe the size and shape of the cervix. Identify the external os, columnar epithelium (red in colour), squamous epithelium (pink) and the squamocolumnar junction. Look for ectropion, cervical polyp, nabothian cysts, healed laceration of the cervical lips, leukoplakia, condylomata and signs of cervicitis. In postmenopausal women, the cervix appears pale and brittle, due to thinning and atrophy of the squamous epithelium.

INTERPRETATION • Discharge: Assess the characteristics of discharge in terms of quantity, colour, odour and thickness. Thread-like, thin mucinous discharge from the external os indicates ovulation. • Menstrual Bleeding: If heavy blood flow through the external os is observed in women during menstruation, subject to VIA after 5 -15 days. • Ectropion the cervix has a large area of red appearance around the external os and the squamocolumnar junction far away from the os. Extensive erosive red areas may be present on the cervix, extending to the vagina in instances of severe cervical infection and inflammation. • Nabothian cysts appear as bulging blue-white or yellow-white nodules, having a smooth delicate lining with branching blood vessels. • Leukoplakia appears as a smooth-surfaced, white area on the cervix before doing VIA that cannot be removed or scraped off.

INTERPRETATION • Observe whethere is any bleeding from the cervix, especially on touch, or ulcer or proliferative growth. More advanced invasive cancers may present as a large exophytic growth with an ulceroproliferative, bulging mass with polypoid or papillary excrescences, arising from the cervix or as a predominantly ulcerating growth replacing most of the cervix. • If no evidence of invasive cancer: Gently, but firmly, apply 5% acetic acid using a cotton swab soaked in acetic acid. Wait for at least 1 minute before reading. The secretions should be gently wiped off. • A significant white lesion is defined as a lesion that appears white, thick and with well-defined margins close to or at the SCJ. The results one minute after application of acetic acid should be reported. Note how rapidly the acetowhite lesion appears and then disappears.

