Science of Biology Biology is the study of

Science of Biology âBiology is the study of living systems âEasy to say, but what does this mean? âAnything alive is an object of study âCan study living systems at many levels â Cells âMolecules â Assemblies of cells âPopulations of cells âPhysiology of multicellular organisms â Individual organisms â Populations of organisms

Characteristics of Life 1. Complexity and organization 2. Respond to environment (homeostasis) 3. Growth and metabolism 4. Reproduction/heredity 5. Evolve None of these alone defines life, but together, they are not a bad definition

Levels of Organization âMacromolecule âCells âTissue âOrgan systems âIndividual organisms âPopulations

Elements Found in Living Systems âNo matter what level we look at âLiving systems are made of only 6 elements âCHONPS âAlso trace amounts of some important ions: âNa+, K+, Mg 2+, Ca 2+, Cl-, Fe 3+ âEven smaller amounts of other ions: âZn 2+, Mn 2+ âSounds chemically simple, but hides the complexity of organic compounds
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Periodic Table 1 H 3 4 5 Li Be 12 Na Mg 20 6 14 C 13 21 22 K Ca Sc Ti 23 24 25 37 38 39 40 41 42 43 55 56 57 72 73 74 87 88 89 104 105 26 Fe 2 O B Si Al 11 19 8 9 7 10 F Ne N 15 P 16 17 18 S Cl Ar 27 28 29 30 31 32 33 34 35 36 44 45 46 47 48 49 50 51 52 53 I 54 Xe 75 76 77 78 79 80 81 82 83 84 85 86 107 108 109 110 58 59 60 61 62 63 64 65 66 67 68 69 70 90 91 92 93 94 95 96 97 98 99 100 V Cr Mn Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Cs Ba La Hf Ta W Re Os Ir Fr Ra Ac He Pt Au Hg Tl Pb Bi Po At Rn 71 (Lanthanide Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu series) 101 102 103 (Actinide series) Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Classes of Macromolecules âCarbohydrates âProteins âNucleic Acids âLipids âConsider structure, and briefly, function

Synthesis and Degradation âBiological polymers have simple synthesis and degradation âSynthesis involves loss of water âCondensation (also called dehydration) âDegradation involves adding water âHydrolysis âEquilibrium favors hydrolysis âEnergy required for synthesis âBiological systems exist far from equilibrium
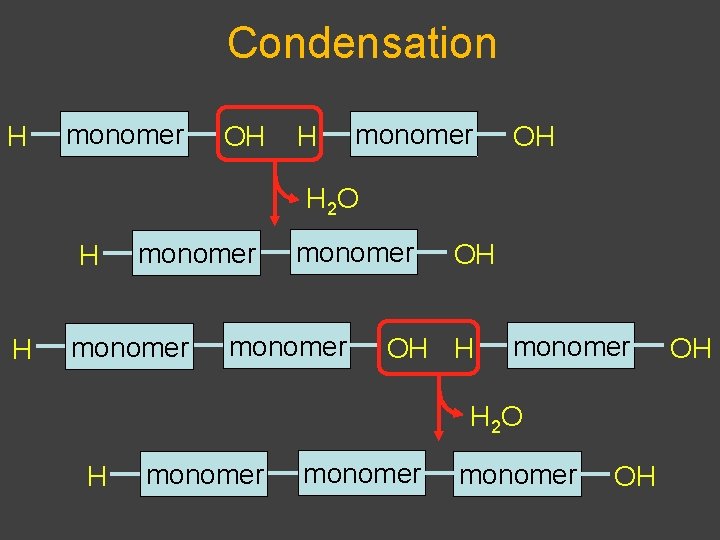
Condensation H monomer OH H 2 O H H monomer OH OH H monomer H 2 O H monomer OH OH
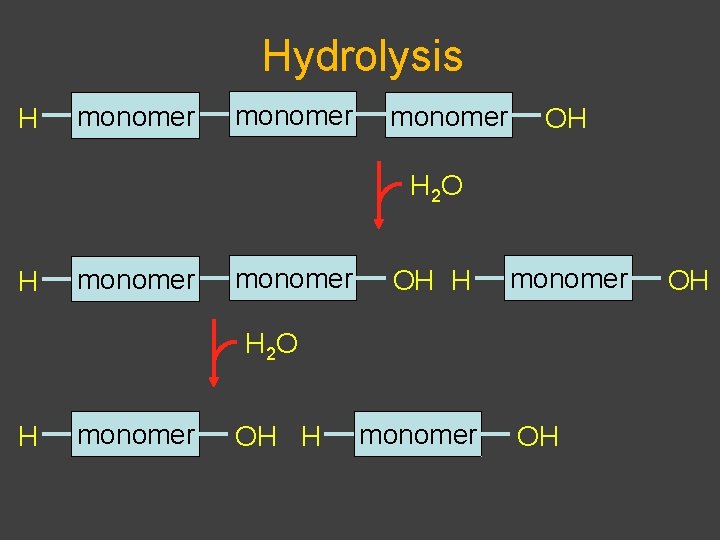
Hydrolysis H monomer OH H 2 O H monomer OH H monomer H 2 O H monomer OH OH

