Scintillators Scintillating fibres emitting blue light from UV

Scintillators Scintillating fibres emitting blue light from UV illumination, SCSF-78 MJ, LHCb Sci. Fi tracker Lecture 11

Scintillators Scintillation light is the light that is emitted when ionizing radiation is the source of the energy. Scintillation is luminescence stimulated by ionizing radiation. Basic principle: The ionising radiation moves electrons into excited states. The de-excitation to lower energetic states emits detectable light! If the deexcitation is fast (immediate), the process is called fluorescence if significantly retarded (>10 -6 s) phosphorescence. General features: 1) The amount of light produced is proportional to the energy deposit, good for spectrometer 2) Fast time response allows for time information of passage of particle to measure time-of-flight 3) Pulse shape discrimination allows for particle identification, particles of different ionizing power produce different time distribution.

Inorganic and organic Scintillators Inorganic (no C content): • Crystals (LYSO, Na. I, Cs. I) • Gases (N 2 and nobel gases), emission fast but in UV • High Z atoms, high density (3 -8 g/cm 3) • High light yield (10 K-100 K photon/Me. V) • Slow decay times • Expensive (production) • Hygroscopic • Temp dependent Light yield (%/K) Organic: • Plastic (organic fluors in a base polymer) • Organic crystals (anthracene) • Liquids Low Z / density (1 -2 g/cm 3) Low light yield (<10 K photons/Me. V) Fast decay times Practical for being cheap, shapeable, machinable • Independent of temperature (-60°C to +20°C) • •

Why are not all materials scintillators? Normally the emission spectrum ≈ absorption spectrum => no visible scintillation light can exit the material due to immediate re-absorption! Scintillators: Emission spectrum ≠ absorption spectrum

Scintillation process in inorganic scintillator The crystal has an energy band structure similar to semi-conductors. Activation centers (impurities) are added to obtain intermediate energy levels. Valence electrons excited by passage of ionizing particle can jump to conduction band. The light emitted from the activation center levels is shifted in energy to allow for low reabsorption. 3 possible return to ground state processes are possible: 1) Luminescence centers: transition to ground state accompanied by emission of photon => scintillation! 2) Quenching centers: return to ground state without radiation => detector inefficiency 3) Electron traps: metastable levels holding electron for some time. Then either radiation less return to ground state or thermal excitation back into conduction band => inefficiency or delayed signal (afterglow, slow component)