• Carefully observe: • The intensity of the white colour of the acetowhite lesion: whether it is shiny white, cloudy-white, pale-white or dull-white. • The borders and demarcations of the white lesion: distinctly clear and sharp or indistinct diffuse margins; raised or flat margins; regular or irregular margins. • Whether the lesions are uniformly white in colour, or the colour intensity varies across the lesion, or if there areas of erosion within the lesion. • Location of the lesion: is it in, near or far away from the transformation zone? Is it abutting (touching) the squamocolumnar junction? Does it extend into the endocervical canal? Does it occupy the entire or part of the transformation zone? Does it involve the entire cervix (which usually indicates early preclinical invasive cancer)? • Size (extent or dimensions) and number of the lesions.
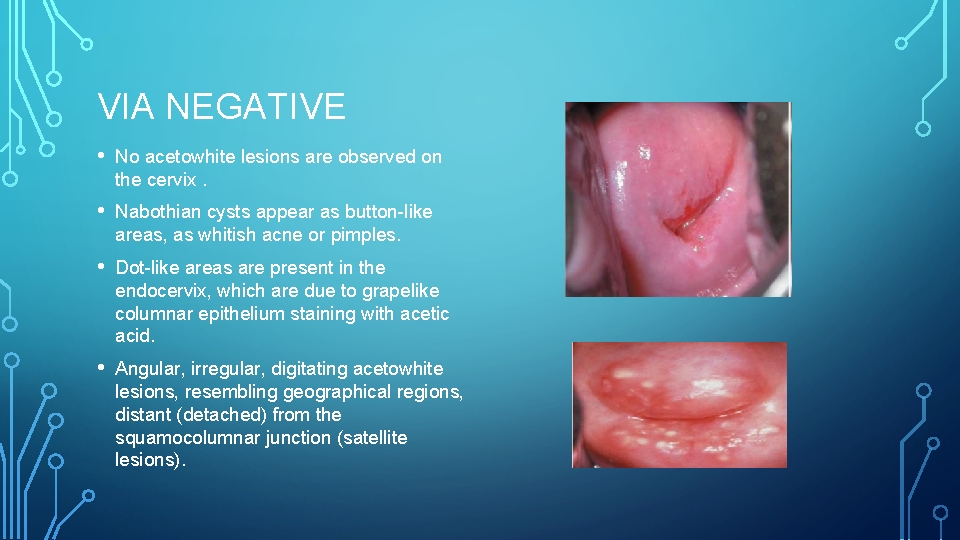
VIA NEGATIVE • No acetowhite lesions are observed on the cervix. • Nabothian cysts appear as button-like areas, as whitish acne or pimples. • Dot-like areas are present in the endocervix, which are due to grapelike columnar epithelium staining with acetic acid. • Angular, irregular, digitating acetowhite lesions, resembling geographical regions, distant (detached) from the squamocolumnar junction (satellite lesions).
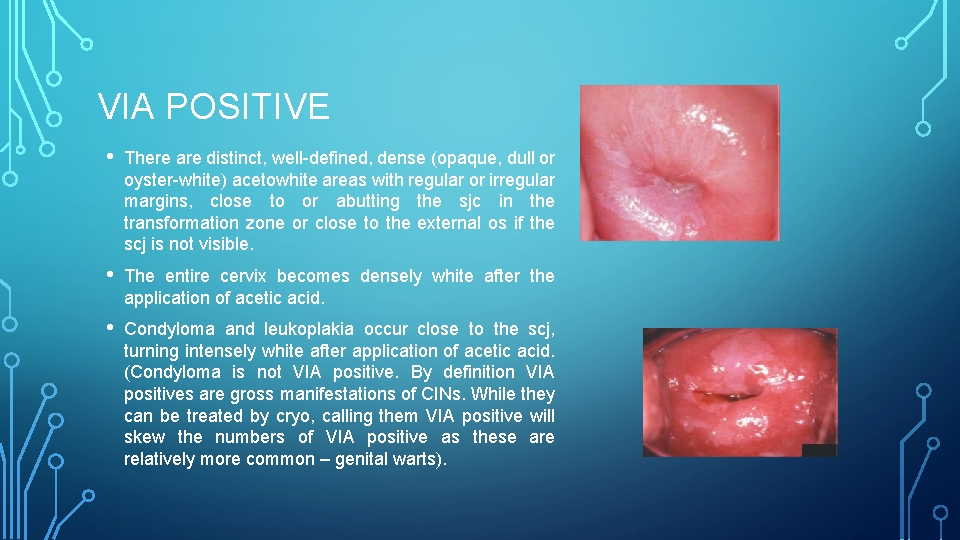
VIA POSITIVE • There are distinct, well-defined, dense (opaque, dull or oyster-white) acetowhite areas with regular or irregular margins, close to or abutting the sjc in the transformation zone or close to the external os if the scj is not visible. • The entire cervix becomes densely white after the application of acetic acid. • Condyloma and leukoplakia occur close to the scj, turning intensely white after application of acetic acid. (Condyloma is not VIA positive. By definition VIA positives are gross manifestations of CINs. While they can be treated by cryo, calling them VIA positive will skew the numbers of VIA positive as these are relatively more common – genital warts).
