Science of Foods for Health Biosynthesis Dr Hisashi

Science of Foods for Health Biosynthesis Dr. Hisashi Kiowa Texas A&M University

Biosynthesis and Engineering of Plant Natural Products • What are natural products? • How natural products are produced in plants • Bioactivity of natural products • Engineering natural products biosynthesis--case studies

Natural Products are often called as secondary metabolites Secondary metabolites Primary metabolites • Not essential for plants • Complicated regulation of the biosynthesis – Can be both constitutive and inducible – Tissue specificity • Bioengineering – Complex chemical structure (difficult though not impossible to chemically synthesize) – Beneficial for human use

Nature may not be the best source of Natural Products Accumulation of natural products can take years
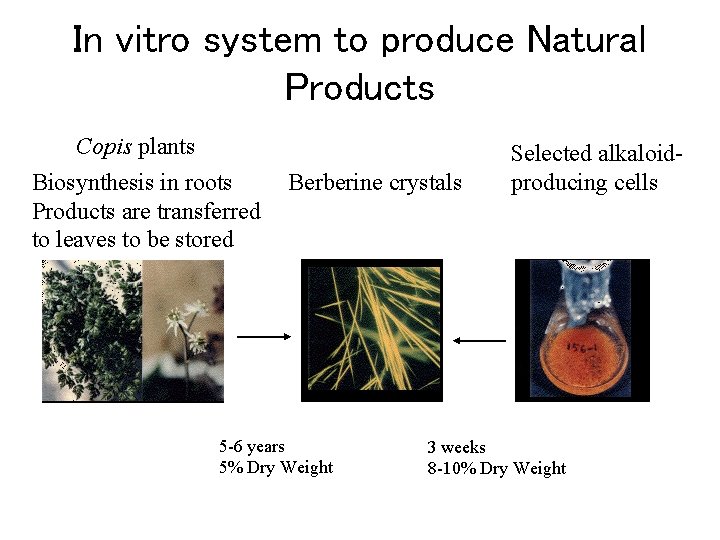
In vitro system to produce Natural Products Copis plants Biosynthesis in roots Products are transferred to leaves to be stored Berberine crystals 5 -6 years 5% Dry Weight Selected alkaloidproducing cells 3 weeks 8 -10% Dry Weight

Tissue culture system has been used to study secondary metabolite synthesis Induction of shikonin biosynthesis Pigments has been used for cosmetics industry Red beet hairy root culture

Topics for natural products 1. Terpenoid (isoprenoid) 2. Alkaloids (N-containing basic compound) 3. Phenylpropanoids and Phenolics Lignins Flavonoids

Terpenoids • Lipid molecules – estimated 22, 000 different types – ~ 400 in tobacco alone – MOST DIVERSE GROUP OF PLANT CHEMICALS • Essential compounds – membrane sterols, carotenoids, groups on chlorophyll, heme a, UQ & PQ, cytokinins, abscisic acid gibberellins – function in photosynthesis, respiration, growth & development, membrane architecture • Secondary metabolites (natural products) – volatiles (essential oils) - major plant odours – tree resins & polyterpenes - insect- & rot-proof – phytoalexins – important in response to environment

