Scientific Notation Significant figures And Dimensional Analysis Scientific

















































- Slides: 52

Scientific Notation Significant figures And Dimensional Analysis

Scientific Notation l l An alternate way of writing numbers – especially those that are very LARGE or very SMALL The only measures you CAN’T use it with is money or temperature!

Scientific Notation l Two Parts to the notation Mantissa – the base 10 number – Characteristic – the exponent to which the base 10 number (mantissa) is raised Mantissa Characteristic – 4. 1 x – 5 10 Correct scientific notation MUST HAVE BOTH PARTS even if the characteristic is zero!

Scientific Notation l Positive characteristic means the number is very LARGE – – l 3. 0 x 1010 = 30, 000, 000 If the original number is > 1 then the characteristic will be positive Negative characteristic means the number is very SMALL – – 3. 0 x 10 -10 = 0. 000003 If the original number is < 1 then the characteristic will be negative

Examples l l l l 17600 0. 00135 10. 2 -67. 30 4. 76 -0. 1544 301. 0 -0. 000130

Scientific Notation to Normal Notation l To expand scientific notation to regular or normal notation look at the characteristic. – If the characteristic is POSITIVE move the decimal that many places to the RIGHT l – 4. 96 x 102 = 496. If the characteristic is NEGATIVE move the decimal that many places to the LEFT l 5. 61 x 10 -3 = 0. 00561

Examples l l l l 36. 7 x 101 -0. 015 x 10 -3 75. 4 x 10 -1 -14. 5 x 10 -2 0. 123 x 104 97723 x 102 851. 6 x 10 -3 94. 2 x 10 -4

Scientific Notation l Multiplying – – MULTIPLY the mantissa as you would any other multiplication ADD the characteristics CONVERT the results to the correct notation if necessary (2 x 104)(3 x 10 -3) l l l 2 x 3 = 6 4 + (-3) = 1 6 x 101 final answer

Examples l (5 x 10 -3) (4 x 10 -4) l (6 x 104) (-7 x 10 -6) l (-4. 5 x 10 -2) (2 x 10 -7)

Dividing Scientific Notation l To divide DIVIDE the mantissa as you normally would – SUBTRACT the characteristic of the denominator (bottom) from the characteristic of the numerator (top) – CONVERT the result if necessary 8 x 10 -5 2 x 10 -3 8/2 = 4 ; (-5) – (-3) =-2 4 x 10 -2 final answer –

Examples l l 4 x 103/8 x 10 -3 6 x 10 -7/3 x 10 -8 4. 5 x 104/9. 0 x 10 -12 (2 x 10 -2)(3 x 10 -3)/(4 x 10 -4)(6 x 10 -2)

Adding and Subtracting l l In order to ADD and SUBTRACT scientific notation the characteristic MUST BE THE SAME First correct or “incorrect” the mantissa so that the characteristics of the numbers are the same Then add or subtract the manitssas as you normally would Write the characteristic as you have it for both numbers DO NOT ADD the characteristics!!

Examples 1. 64 x 10 -2 +3. 78 x 10 -3 4. 77 x 10 -2 +2. 13 x 10 -4 9. 88 x 101 +6. 37 x 103

Significant Figures l l Two basic rules All non-zero digits are ALWAYS significant – l Any number 1 -9 Zeros have three rules: – – – Zeroes at the beginning of a number are NEVER significant – they are only place holders (0. 0001) Zeroes between NON-zero digits are ALWAYS significant (101) Zeros a the END of numbers are significant only if the number contains a decimal point (100 vs 100. )

Significant figures Zeros that come BEFORE significant numbers are NEVER significant But just like paparazzi you can’t get rid of them!!