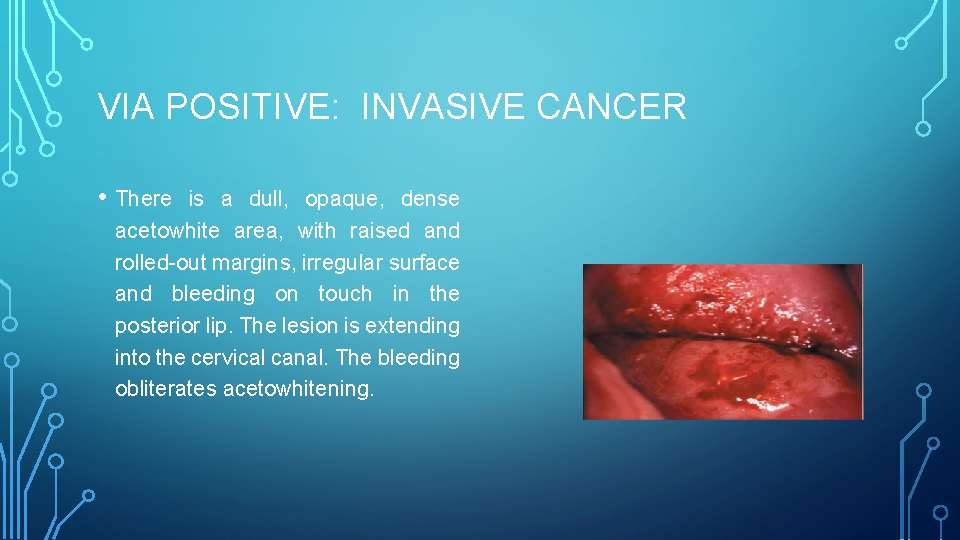
VIA POSITIVE: INVASIVE CANCER • There is a dull, opaque, dense acetowhite area, with raised and rolled-out margins, irregular surface and bleeding on touch in the posterior lip. The lesion is extending into the cervical canal. The bleeding obliterates acetowhitening.
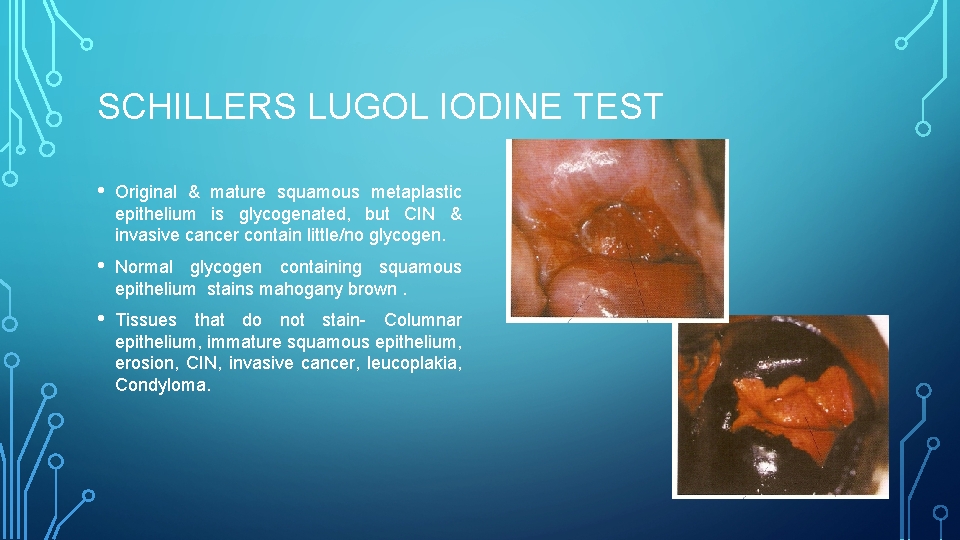
SCHILLERS LUGOL IODINE TEST • Original & mature squamous metaplastic epithelium is glycogenated, but CIN & invasive cancer contain little/no glycogen. • Normal glycogen containing squamous epithelium stains mahogany brown. • Tissues that do not stain- Columnar epithelium, immature squamous epithelium, erosion, CIN, invasive cancer, leucoplakia, Condyloma.
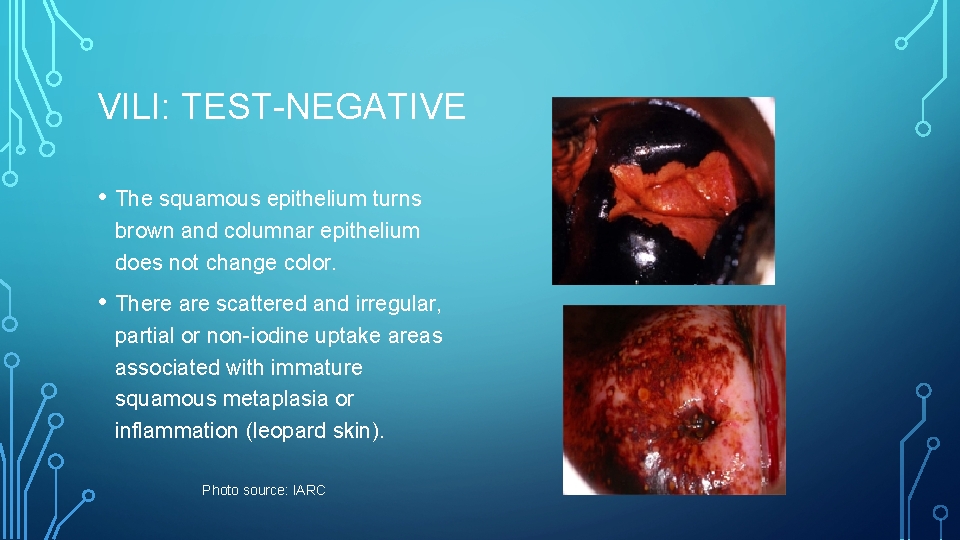
VILI: TEST-NEGATIVE • The squamous epithelium turns brown and columnar epithelium does not change color. • There are scattered and irregular, partial or non-iodine uptake areas associated with immature squamous metaplasia or inflammation (leopard skin). Photo source: IARC
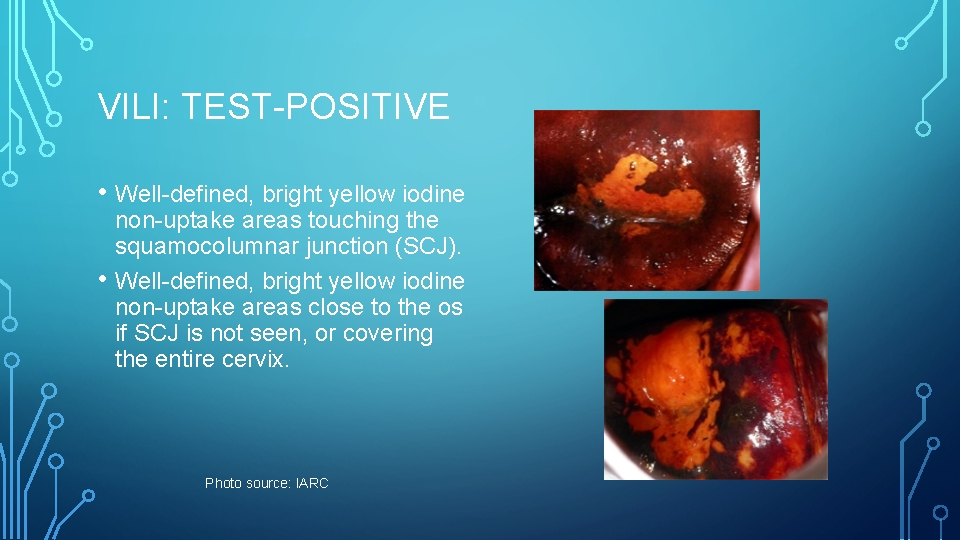
VILI: TEST-POSITIVE • Well-defined, bright yellow iodine • non-uptake areas touching the squamocolumnar junction (SCJ). Well-defined, bright yellow iodine non-uptake areas close to the os if SCJ is not seen, or covering the entire cervix. Photo source: IARC
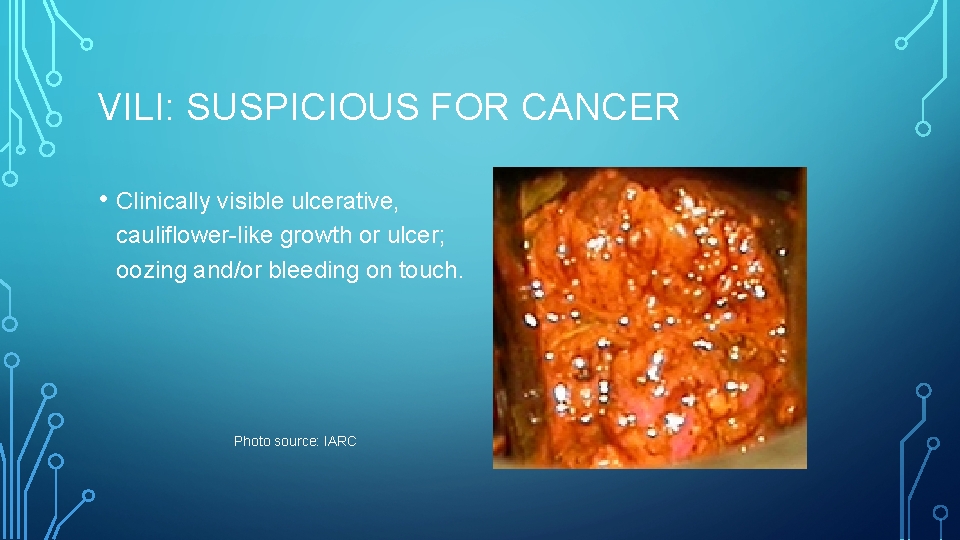
VILI: SUSPICIOUS FOR CANCER • Clinically visible ulcerative, cauliflower-like growth or ulcer; oozing and/or bleeding on touch. Photo source: IARC
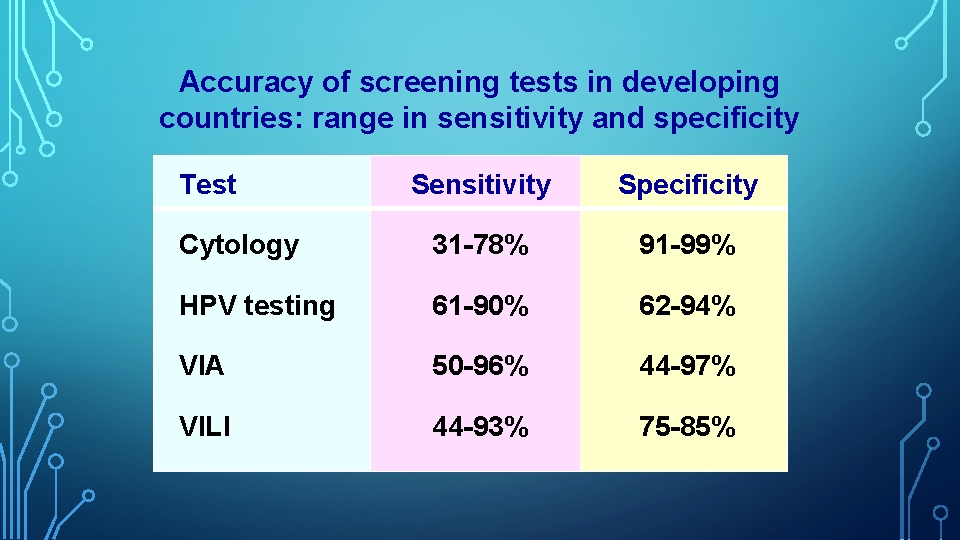
Accuracy of screening tests in developing countries: range in sensitivity and specificity Test Sensitivity Specificity Cytology 31 -78% 91 -99% HPV testing 61 -90% 62 -94% VIA 50 -96% 44 -97% VILI 44 -93% 75 -85%