Reasons for the greater diversity in plan terpenoids • Mode of connections of C 5 units • Length of connected C 5 units • Diverse cyclization reaction • Various secondary modifications
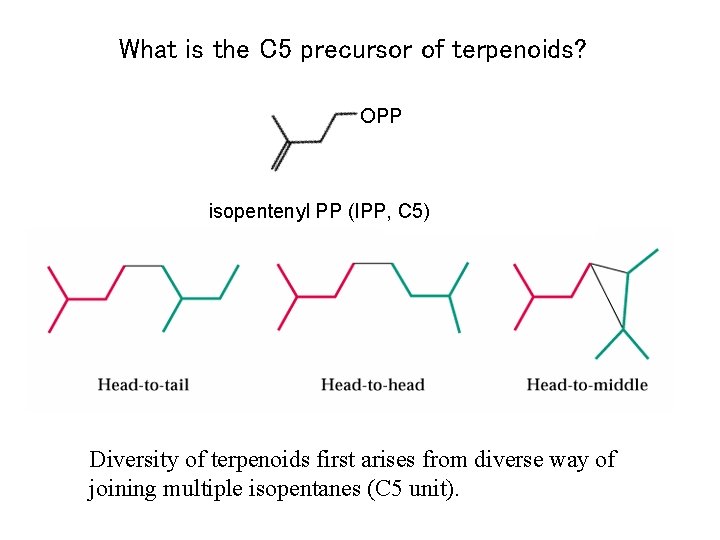
What is the C 5 precursor of terpenoids? OPP isopentenyl PP (IPP, C 5) Diversity of terpenoids first arises from diverse way of joining multiple isopentanes (C 5 unit).
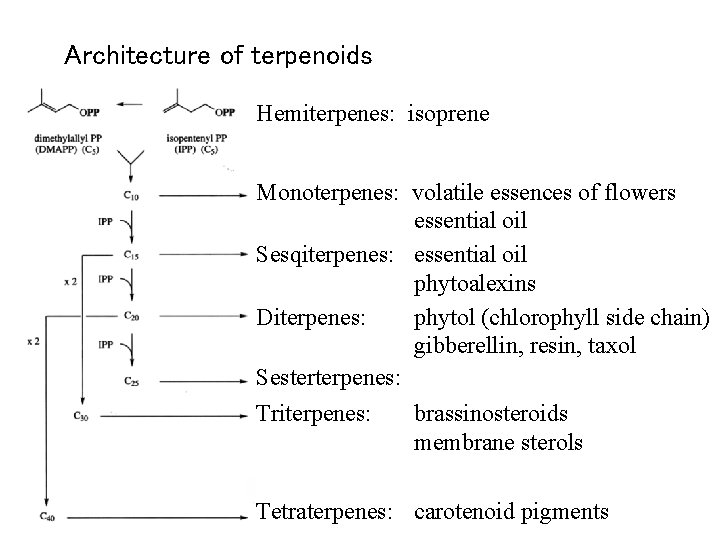
Architecture of terpenoids Hemiterpenes: isoprene Monoterpenes: volatile essences of flowers essential oil Sesqiterpenes: essential oil phytoalexins Diterpenes: phytol (chlorophyll side chain) gibberellin, resin, taxol Sesterterpenes: Triterpenes: brassinosteroids membrane sterols Tetraterpenes: carotenoid pigments
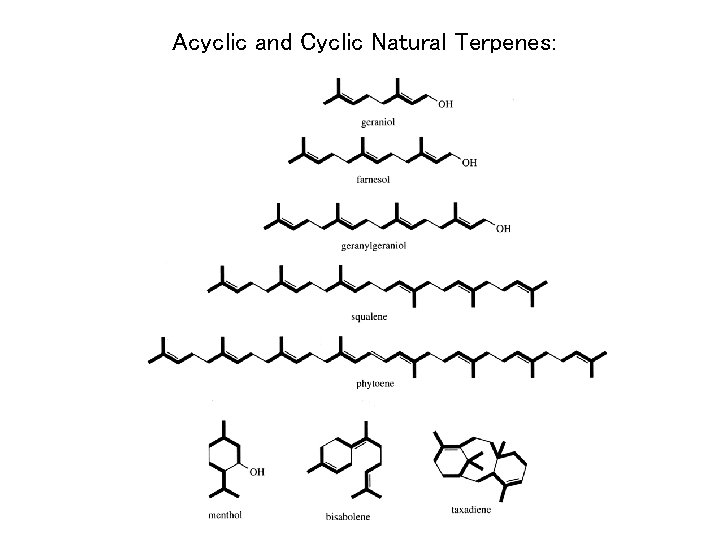
Acyclic and Cyclic Natural Terpenes:

IPP synthesised by 2 different pathways MVA pathway MEP pathway Archaebacteria, fungi and animals Most eubacteria (cytosol and ER) (plastid) Monoterpenes Diterpenes Cytokinins Abscisic acid Gibberellins Carotenoids Phytol Plastoquinone Tocopherols Brassinosteroids Phytosterols Phytoalexins Prenyl groups Ubiquinone (mitos) • Most organisms only use one of the two pathways for the biosynthesis of their precursors. • Plants use both the MEP pathway and the MVA pathway for isoprenoid biosynthesis, although they are localized in different compartments
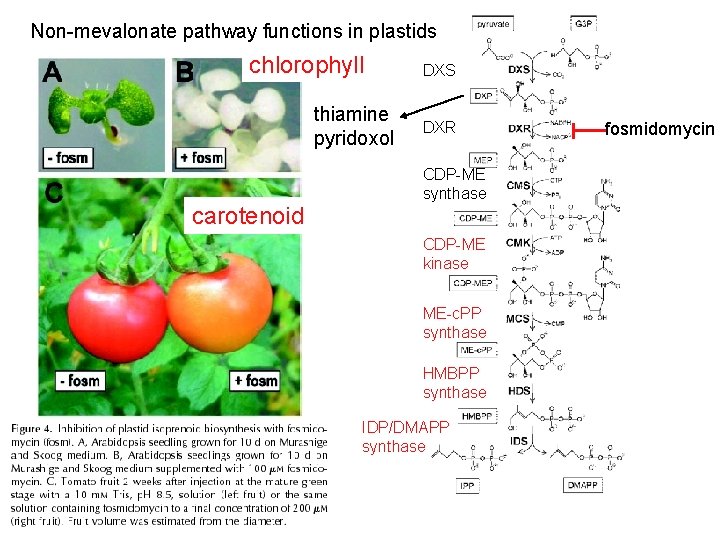
Non-mevalonate pathway functions in plastids chlorophyll thiamine pyridoxol carotenoid DXS DXR CDP-ME synthase CDP-ME kinase ME-c. PP synthase HMBPP synthase IDP/DMAPP synthase fosmidomycin
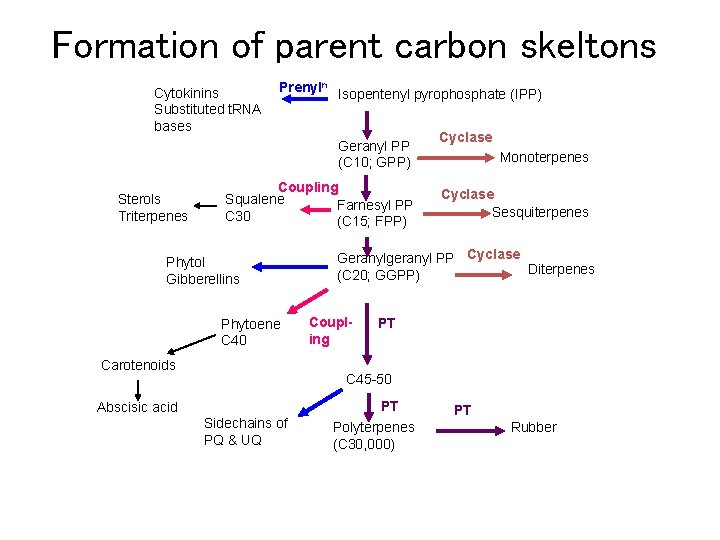
