SCIENCE PHYSICAL SCIENCE GED SUPPORT Kristina Gonzales BS

SCIENCE PHYSICAL SCIENCE GED SUPPORT Kristina Gonzales, BS, MHA

PHYSICAL SCIENCE

Lesson 1: Atoms and Molecules • All matter is made up of atom – Smallest unit of matter that can combine chemically with other matter and cannot be broken down into smaller particles by chemical means • Atoms are made of subatomic particles: – Protons = + – Neutrons = 0 – Electrons = -- • Equal protons and electrons = electrically neutral • When an atom gains or loses electrons, it becomes a negatively charged or positively charged ion

Lesson 1: Atoms and Molecules • Matter – Elements, compounds, or mixtures • Elements substances that cannot be broken down into other substances – A single atom; gold, iron, hydrogen, etc. – Each element has a chemical symbol • Gold=Au • Compounds substances formed of 2 or more elements chemically combined in a definite proportion – Water = H 2 O, chemical formulas • Mixtures physical combos of 2 or more substances that keep their own properties – Salt water
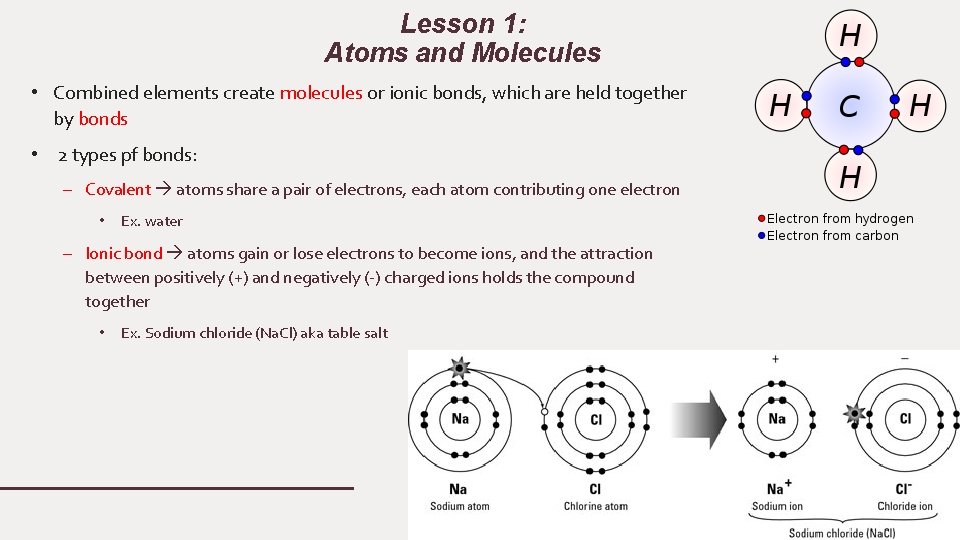
Lesson 1: Atoms and Molecules • Combined elements create molecules or ionic bonds, which are held together by bonds • 2 types pf bonds: – Covalent atoms share a pair of electrons, each atom contributing one electron • Ex. water – Ionic bond atoms gain or lose electrons to become ions, and the attraction between positively (+) and negatively (-) charged ions holds the compound together • Ex. Sodium chloride (Na. Cl) aka table salt

Lesson 2: Properties & States of Matter • Matter anything that has mass and takes up space • Mass of an object is the amount of matter that it contains • Weight measure of the gravitational force exerted on it – Ex. Shovel; mass never changes but the weight can • 3 states of matter: – Solid: a shape & volume; ex. Crystal – Liquids: a volume but no shape; – Gases: no shape or volume; weak attraction between molecules, always in motion & colliding with one another

Lesson 2: Properties & States of Matter • The states of matter can be altered by heat • Heat to solid = melts – Melting point = temperature at which a solid becomes a liquid • Heat to liquid = boils & evaporates, turning into a gas as the motion of its molecules increases – Boiling point = temperature at which a liquid becomes a gas • Heat removed from gas = motion of its molecules decreases and it turns into a liquid – Condensation point = temperature at which a gas becomes a liquid • Heat removed from liquid = motion of its molecules slows until it solidifies – Freezing point = temperature at which a liquid becomes a solid

Lesson 2: Properties & States of Matter • Solution mixture that is uniform throughout and that contains ions, atoms, or molecules of 2 or more substances • Solute substance in a solution that is dissolved • Solvent substance in which the solute is dissolved • Ex. Salt water: salt= solute, water= solvent • Water is the universal solvent • Solutions are not always liquids; alloy = when 2 or more metals are combined , or they can be gases