MANAGEMENT • Mild dysplasia • • • Observation Treat infection, trichomoniasis Menopausal, give hormones Repeat Pap’s 3 monthly, till 3 negative smear Moderate dysplasia/ CIN III • • Colposcpic directed biopsy Cone biopsy, cold knife/ laser conization LEEP/LLETZ, cryocautery, CO 2 laser Hysterectomy Loop electrosurgical excision procedure – large loop excision of transformation zone

SEE & TREAT Cytology, Colposcopy, VIA, LLETZ • No evidence of malignancy on cytology or colposcopy • No endocervical involvement, ECC –ve • Whole of transformation zone visualised • No disparity between cytology & HPE • Patient amenable to follow up.

CRYOCAUTERY • Apply acetic acid to outline the abnormality and begin cryotherapy. There are 2 techniques for freezing the cervical lesion. The double freeze of 3 minute freeze + 5 minute rest + 3 minute freeze or the single freeze of 5 min straight. • • • Tell the woman she might feel some discomfort or cramping while freezing. • Ensure that the vaginal wall is not in contact with the cryoprobe or which may cause a freezing injury to the vagina. • Set the timer and release the gas trigger to cool the probe. Wipe the cryoprobe surface with saline to ensure optimum effectiveness. Apply the cryoprobe tip in the centre of the os and make sure the probe adequately covers the lesion. If the lesion extends more than 2 mm beyond the probe, discontinue the Procedure. Explain to the woman what needs to be done for her as an alternative.

• Ice formation on the tip of the cryoprobe is observed and on the cervix. When the frozen area extends 4– 5 mm beyond the edge of the cryoprobe, freezing is adequate. • Allow two cycles of freezing and thawing: 3 minutes freezing + 5 minutes thawing + 3 minutes freezing. • Once the second freezing is complete, allow time for thawing before attempting to remove the probe from the cervix. Removing it before it is fully thawed will pull tissue off the cervix. • Gently rotate the probe on the cervix to remove it. The area you have frozen will appear white. • • • Examine the cervix for bleeding. If bleeding is noted, apply Monsel’s paste. Do not pack the vagina. Remove the speculum

POST PROCEDURE • Instruct the woman to abstain from intercourse and not to use vaginal tampons for 4 weeks, until the discharge stops completely. This to avoid infection. She should use clean pads • Provide condoms for use if she cannot abstain from intercourse as instructed. Teach her how to use them. This may usually be allowed from the 3 rd week. • Invite her to return in 2– 6 weeks to be checked for healing, and again in 12 months for a repeat VIA or colposcopy. • Inform her of possible complications and ask her to return immediately if she notes: • • fever with temperature higher than 38 °C or shaking chills; severe lower abdominal pain; foul-smelling or pus-like discharge; bleeding for more than two days or bleeding with clots. Perform a pelvic examination to check for healing 2– 6 weeks after the cryotherapy. • At 12 months do a repeat VIA or colposcopy and take a biopsy if necessary.