Formation of parent carbon skeltons Cytokinins Substituted t. RNA bases Prenyln Isopentenyl pyrophosphate (IPP) Geranyl PP (C 10; GPP) Sterols Triterpenes Coupling Squalene Farnesyl PP C 30 (C 15; FPP) Phytol Gibberellins Phytoene C 40 Carotenoids Abscisic acid Cyclase Monoterpenes Cyclase Sesquiterpenes Geranylgeranyl PP Cyclase Diterpenes (C 20; GGPP) Coupling PT C 45 -50 PT Sidechains of PQ & UQ Polyterpenes (C 30, 000) PT Rubber
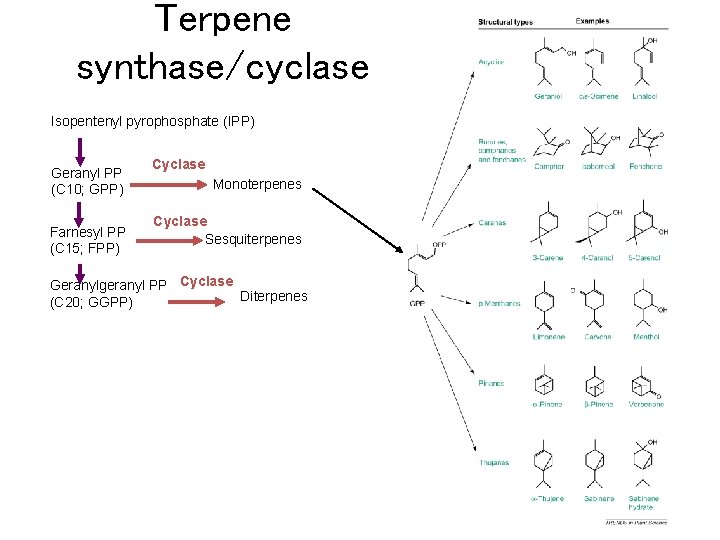
Terpene synthase/cyclase Isopentenyl pyrophosphate (IPP) Geranyl PP (C 10; GPP) Farnesyl PP (C 15; FPP) Cyclase Monoterpenes Cyclase Sesquiterpenes Geranylgeranyl PP Cyclase Diterpenes (C 20; GGPP)
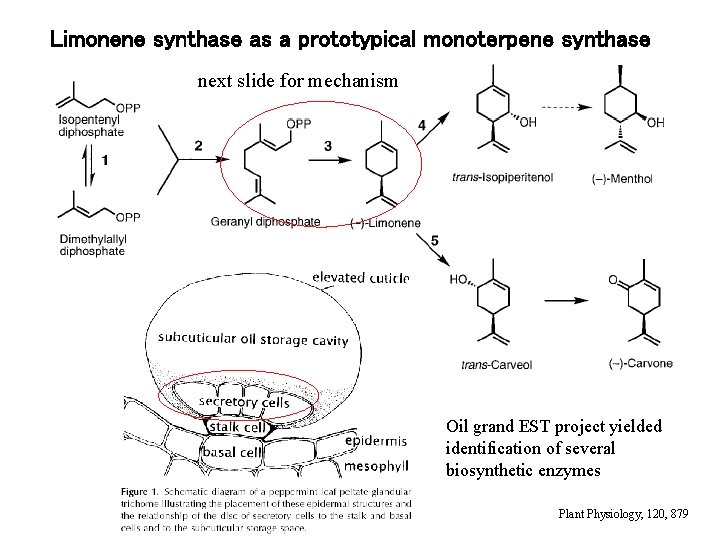
Limonene synthase as a prototypical monoterpene synthase next slide for mechanism Oil grand EST project yielded identification of several biosynthetic enzymes Plant Physiology, 120, 879

