Scientists Greek Philosophers Many ancient scholars believed matter

Scientists

Greek Philosophers • Many ancient scholars believed matter was composed of such things as earth, water, air, and fire. • Many believed matter could be endlessly divided into smaller and smaller pieces.

Greek Philosophers (cont. ) • Democritus (460– 370 B. C. ) was the first person to propose matter was not infinitely divisible, but made up of individual particles called atomos. • Aristotle (484– 322 B. C. ) disagreed with Democritus because he did not believe empty space could exist. • Aristotle’s views went unchallenged for 2, 000 years until science developed methods to test the validity of his ideas.

Alchemists

Dalton • In 1804 Dalton stated that atoms were – Tiny individual particles – All identical in a given element with identical properties – Two or more elements atoms will combine in fixed ratios – “Billiard Ball Model”

Dalton’s Matter Model • All matter is made of atoms • All atoms of an element are identical in properties • Atoms of different elements can combine in specific ratios to form new substances

Law of Definite Proportions • A compound always has the same ratio of atoms, regardless of how it is made. Example: water is always H 2 O, not HO, or HO 2, or H 2 O 2 7

J. J. Thompson • Discovered that atoms had negative particles (electron) contained inside the atom • “Raisin Bun Model”
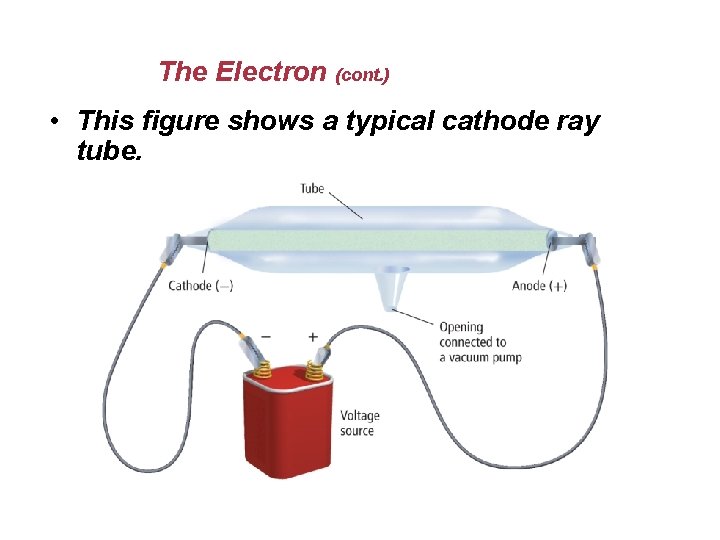
The Electron (cont. ) • This figure shows a typical cathode ray tube.

The Electron • When an electric charge is applied, a ray of radiation travels from the cathode to the anode, called a cathode ray. • Cathode rays are a stream of particles carrying a negative charge. • The particles carrying a negative charge are known as electrons.