LOOP ELECTRICAL EXCISION PROCEDURE • Colposcopic assessment should be carried out immediately before LEEP to confirm that the location and linear extent of the lesion are amenable to effective LEEP. • The application of Lugol’s iodine solution is helpful to outline lesion margins before the start of treatment. • An insulated vaginal speculum with an electrically insulating coating or a speculum covered with a latex condom should be used. • Avoid causing pain by inadvertently touching the vaginal walls with the activated electrode. The later possibility may be avoided by using an insulated vaginal sidewall retractor in addition to an insulated vaginal speculum or by using a speculum covered by a condom • A smoke evacuation system with a high rate of flow and a means of filtering out the smoke particles and odour is mandatory.

PROCEDURE • Local anesthesia is achieved 30 seconds after injection of a total of 5 ml or less of 1% xylocaine into the stromal tissue of the ectocervix. The injections are given in a ring pattern 12 mm deep (at 3, 6, 9 and 12 o’clock) at the periphery of the lesion and transformation zone. • • A vasoconstrictor agent such as vasopressin may be added to LA. Use least amount of power that will effectively perform the electro surgery, so as to minimize the risk to the patient’s normal tissues and ensure that the excised specimen is inacceptable condition for pathological assessment. Use blend mode for cutting and coagulation mode for hemostasis. The commonly used power settings for the different loop electrodes are as follows: • • • 1. 0 x 1. 0 cm: 30 watts; 1. 5 x 0. 5 cm: 35 watts; 2. 0 x 0. 8 cm: 40 watts; 2. 0 x 1. 2 cm: 50 watts. 3 mm and 5 mm ball electrodes: 30 watts and 50 watts, coagulation mode. When possible, remove lesion with one pass of the loop electrode using a loop that is wider than the lesion(s) and the transformation zone to be removed. The depth of the loop should be at least 5 mm.

• Introduce loop into the tissue 5 mm outside the outer boundary of the lesion. It is important not to push the electrode in, but to let it cut its own way. Direct the loop gradually into the cervix until the cross bar nearly comes in contact with the epithelial surface. Then the loop is guided along parallel to the surface (horizontally or vertically, depending on the orientation of the direction of cutting until the point is reached just outside the opposite border of the lesion. Withdraw the loop is then slowly, still keeping it at right angles to the surface. The current is switched off as soon as the loop exits the tissue. • The setting on the electrosurgical generator is changed to fulguration and the appropriate power is selected. The surface of the excisional crater is fulgurated using 3 or 5 mm ball electrode. The edges of the crater should also be fulgurated to preserve the squamocolumnar junction in the visible ectocervix. • If active bleeding occurs and is difficult to control using the ball electrode, use macroneedle style electrode to apply the fulguration current in a much more concentrated and localized fashion to a bleeding site. • If satisfactory hemostasis has been obtained, coat the surface of the crater with Monsel’s paste and remove the speculum. • If a lesion involves the endocervical canal and is not likely to be removed with the depth of the usual single-layer pass ( this type of excision can reach a maximum of 1. 6 cm into the endocervical canal). Or if the distal or upper extent of the lesion in the canal cannot be seen and if the distal end of the lesion extends more than 1 cm into the canal, such patients should undergo cold-knife conization.

FOLLOW UP • Give instructions on self-care and what symptoms to expect after treatment. Women should be advised that they will have a brown or black discharge lasting between a few days and two weeks. • To promptly report back if the discharge persists for more than two weeks, if discharge becomes malodorous and/or is associated with lower abdominal pain or if profuse bleeding develops. • The risk of post-operative infection can be reduced by treating adequately the woman with a likely diagnosis of PID, cervicitis, vaginal trichomoniasis or bacterial vaginosis before performing LEEP. If a woman presents post-operatively with a malodorous discharge, it should be cultured if possible and empirical treatment prescribed with antibiotics for PID (doxycycline 100 mg BD x 7 days and metronidazole 400 mg orally TDS X 7 days). • • • If post-operative pain occurs, use acetaminophen or ibuprofen. Not to use a vaginal douche or tampons, or to have sexual intercourse for one month. A follow-up appointment should be made for review at 9 -12 months after treatment.
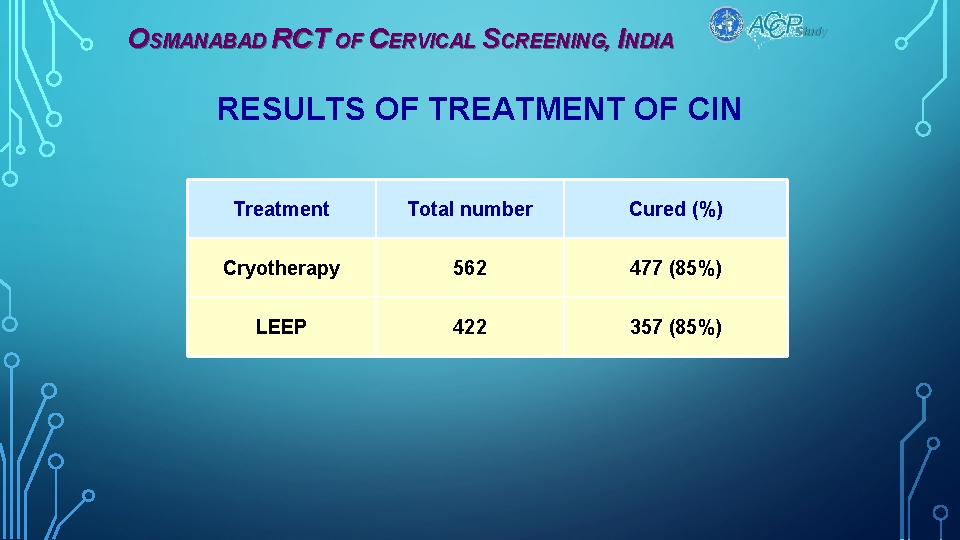
OSMANABAD RCT OF CERVICAL SCREENING, INDIA RESULTS OF TREATMENT OF CIN Treatment Total number Cured (%) Cryotherapy 562 477 (85%) LEEP 422 357 (85%) Study