Which crystal to chose?
![Inorganic material properties: Scintillator Material Density [g/cm 3] Radiation length [cm] Wavelength Decay emission Inorganic material properties: Scintillator Material Density [g/cm 3] Radiation length [cm] Wavelength Decay emission](http://slidetodoc.com/presentation_image_h2/7060d89fd501857bdde0079345f01c1b/image-7.jpg)
Inorganic material properties: Scintillator Material Density [g/cm 3] Radiation length [cm] Wavelength Decay emission time[ns] Photons /Me. V (LY) Na. I (Tl)* 3. 67 2. 56 415 230 43 k Cs. I (Tl)* 4. 51 2. 43 560 630 38. 6 k BGO 7. 13 1. 12 505 300 8. 2 k Pb. WO 4 8. 3 0. 89 420 6 100 LYSO (Ce) 7. 3 1. 16 428 50 33 k Cs. F 4. 64 2. 69 390 2 -4 1. 9 k Ba. F 2 4. 88 1. 25 220/310 0. 6/620 1. 43 k/10 k LAr 1. 4 14 120 -170 5/860 ~40 k SCSF-78 1. 06 41. 3 440 2. 7 8 k *Thallium doped LYSO is the most commonly used crystal for PET and TOFPET applications Organic plastic scintillator used for the Sci. Fi tracker in LHCb for comparison
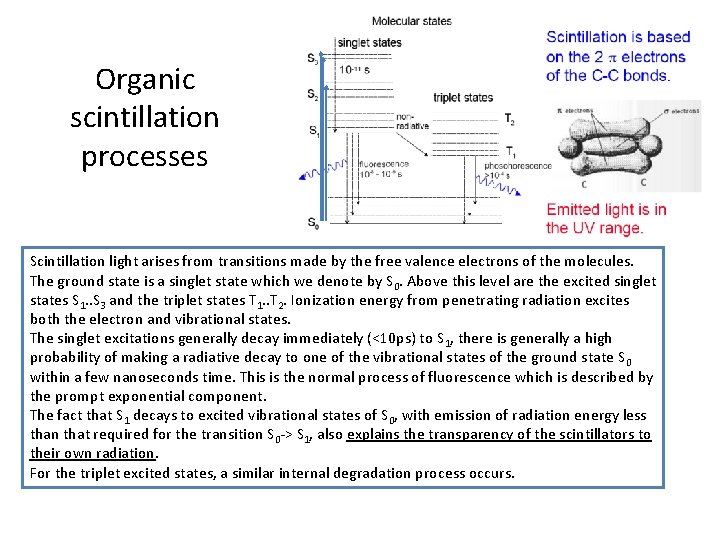
Organic scintillation processes Scintillation light arises from transitions made by the free valence electrons of the molecules. The ground state is a singlet state which we denote by S 0. Above this level are the excited singlet states S 1. . S 3 and the triplet states T 1. . T 2. Ionization energy from penetrating radiation excites both the electron and vibrational states. The singlet excitations generally decay immediately (<10 ps) to S 1, there is generally a high probability of making a radiative decay to one of the vibrational states of the ground state S 0 within a few nanoseconds time. This is the normal process of fluorescence which is described by the prompt exponential component. The fact that S 1 decays to excited vibrational states of S 0, with emission of radiation energy less than that required for the transition S 0 -> S 1, also explains the transparency of the scintillators to their own radiation. For the triplet excited states, a similar internal degradation process occurs.
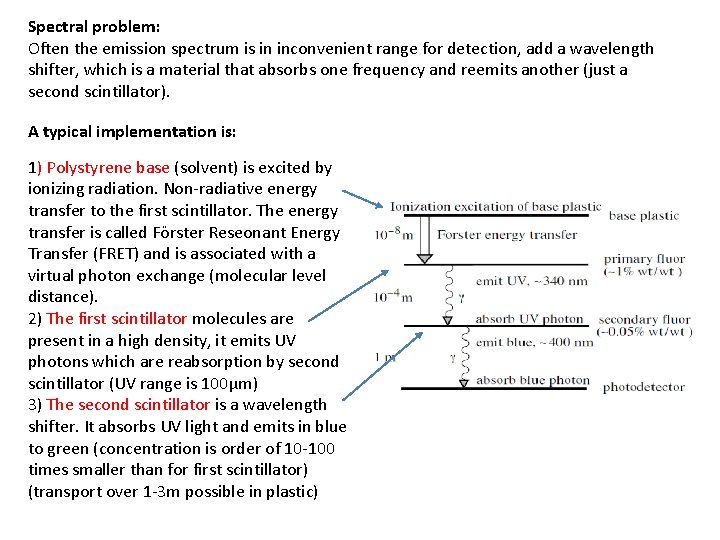
Spectral problem: Often the emission spectrum is in inconvenient range for detection, add a wavelength shifter, which is a material that absorbs one frequency and reemits another (just a second scintillator). A typical implementation is: 1) Polystyrene base (solvent) is excited by ionizing radiation. Non-radiative energy transfer to the first scintillator. The energy transfer is called Förster Reseonant Energy Transfer (FRET) and is associated with a virtual photon exchange (molecular level distance). 2) The first scintillator molecules are present in a high density, it emits UV photons which are reabsorption by second scintillator (UV range is 100μm) 3) The second scintillator is a wavelength shifter. It absorbs UV light and emits in blue to green (concentration is order of 10 -100 times smaller than for first scintillator) (transport over 1 -3 m possible in plastic)
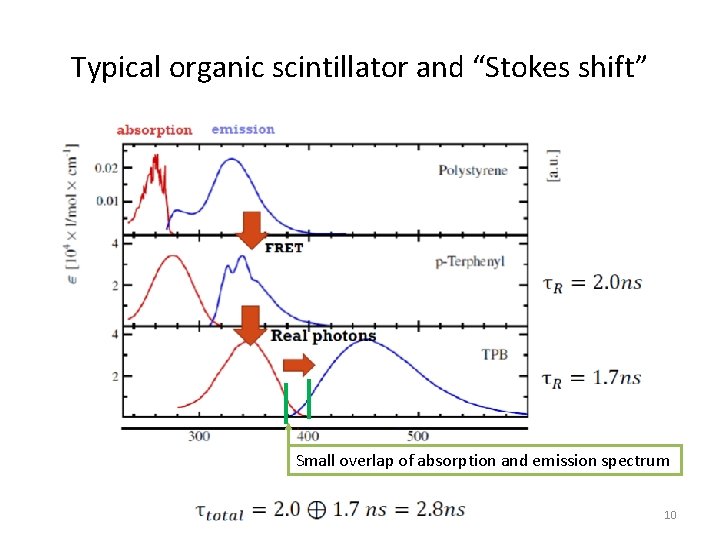
Typical organic scintillator and “Stokes shift” Small overlap of absorption and emission spectrum 10