Modification of (peppermint) P 450 limonene to menthol (spearmint) Dehydrogenease P 450 menthofuran Isomerase synthase Dehydrogenease Reductase Plant Physiol 122, 215

Manipulating Peppermint Oil Synthesis • • Plant WT DXR 6 MFS 1 Introduction of sense DXR increases the flux of MEP pathway – Increase of up to 50% essential oil Antisense menthofuran synthase decreases by product of menthol synthesis – Decrease of 50% menthanofuran Oil weight 1. 8 2. 6 1. 7 Menthol(%) Menthofuran (mg/g FW) 6. 9 12. 7 23. 2 16. 8 15. 7 2. 5 PNAS 98, 8915

Alkaloids Definition • Pharmacologically active, nitrogen-containing basic compounds of plant origin • Found in about 20 % of plant species • Many of them serves as a chemical defense of plants against herbivores – Many alkaloids are toxic to insects (nicotine, caffeine, etc) – Herbivory stimulates biosynthesis • Ingredients of medicinal plants – Pharmaceuticals – Narcotics – Stimulants – Poisons Ref: Alkaloid biosynthesis in plants Ammu. Rev. Plant Physiol. Plant Mol. Biol. (2001), 52, 29 -66

Four classes of alkaloids Terpenoid-indole alkaloid (Trp) Benzylisoquinoline alkaloid (Tyr) Tropane alkaloid (SAM) Purine alkaloid
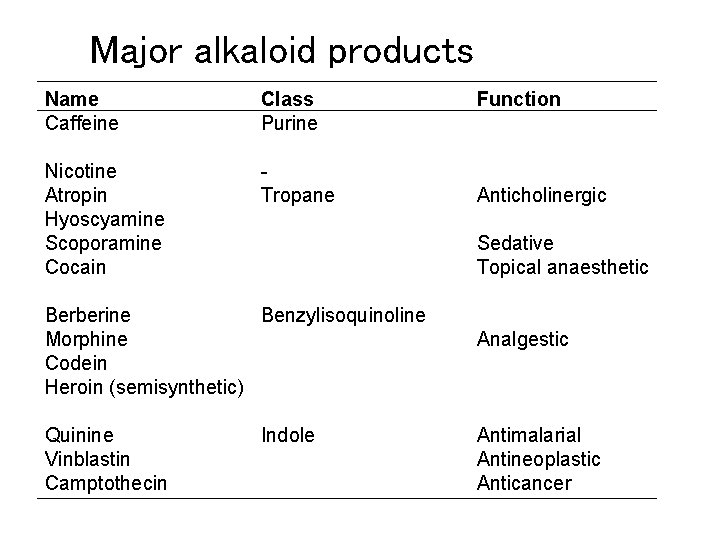
Major alkaloid products Name Caffeine Class Purine Nicotine Atropin Hyoscyamine Scoporamine Cocain Tropane Anticholinergic Sedative Topical anaesthetic Berberine Benzylisoquinoline Morphine Codein Heroin (semisynthetic) Quinine Vinblastin Camptothecin Function Indole Analgestic Antimalarial Antineoplastic Anticancer
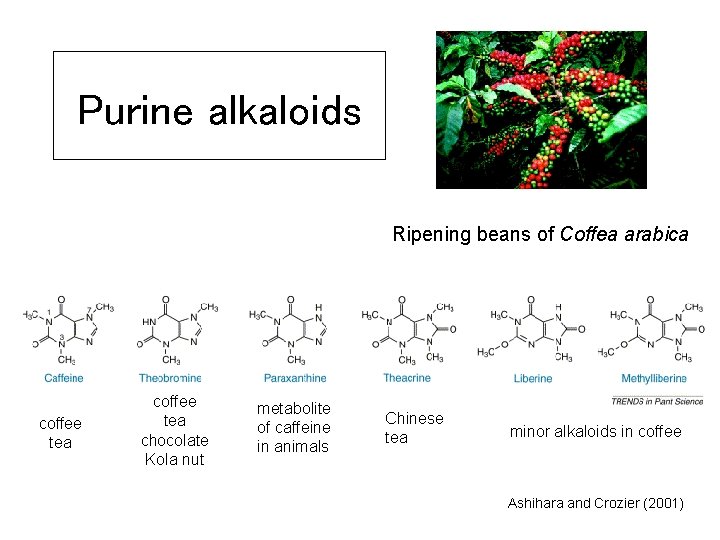
Purine alkaloids Ripening beans of Coffea arabica coffee tea chocolate Kola nut metabolite of caffeine in animals Chinese tea minor alkaloids in coffee Ashihara and Crozier (2001)
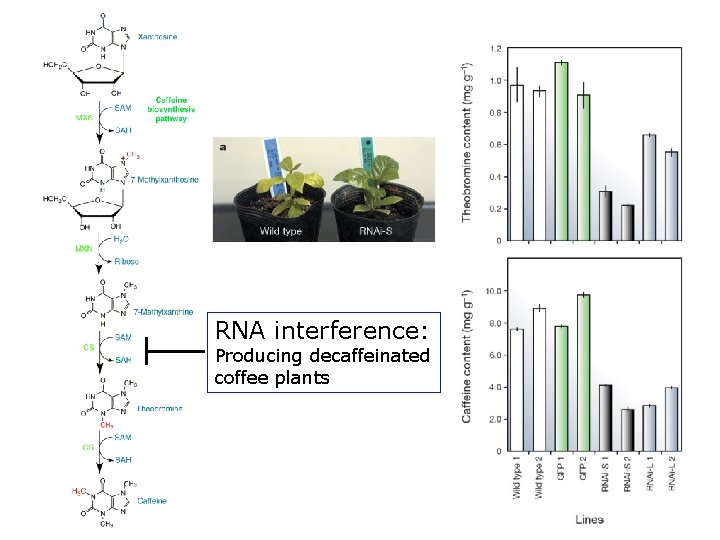
RNA interference: Producing decaffeinated coffee plants
Phenylpropanoids and phenolics • Aromatic metabolites that possess one or more “acidic”-OH groups attachd to the phenyl ring. • Major classes – Lignin/Lignans • Polymeric (lignins) structure that reinforce cell wall • Unique to land plants and not found in