HPV VACCINE Gardasil ® What is it? Gardasil ® is a vaccine approved by the Food and Drug Administration (FDA) to protect against four types of HPV: 6, 11, 16, and 18 Who can get it? ü The CDC and FDA recommends girls and women aged 9 -26 should get the vaccine. Why 6, 11, 16, and 18? ü Types 6 and 11 cause most cases of genital warts ü Types 16 and 18 are high-risk types that can cause cervical cancer (~70%) How effective is it? ü Studies have shown the vaccine to be almost 100% effective in preventing diseases caused by the 4 types of HPV it covers ü

OF GREAT IMPORTANCE… Cervarix™ What is it? ü Cervarix™ is an HPV vaccine produced by Glaxo. Smith. Kline that has not yet been approved by the Food and Drug Administration (FDA). It has been approved in Australia and Europe. What does it protect against? ü Cervarix™ protects against types 16 and 18. These are high-risk types that cause cervical cancer over 70% of all cervical cancer cases.

DOWN STAGING OF CERVICAL CANCER WHO • In resource poor settings • Aims to pick up cervical cancer in early stage when amenable to treatment • Health workers trained to do per speculum examination of cervix and refer bad cervices for cytology/ biopsy • Low sensitivity & specificity • Can be helped by VIA / VILI

MANAGEMENT OF CERVICAL CANCER

INVASIVE CANCER CERVIX


ETIOLOGY • Age, 2 peaks – 35 yrs, 50 -55 yrs • Genetic & racial factors, rare in Jews & Muslims, high in Africans. • Low SE status, lack of hygiene • Early age at coitus, frequent intercourse & change of partners, 1 st wife • • • had cervical cancer, parous women HPV 16, 18, 31, 45 OCPs favour CIN Smokers