Significant Figures Zeros BETWEEN two significant figures are ALWAYS significant

Significant figures l Zeros at the END are ALWAYS significant

Examples l l l l 114. 0 733. 02 0. 000310 9. 080 4. 50 x 103 -23. 40 6. 0040 -0. 0004010

Multiplying & dividing sig figs l l l Count the significant figures in the given numbers. The answer must be rounded to the LEAST number of digits in the given numbers 4. 51 x 3. 4 = 15. 334 = 15 3 digits 2 digits Final answer = 2 digits

Adding & subtracting sig figs l l Count the number of DECIMALS PLACES in the given numbers. The answer must be rounded to the LEAST number of DECIMALS PLACES in the given numbers 13. 452 145 13. 000 875. 03

Examples l 67. 4432 + 3. 07 +(-245. 21) – 2. 9 l -861. 2 + 78. 554 – 91. 0045 +0. 0203

l If the problem contains both addition/ subtraction AND multiplication/division you must take the problem apart and deal with each part appropriately 78. 4 x 0. 00513 3. 2 +1. 41

Metric Units l l l System International units or SI are the units used in science. English units are the COMMON unit used in the United States. BOTH systems are both accurate There are 88 English units There are only 3 major SI units and 9 prefixes

Metric Units l What are the English units for length? l The metric units for length is the meter (m) What are the English units for volume l l The metric unit for volume is the liter (L) What are the English units of mass (weight)? l The metric unit for mass is the gram (g) l

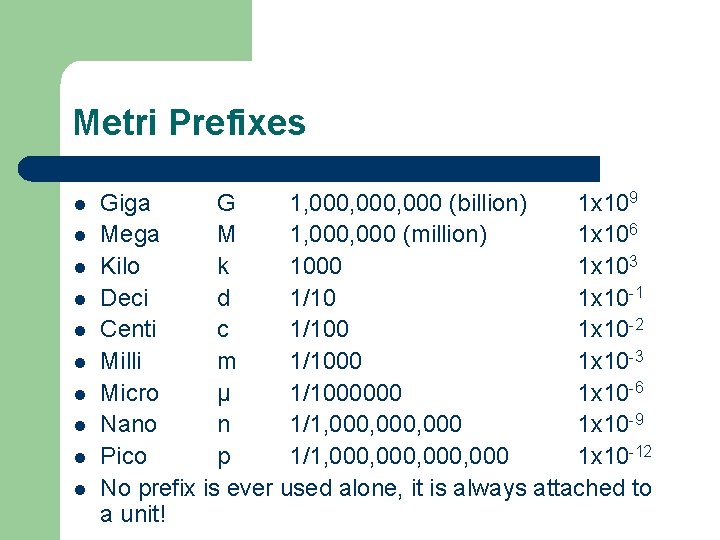
Metri Prefixes l l l l l Giga G 1, 000, 000 (billion) 1 x 109 Mega M 1, 000 (million) 1 x 106 Kilo k 1000 1 x 103 Deci d 1/10 1 x 10 -1 Centi c 1/100 1 x 10 -2 Milli m 1/1000 1 x 10 -3 Micro µ 1/1000000 1 x 10 -6 Nano n 1/1, 000, 000 1 x 10 -9 Pico p 1/1, 000, 000 1 x 10 -12 No prefix is ever used alone, it is always attached to a unit!

Examples l l l l Give the metric abbreviation for each One-millionth of a gram One billion liters On-hundredth of a liter On thousand grams On-thousandth of a meter On million grams

Dimensional Analysis l l Organized method of unit conversion What is 3 hours in minutes? How did you do that? How about 3. 179 hours? – – Not so easy in your head… So HOW do you do it?

Dimensional Analysis There are two relationships which exist between hours and minutes l 60 minutes “per” 1 hour OR 60 minutes 1 hour And 1 hour “per” 60 minutes OR 1 hour 60 minutes l

Dimensional Analysis To convert 3 hours to minutes your brain took the value of 3 hours and multiplied it by one of the two fractions 3 hours x (1 hour) OR 3 hours x (60 minutes) 60 minutes 1 hour Only ONE of these will cancel out the units of hours when multiplied. That’s the one you want! l

Dimensional analysis is a way of arranging the labels (units) so they will cancel out. l Instead of writing fractions in parentheses we are going to place them on a straight line dimensional analysis grid. 3. 179 hours 60 min = 190. 74 min = 190. 7 min 1 hour l