Lesson 3: Chemical Reactions • Chemical reaction the atoms or ions of one or more substances, called reactants, which are arranged and result in one or more different substances called products – Ex. Iron, water, oxygen react and form Hydrated Iron Oxide aka rust • Matter is neither created nor destroyed during a chemical reaction, therefore the mass of the products always equals the mass of the reactants = law of conservation of mass • Chemical equations they show the reactants on the left side and the products on the right – The equation must balance; same amount of atoms • Energy is involved • Endothermic reaction in which the reactants absorb energy from their surroundings ex. Eggs • Exothermic reaction in which energy is given off with the products, in the form of heat or light

Lesson 3: Chemical Reactions • Energy is involved • Endothermic reaction in which the reactants absorb energy from their surroundings – ex. Eggs • Exothermic reaction in which energy is given off with the products, in the form of heat or light – Wood in a fireplace • Activation energy the amount of energy needed to get a reaction going • https: //www. youtube. com/watch? v=e. JXL 0 Irbtq. E

Lesson 4: The Nature of Energy • Energy capacity to do work • Work done whenever a force is applied to an object to set it in motion • Anything that can force matter to move, change direction, or change speed has energy • Forms of energy: – Heat energy: change a solid to a liquid and a liquid to a gas – Light energy: create an image by causing the chemicals on a piece of film to react – Electrical energy: turn a motor, plate a set of flatware with a layer of silver, or store data on a hard drive – Chemical energy: in food, provides energy to humans – Nuclear energy: from breaking apart the nuclei of atoms provides energy to produce electricity or power a submarine – Mechanical energy: turns the axels of a car or the blades of a fan

Lesson 4: The Nature of Energy • Energy can be converted from one form to another – Fossil fuel burned heat energy water=steam turns blades of turbine produces mechanical energy turbine powers the generator produces electrical energy this energy is used to power homes to provide heat energy, light energy, sound energy, & mechanical energy • While energy undergoes changes in form, the amount of energy in a closed system remains the same = law of conservation of energy • 2 basic types of energy: – Potential energy: because of its position – Kinetic energy: when it moves • Ex. Hitting someone with a water bottle • The energy transferred is equal to the work done by the waterbottle and its measured in = joules

Lesson 4: The Nature of Energy • The rate if doing work or consuming energy = power • Measured in horsepower or watts (joules per second) • Albert Einstein: discovered the relationship between energy and mass and expressed it through an equation – E=mc 2 – E=energy, m=mass, c=speed of light

Lesson 5: Motion & Forces • Everything in the universe is in motion! • Speed= rate at which an object moves • Velocity = is its speed in a particular direction • Acceleration = the rate at which velocity changes • Force = anything that tends to change the state of rest or motion of an object • https: //bestgedclasses. org/science-velocity-acceleration/

Lesson 5: Motion & Forces • Sir Isaac Newton – Created the 3 laws of motion • Law of inertia: – States that an object at rest will stay at rest until a force acts upon it and an object in motion will stay in motion at a constant speed in a straight line until a force acts upon it • Law of motion acceleration: – States that acceleration of an object depends on its mass and the force acting upon it – Greater the force, the greater the acceleration – More massive an object, the more force it takes to accelerate it

Lesson 5: Motion & Forces 2 nd law: law of inertia

Lesson 5: Motion & Forces • For every action, there is an equal and opposite reaction; law of conservation of momentum: – Momentum = amount of energy that a moving object has & it depends on the mass of the object & its velocity – https: //www. youtube. com/watch? v=e 1 lz. B 36 a. HD 4

Lesson 6: Electricity & Magnetism • Remember…charges? • Electric charge causes them to exert forces on one another – Like charges=repel one another; unlike charges= attract one another • Static energy electrons are temporarily pulled away from atoms, creating stationary areas of positive and negative charge • Electric current movement of changed particles, usually electrons – In battery operated devices – Material allows electrons to move freely from atom to atom = conductor; exs. Metals – Material that doesn’t allow electrons to move freely from atom to atom = insulator; exs. Rubber and plastic – Semiconductors= substances whose ability to conduct electricity is midway between that of a conductor and an insulator; exs. silicon

Lesson 6: Electricity & Magnetism • Magnetic field electric current produces this; which affects magnetic substances; they are produced by moving charged particles • Electromagnet charged particles move along a coil of wire connected to a battery or other power source • Permanent magnet spinning of electrons creates a magnetic field • Magnets have 2 ends, which repel one another; north attracts to south

Lesson 6: Electricity & Magnetism • A moving magnetic field produces an electric current; this principle underlies electric motors, generators, and transformers • Electric motor magnetic fields push against one another, turning the shaft of the motor • Generator moving magnetic field produces electric current • Transformer incoming electric current in coiled wire produces fluctuating magnetic fields, which in turn produce an outgoing electric current of a different voltage
- Slides: 20