PATHOGENESIS l There are 3 steps necessary for cervical cancer development 1. HPV infection 2. Progression to cervical intraepithelial neoplasia (CIN) 3. Invasion HPV infection must persist for more than one year Normal Cervix HPV Infection HPV Clearance Infected Cervix Progression Invasion CIN Regression Cancer
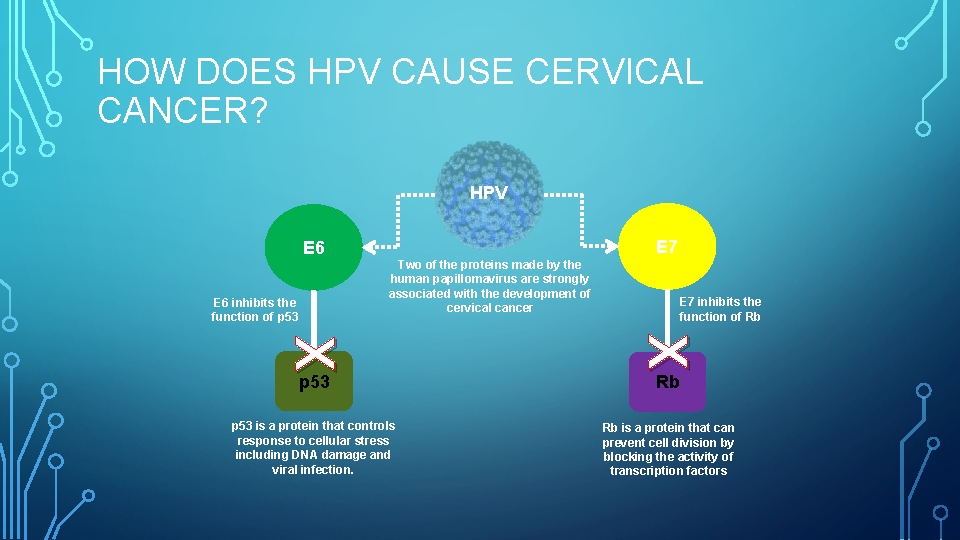
HOW DOES HPV CAUSE CERVICAL CANCER? HPV E 7 E 6 Two of the proteins made by the human papillomavirus are strongly associated with the development of cervical cancer E 6 inhibits the function of p 53 E 7 inhibits the function of Rb X X p 53 is a protein that controls response to cellular stress including DNA damage and viral infection. Rb is a protein that can prevent cell division by blocking the activity of transcription factors p 53 Rb
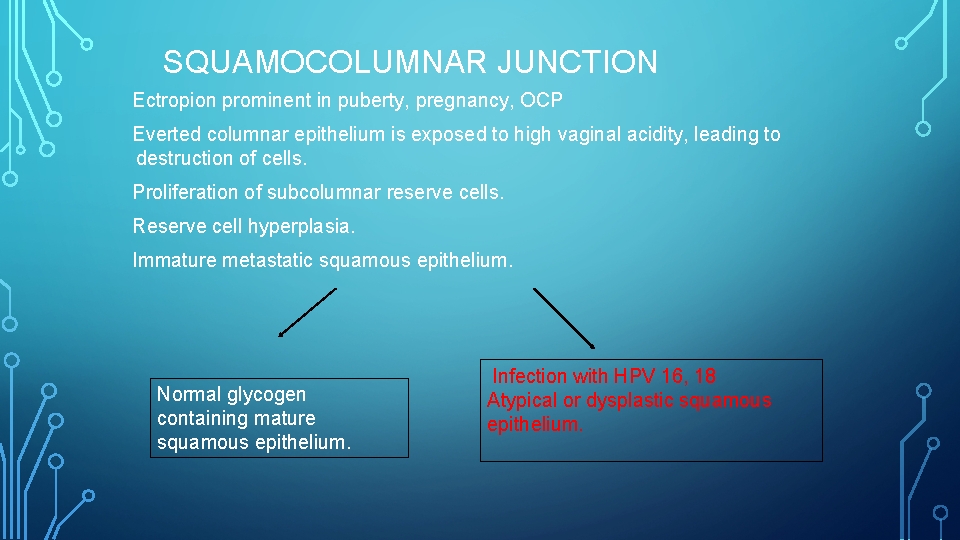
SQUAMOCOLUMNAR JUNCTION Ectropion prominent in puberty, pregnancy, OCP Everted columnar epithelium is exposed to high vaginal acidity, leading to destruction of cells. Proliferation of subcolumnar reserve cells. Reserve cell hyperplasia. Immature metastatic squamous epithelium. Normal glycogen containing mature squamous epithelium. Infection with HPV 16, 18 Atypical or dysplastic squamous epithelium.
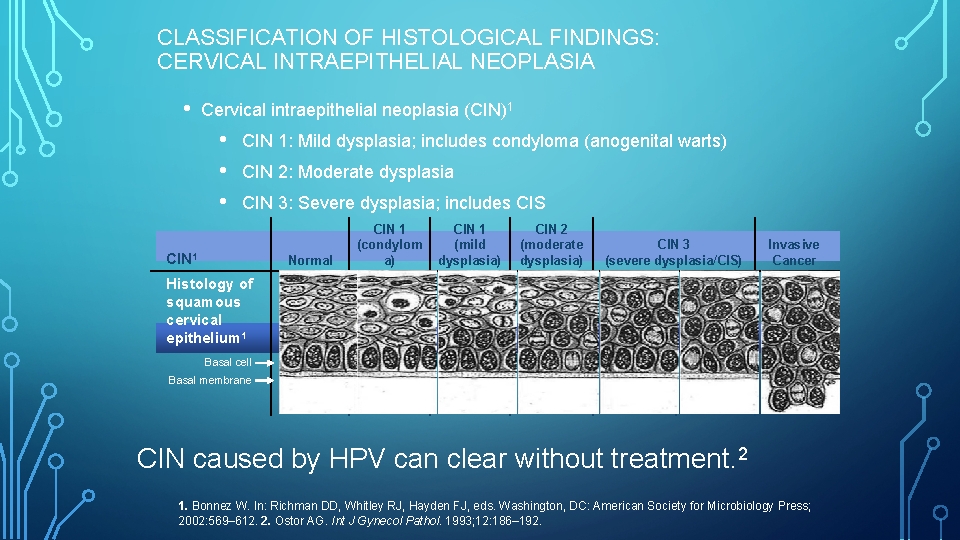
CLASSIFICATION OF HISTOLOGICAL FINDINGS: CERVICAL INTRAEPITHELIAL NEOPLASIA • Cervical intraepithelial neoplasia (CIN)1 • • • CIN 1: Mild dysplasia; includes condyloma (anogenital warts) CIN 2: Moderate dysplasia CIN 3: Severe dysplasia; includes CIS CIN 1 Normal CIN 1 (condylom a) CIN 1 (mild dysplasia) CIN 2 (moderate dysplasia) CIN 3 (severe dysplasia/CIS) Invasive Cancer Histology of squamous cervical epithelium 1 Basal cell Basal membrane CIN caused by HPV can clear without treatment. 2 1. Bonnez W. In: Richman DD, Whitley RJ, Hayden FJ, eds. Washington, DC: American Society for Microbiology Press; 2002: 569– 612. 2. Ostor AG. Int J Gynecol Pathol. 1993; 12: 186– 192.

CERVICAL CANCER RISK FACTORS • Risk factors for persistent HPV infection • • • Lifetime number of sexual partners Age at first intercourse Smoking Oral contraceptive use Male partner sexual behavior • Additional risk factors • • • Age Genetics Low socioeconomic class Nutrition Immune suppression

HISTOPATHOLOGICAL TYPES • • Squamous cell carcinoma – 80 -90% • Large cell keratinizing – 20% • Large cell non-keratinizing – 60% • Small cell non-keratinizing – 20% • Verrucous Adenocarcinoma – 5 -10% Endometriod adenocarcinoma Clear cell adenocarcinoma Adenosquamous carcinoma Small cell carcinoma Undifferentiated carcinoma • • Grade I well differentiated, Grade II moderately differentiated, Grade III poorly differentiated.