aquatic plants – Hydrolizable tannins: polyphenols – Flavonoids: most diverse group, includes anthocyanin, anthocyanidins, isoflavonoids, etc. – Condensed tannins – Coumarins: defense chemicals, toxic to mammals. – Stilbenes: antifungal property, target of biotechnology. – Suberin: Structures of polyaromatic, hydrophobic layer alternated with phenolic hydrophillic layer
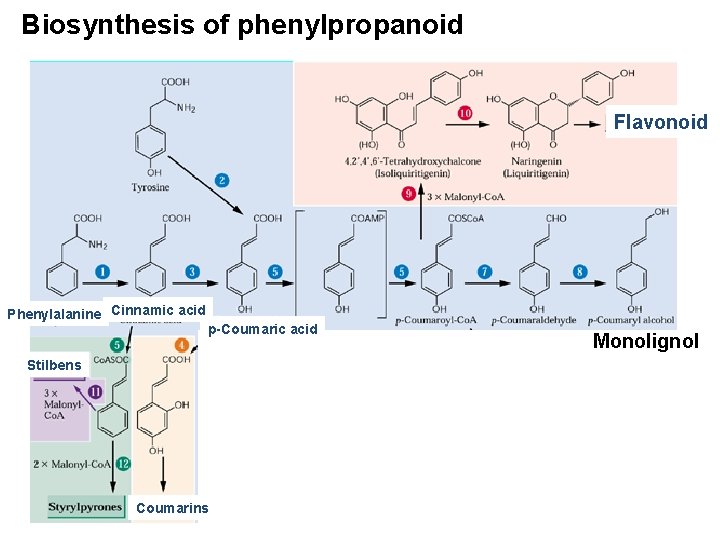
Biosynthesis of phenylpropanoid Flavonoid Phenylalanine Cinnamic acid p-Coumaric acid Stilbens Coumarins Monolignol
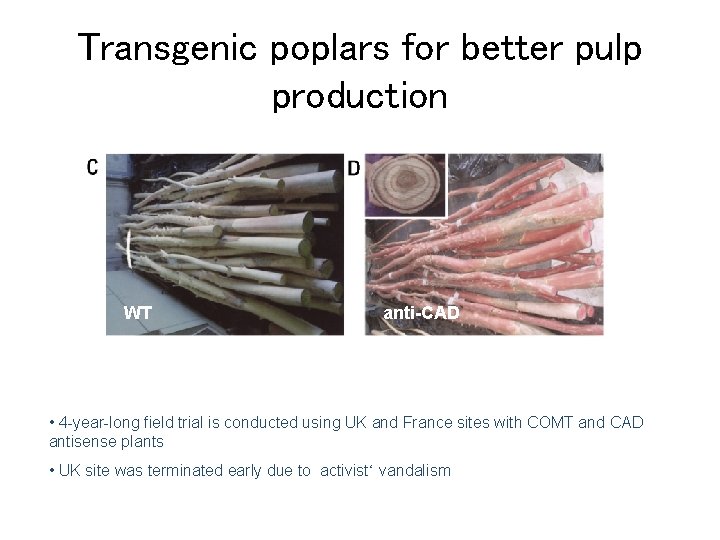
Transgenic poplars for better pulp production WT anti-CAD • 4 -year-long field trial is conducted using UK and France sites with COMT and CAD antisense plants • UK site was terminated early due to activist‘ vandalism
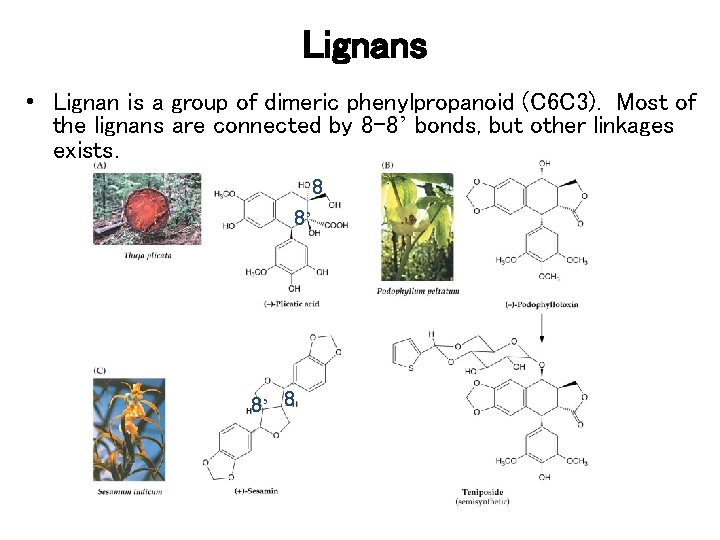
Lignans • Lignan is a group of dimeric phenylpropanoid (C 6 C 3). Most of the lignans are connected by 8 -8’ bonds, but other linkages exists. 8 8’ 8’ 8
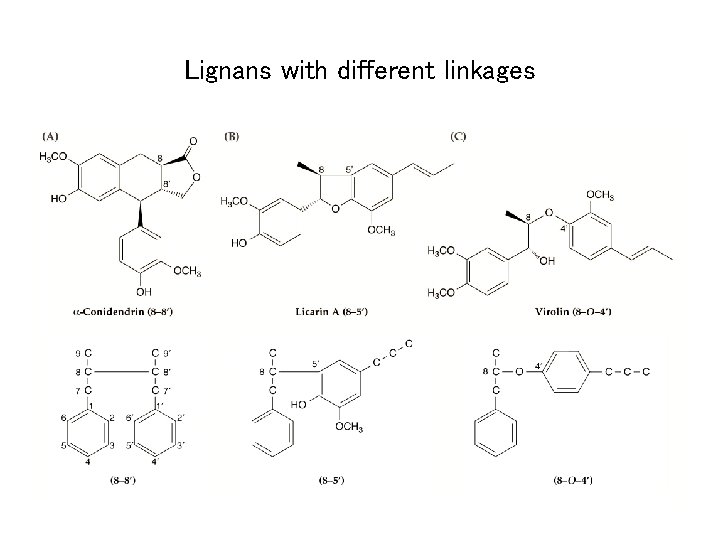
Lignans with different linkages