Dimensional analysis l Draw the dimensional analysis grid like so Put what is GIVEN in the UPPER LEFT corner with the unit label GIVEN UNIT LABEL

Dimensional Analysis l Put the SAME label that you put in the upper left in the LOWER RIGHT corner GIVEN UNIT LABEL MATCH THE GIVEN UNIT LABEL l Put the label of what you are converting to in GIVEN UNIT LABEL the UPPER RIGHT LABEL YOU WANT MATCH THE GIVEN UNIT LABEL

Dimensional Analysis l Fill in the correct numerical relationship that exists between the two labels i. e. 60 seconds = 1 minutes GIVEN UNIT LABEL YOU WANT MATCH THE GIVEN UNIT LABEL l l 3. 179 hours 60 seconds 1 hour 190. 74 seconds Cancel out any labels that appear in BOTH the numerator and denominator. Multiply everything together that is above the grid line; Divide everything that is below

Examples l l How many eggs are there in 10. 25 dozen? Metric system unit conversions – – Convert 378. 4 cm to meters Convert 4. 32 x 10 -4 g to micrograms How many picoliters are in 4. 56 x 10 -7 liters? Convert 88. 1 km to meters

Multiple Prefixes l l l How do you know where to put the unit labels for a problem with multiple prefixes? Use one dimension to convert from one prefix to the “plain” unit Then convert from the “plain” unit to the other prefix 231 mm 1 x 10 -3 m 1 nm = 231 x 10 -3 = 2. 31 x 108 nm 1 mm 1 x 10 -9

Examples l l l Convert 5. 43 GL to d. L Convert 6. 99 x 108 pg to cg How many micrometers are there in 45. 2 kilometers?

English Conversions l l l l l The following are conversions you are EXPECTED to KNOW! 1 ton = _____ pounds 1 day = _______ hours 1 pound = _____ounces 1 hours = ______ minutes 1 mile = _____ feet 1 minute = _____seconds 1 foot = _____ inches 1 year = ______days 1 yard = _____ feet 1 month = ____ days 1 pint = ______ cups 1 quart =_____ pints 1 gallon = _____quarts

English to metric unit conversions l l l 2. 54 cm = 1 inch (length) 1. 06 quarts = 1 liter (volume) 454 grams = 1 pound (mass)

Examples l l l Convert 3. 92 yards to picometers Convert 1. 75 x 106 grams to ounces Convert 25. 6 tons to mg

Dimensional analysis of Double units l l Put the number on the TOP label in the upper left corner of the grid Put the number on the bottom label in the lower left corner of the grid You may begin working with either label just be sure to arrange the labels diagonally so they will cancel Work until you get ONE of the labels in the unit you want THEN work on the other label.

Example l Convert 35 miles/hour to km/sec 35 mi 1 hr 1 min 5280 ft 12 in 2. 54 cm 1 x 10 -2 m 1 km 1 hr 60 min 60 s 1 mi 1 ft 1 in 1 cm 1 x 103 m

Examples l Convert 45. 8 Pounds/gallon to ng/microliter l Convert 1. 023 X 107 pm/day to inches/sec

Cubic units l l l Write what is given in the problem in the upper left part of the grid with label STOP AND THINK! You do not know one SINGLE conversion which utilizes cubic units …so transfer just the “plain” unit in the lower right corner and the other “plain” unit in the upper right Now go ahead and fill in the known relationship (i. e. the numbers) BUT!!! Since the label that you started with is CUBIC you MUST CUBE THE ENTIRE DIMENSION both NUMBER AND LABEL!! Sometimes it is necessary to cube more than one of the dimensions to get to the label you want…don’t panic!

Example l Convert 70. 6 cm 3 to m 3 70. 6 cm 3 1 x 10 -2 m 1 cm 3 7. 06 x 10 -5 m 3

Examples l Convert 35 km 3 to cubic feet l Convert 231. 4 cubic yards to cubic millimeters

Final Notes on Dimensional Analysis l l l Read the problem and write down ALL “double label” conversions Begin your dimensional analysis with anything in the problem which is NOT a double label conversion Use each double label conversion factor you have written down only ONCE and arrange the conversion factors on the dimensional analysis grid so that the labels cancel out.