• Matter is neutral. • J. J. Thomson's plum pudding model of the atom states that the atom is a uniform, positively changed sphere containing electrons.
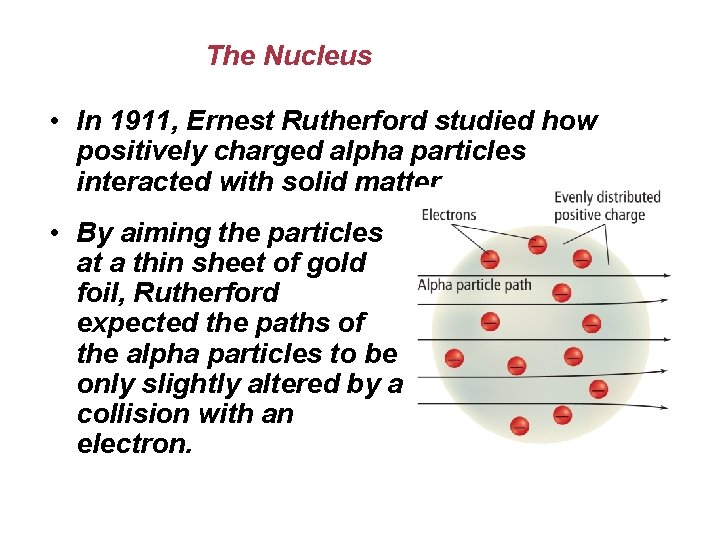
The Nucleus • In 1911, Ernest Rutherford studied how positively charged alpha particles interacted with solid matter. • By aiming the particles at a thin sheet of gold foil, Rutherford expected the paths of the alpha particles to be only slightly altered by a collision with an electron.
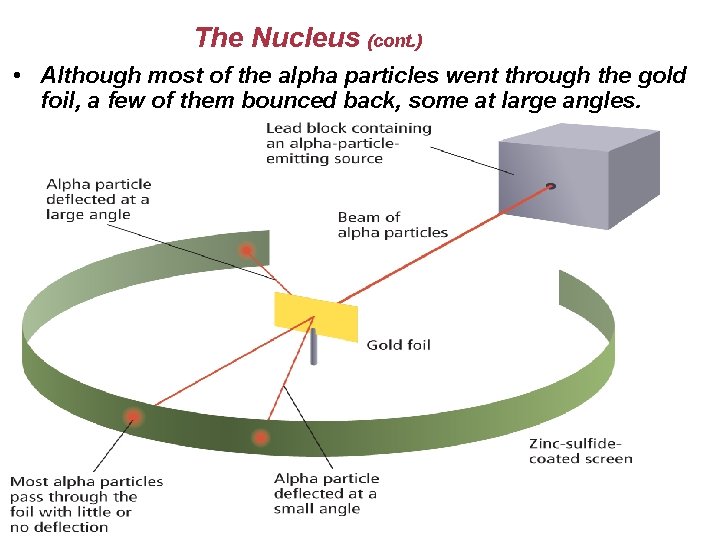
The Nucleus (cont. ) • Although most of the alpha particles went through the gold foil, a few of them bounced back, some at large angles.
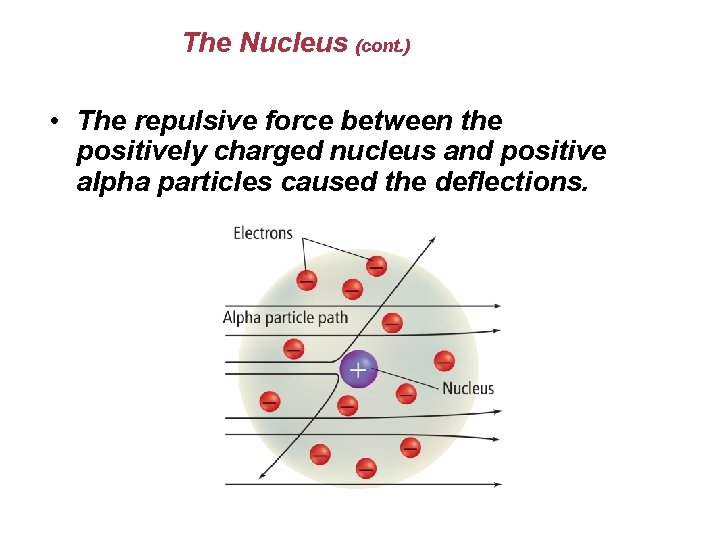
The Nucleus (cont. ) • The repulsive force between the positively charged nucleus and positive alpha particles caused the deflections.

Bohr • Thought that electrons were arranged in certain energy levels around a positive nucleus • Electrons in orbitals • “Bohr Model”

The Periodic Table https: //www. youtube. com/watch? v= u. Pk. EGAHo 78 o. 15 minutes

Relationship of Chemical Symbols to Chemical Names • Alchemists used artistic symbols to represent the elements • Dalton updated the symbols but they were still cumbersome 17

Elements • The number of protons in the nucleus and the distribution of electrons around it determines the type of element and the chemical and physical properties of the element. Eg. hydrogen is 1 p surrounded by 1 e- bromine is 35 p and 45 n surrounded by 35 e- 18

Elements • Most elements occur naturally as single atoms (monoatomic) eg. Ne • Other elements occur naturally as combinations of two or more atoms (molecular elements) – diatomics eg. O 2, N 2, H 2, Cl 2, – polyatomics eg. P 4, S 8 19

SIMPLE EXPERIMENT CAN YOU PROVE, PLANTS NEED SUNLIGHT TO GROW? OK, SO PROVE IT……

• The independent (manipulated) variable is the one that is changed by the scientist. As the scientist changes the independent variable, he or she observes what happens.

• The scientist focuses his or her observations on the dependent (responding) variable to see how it responds to the change made to the independent variable. What is the ruler for?

• Controlled variables are quantities remain constant. So , could we use a ctaus and compare that to a rose bush? Why or why not. Is it ok to water one plant every day but not water the other one at all?

Summary • Independent (Manipulated) Variable: What we are changing • Dependent (Responding) Variable: The Response (what happens) • Controlled Variable: What remains the same
- Slides: 24