Scintillator read-out, light guide Crystals: Usually direct coupling to photon detector (small blocks) with optical grease. Plastics: Often collection and guidance of light is needed to fit to sensitive surface of photodetector, due to space constraints, to avoid magnetic field or to avoid high radiation. Use light guides as fibres or bend plastic pieces:
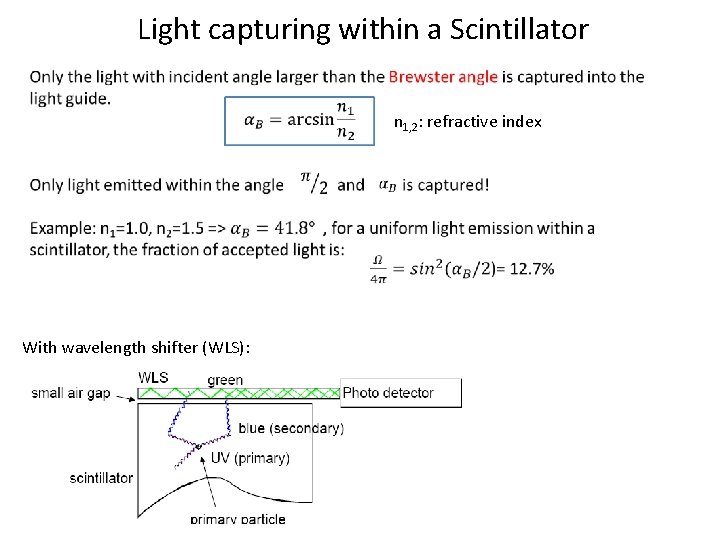
Light capturing within a Scintillator n 1, 2: refractive index With wavelength shifter (WLS):
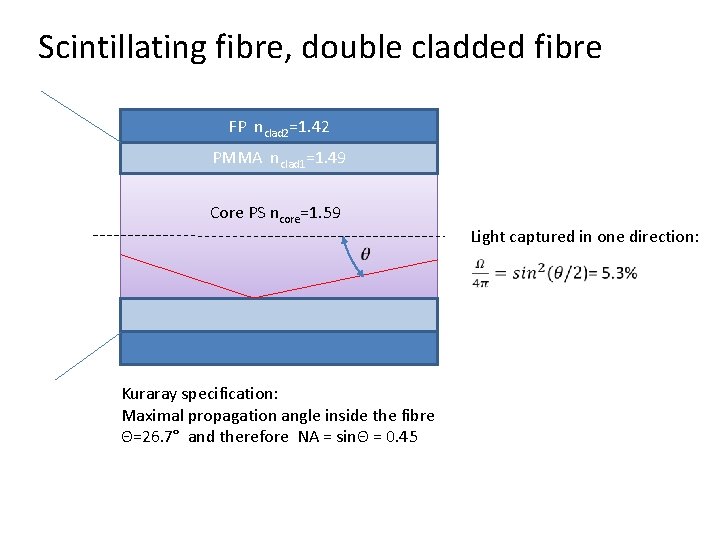
Scintillating fibre, double cladded fibre FP nclad 2=1. 42 PMMA nclad 1=1. 49 Core PS ncore=1. 59 Kuraray specification: Maximal propagation angle inside the fibre Θ=26. 7° and therefore NA = sinΘ = 0. 45 Light captured in one direction:

Timing performance for Scintillators The best timing performance is achieved by measuring the first arriving photon from the scintillation process. First single photon timing performance (width of distribution of the arrival time): Photon emission time dependence or expressed in Coincidence Time Resolution (CTR) for single photon detection between two detectors: [1] S. Gundacker, R. M. Turtos, E. Auffray, P. Lecoq. Nuclear inst. and Methods in Physiscs Research, A 891 (2018) 42 -52
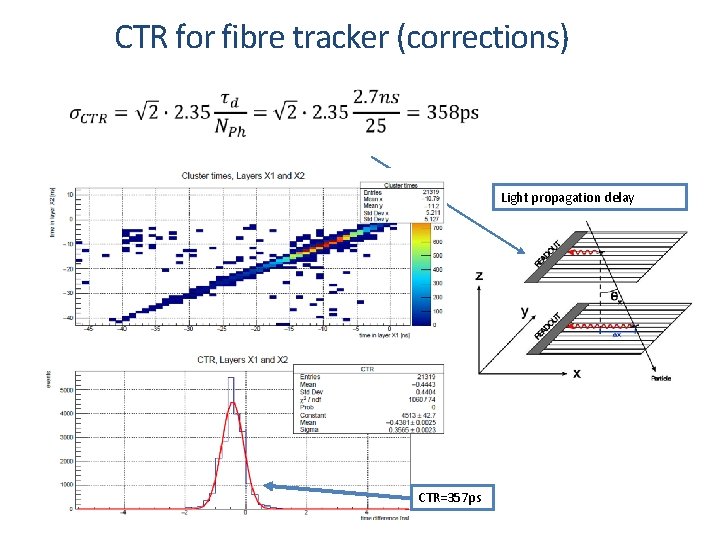
CTR for fibre tracker (corrections) Light propagation delay CTR=357 ps
- Slides: 15