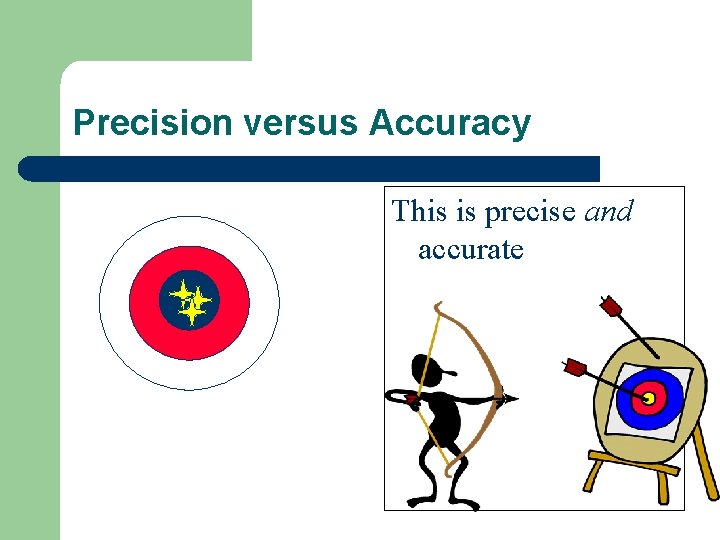
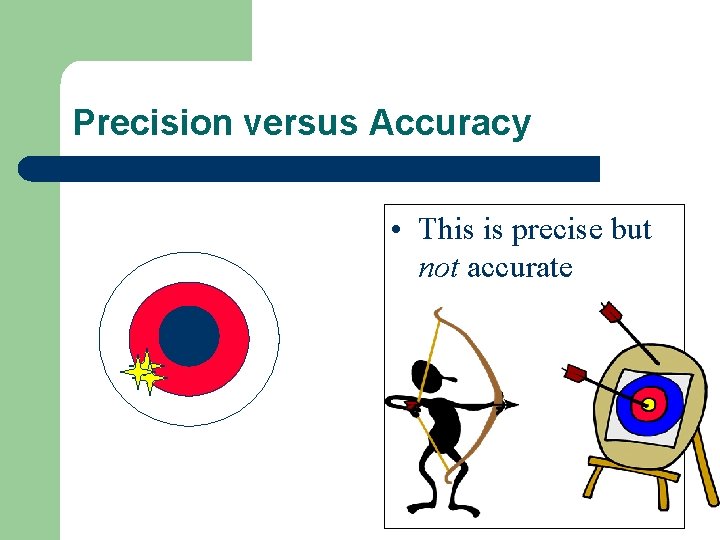
Precision versus Accuracy l if your data is accurate it is close to true value l if your data is precise it is consistent from trial to trial – How close a set of measurements are to EACH OTHER

Precision versus Accuracy This is precise and accurate

Precision versus Accuracy • This is precise but not accurate

Precision versus accuracy l l Student weighs a beaker and records its mass as 47. 21 g, 47. 23, and 47. 19, The actual mass of the beaker is 48. 44 g is this student accurate? Precise? What is the accuracy and/or precision of these basketball free throw shooters? – – – 99 out of 100 shots made 99 out of 100 hit the front of the rim and bounced off 33 out of 100 shots are made and the rest miss the rim completely or rebound off the backboard

Percent Error l l Sometimes you need to know the percent ERROR in your measurements whether it is too high or too low. This is the formula for % error – MEMORIZE IT!! % error = experimental or observed value – actual or true value x 100 actual or true value

Examples A student weighs a beaker and records its mass as 47. 21 g. The actual mass of the beaker is 47. 93 g. What is the student’s % error? % error = 47. 2 g - 47. 93 g x 100 = -1. 502% 47. 93 g l HINT: a negative sign means your answer is that percentage TOO LOW a positive sign means your answer is that percentage TOO HIGH