ADENOCARCINOMA CERVIX • Younger women, smokers or pill users • Arises in endocervix • Ulcerative, infilterative, hard indurated barrel shaped cervix • Anaplasia common

SQUAMOUS CELL CANCER • Starts in squamocolumnar junction • • • Hypertrophic or exophytic Eroding or ulcerative Infilterative

SQUAMOUS CELL CANCER WELL DIFFERENTIATED

SPREAD • Direct extension to body of uterus, vagina, bladder & cellular tissues of broad ligament, & uterosacral ligament. • Blood stream to ovaries, brain, bones, lung. • Lymphatic from lymphatics in base of broad & uterosacral ligament to Obturator, Sacral, External & Internal Iliac Lymph nodes.

SYMPTOMS & SIGNS • Symptomless • Postcoital bleeding • Irregular uterine bleeding or • • • discharge Late frequency, dysuria, incontinence Rectal pain, backache sciatica, leg edema Loss of weight, anorexia, malaise • Hard, indurated • Irregular • Bleeds on touch • Friable • Fixity

DIAGNOSIS & CLINICAL STAGING • • • Per speculum, bimanual & per rectal examination Colposcopy, directed biopsy, endocervical curettage Hysteroscopy, cystoscopy, proctoscopy Intravenous pyelography X-Ray Chest & skeleton. Optional – laparoscopy, ultrasonography, CT scan, MRI scan, PET scan.

FIGO STAGING • Stage 0 carcinoma in situ • Stage 1 Cervical carcinoma confined to uterus. Spread to corpus disregarded. • Stage 1 A invasive cancer diagnosed only on microscopy • Stage 1 A 1 stromal invasion <3 mm depth & <7 mm horizontal spread. • Stage 1 A 2 stromal invasion >3 mm but <5 mm in depth, horizontal spread <7 mm.

STAGE 1 B OVERT CANCER CONFINED TO CERVIX • Stage 1 B 1 clinically visible lesion 4 cm or less in greatest dimension. • Stage 1 B 2 clinically visible lesion >4 cm in greatest dimension.

• Stage 2 Tumor invades beyond the uterus but not to the pelvic wall, or • • to the lower third of vagina. Stage 2 A without parametrial involvement Stage 2 B with parametrial involvement. • Stage 3 tumor extends to lateral pelvic wall, and/or involves lower third • • of vagina and/or causes hydronephrosis, nonfunctioning kidney. Stage 3 A tumor extends to lower third vagina, no extension to pelvic wall. Stage 3 B tumor extends to pelvic wall.

STAGE III A

• Stage 4 A tumor invades mucosa of bladder rectum and/or extends beyond true pelvis. • Stage B distant metastasis.

PROGNOSIS • • • Extent of growth at time of treatment Site - endocervical more dangerous Size – hypertrophic, florid, massive growth filling vagina Histology - adenocarcinoma Age – young person Ureteric obstruction Grade - poorly differentiated or anaplastic Lymph node involvement Metastasis

TREATMENT PATTERNS IN CARCINOMA CERVIX Sx (5%) Comb (12%) RT (83%) TATA HOSPITAL CANCER REGISTRY 2000

MANAGEMENT • Stage 1 A 1 • Conisation • Extrafascial total abdominal hysterectomy (type 1 Radical) • Stage 1 A 2 • Type 2 Radical Werthiem’s hysterectomy • Pelvic node dissection

Stage 1 B + 2 A • Type III radical hysterectomy with pelvic lymph node sampling. Meigs hysterectomy Schauta Amreich opt. Mitra’s hysterectomy with bilateral extraperitoneal lymphadenectomy Stage 1 B exophytic

Indications of surgery • Stage 1 & 2 A • Young patient • Preserve ovarian function, ovaries involved in <1% • Pregnancy • Chronic PID, fibroids • Endocervical barrel shape, columnar adenocarcinoma. Stage 1 B

Complications • Mortality 1 -2% • Hemorrhage, shock, peritonitis, paralytic ileus, intestinal obstruction, thromboembolism • Bladder atony, cystitis, pyelitis, hydronephrosis, stenosis of ureter. Stage 1 B endocervical

Chemotherapy • Cisplatinium based • Neoadjuvant chemotherapy, for preopt. large bulky tumors • Concurrent chemotherapy with RT for bulky advanced cancer. Stage 2 A

B Stage 2 B, 3 & 4 Radiotherapy • EBRT+ ICRT • Primary target, Point A EBRT 50 Gy/5 -6 weeks (Linear accelerator Cobalt 60) + LDR ICRT 3035 GY (LDR or HDR) • Secondary target, Point B EBRT 50 Gy/5 weeks A Stage 2 B 1 Gray == 100 rads

Radiotherapy + Concurrent chemotherapy Cis platinium 40 mg/m 2 weekly during external radiation. Stage 3 A

Interstial brachytherapy • Transperineal perforated templates with iridium 192 or iodine 125 • Advanced parametrial disease • Distorted anatomy • Postopt. or postradiation recurrence Stage 3 B

Stage 4 A Complications • Flare up PID • Diarrhoea, abdominal cramps, nausea, bowel bleed • Arteritis, fibrosis, • Radiation proctitis • Subacute intestinal obstruction • Fistula • Avascular necrosis femur neck • Menopausal symptoms

5 YEAR SURVIVAL RATES • Overall • Radiotherapy Stage 1 - 85% Stage 1 - 80 -90% Stage 1 B Stage 2 - 55 -70% 87 -91% - node -ve Stage 3 - 30 -35% 51 -67% - node +ve Stage 4 - 10% Stage 2 - 60% • Stage 3 - 33% Stage 1 - 70% Stage 4 – 10% Stage 2 - 45 -50% Surgery
- Slides: 100