Lignans are absorbed by the digestive system and can protect against breast and prostate cancer
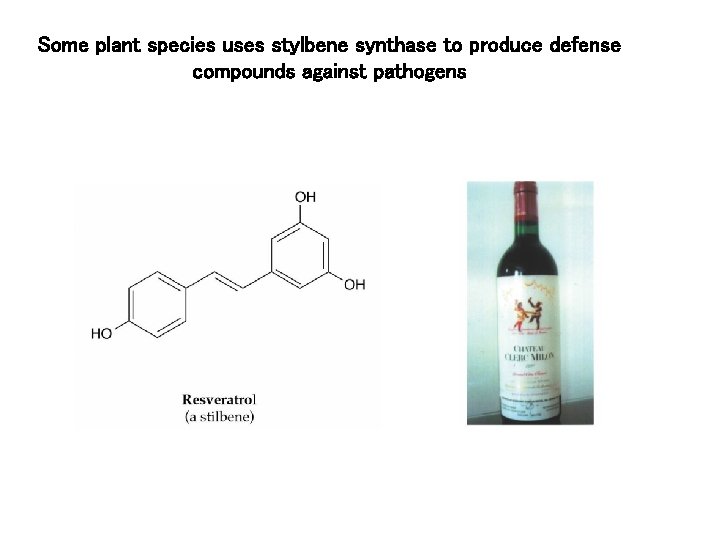
Some plant species uses stylbene synthase to produce defense compounds against pathogens
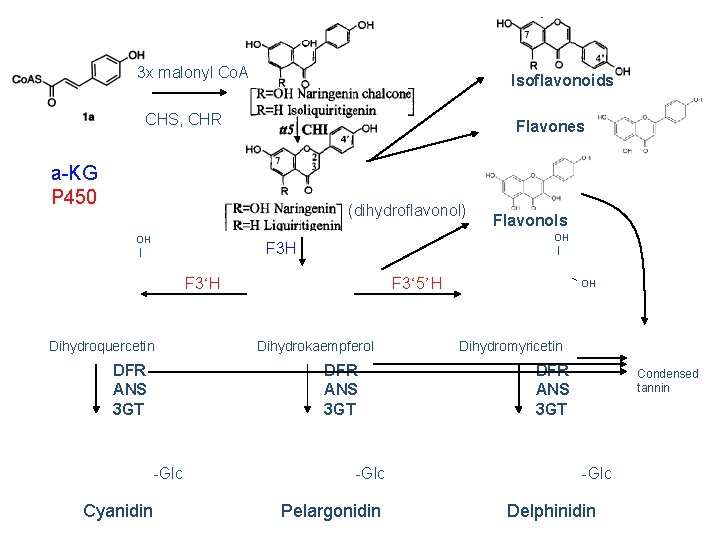
3 x malonyl Co. A Isoflavonoids CHS, CHR Flavones a-KG P 450 (dihydroflavonol) OH | F 3 H F 3‘H Dihydroquercetin DFR ANS 3 GT Cyanidin F 3‘ 5’H Dihydrokaempferol DFR ANS 3 GT -Glc Flavonols -Glc Pelargonidin OH Dihydromyricetin DFR ANS 3 GT Condensed tannin -Glc Delphinidin
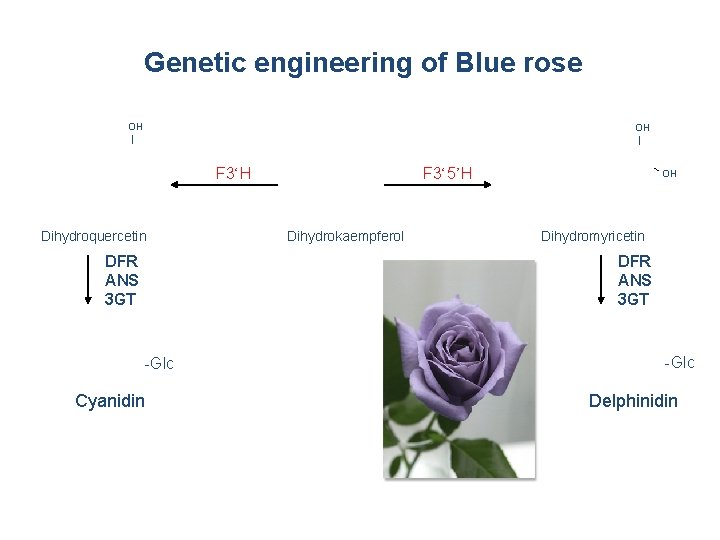
Genetic engineering of Blue rose OH | F 3‘H Dihydroquercetin DFR ANS 3 GT F 3‘ 5’H Dihydrokaempferol OH Dihydromyricetin DFR ANS 3 GT -Glc Cyanidin -Glc Delphinidin

Discussion topics • Controversy about genetically modifying the biochemical profile of plants for consumption
- Slides: 34