Carbohydrates âMonomers: simple carbohydrates are called simple sugars âMonosaccharides: glucose, fructose âDisaccharides: sucrose, maltose, lactose âPolymers: âCellulose: polymer of glucose -1, 4 bonds âStarch: polymer of glucose -1, 4 bonds âGlycogen: polymer of glucose -1, 4 bonds âFunction: energy storage, structural
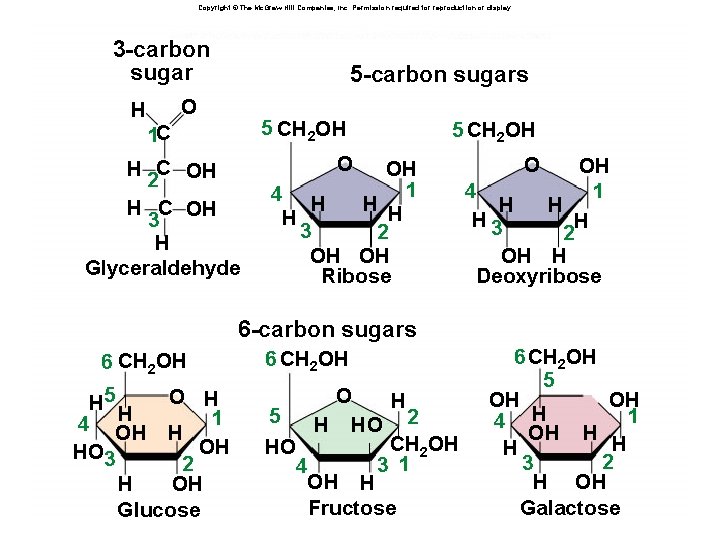
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Fig. 3. 23(TE Art) 5 -carbon sugars 3 -carbon sugar H O 5 CH 2 OH 1 C H C OH 2 H C OH 3 H Glyceraldehyde 5 CH 2 OH O OH 1 4 H H 3 2 OH OH Ribose O OH 4 1 H H H 3 H 2 OH H Deoxyribose 6 -carbon sugars 6 CH 2 OH H 5 H 4 OH HO 3 O H 1 H OH 2 H OH Glucose 6 CH 2 OH O H 5 H HO 2 CH 2 OH HO 3 1 4 OH H Fructose 6 CH 2 OH 5 OH OH 1 4 H OH H 2 3 H OH Galactose

Proteins âMonmers: amino acids (20 unique) âPolymers: polypeptides âProteins: can be multiple polypeptides âFunction: âEnzymes âHormones/neurotransmitters âSurface receptors âMovement: motor proteins âStructural… etc.

Proteins: Structure âMonomer is amino acid âHas carboxylic acid âHas amino group âHas “R” group that defines each aa â 20 different R groups âPolar âNonpolar âacidic (- charge) âbasic (+ charge)
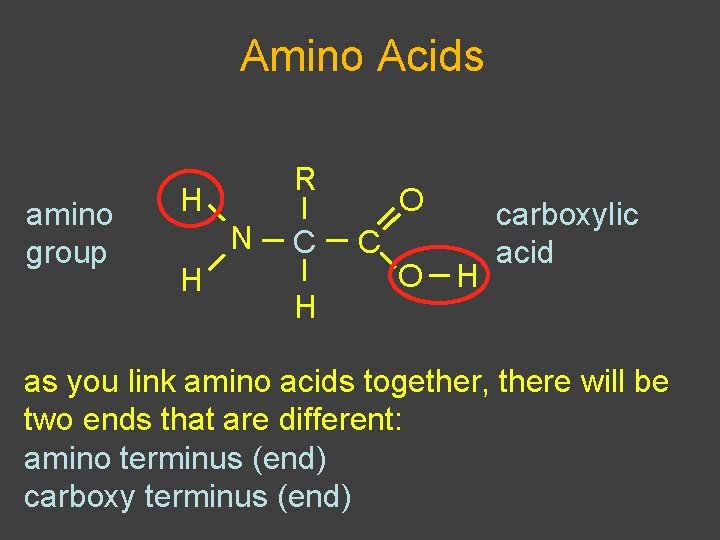
Amino Acids amino group R H N H C H O C O H carboxylic acid as you link amino acids together, there will be two ends that are different: amino terminus (end) carboxy terminus (end)
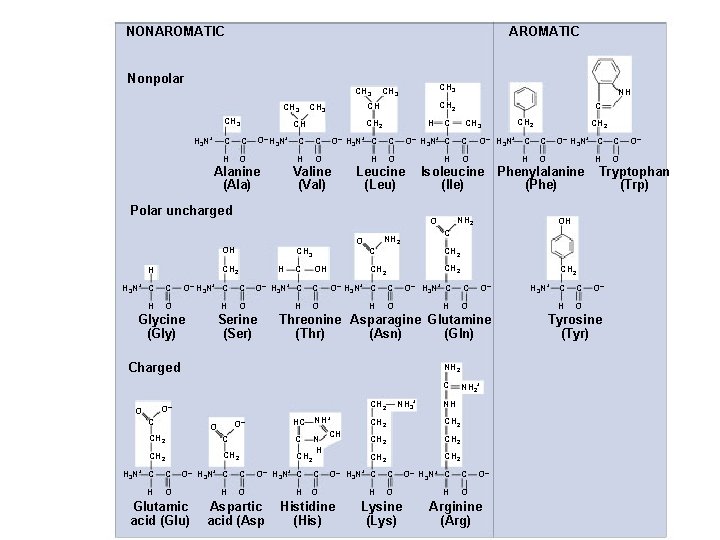
NONAROMATIC Nonpolar CH 3 H 3 N+ C H O O– H 3 N+ CH 2 Alanine (Ala) C C H O– H 3 N+ C C H O Valine (Val) H 3 N+ CH 3 C C H O O– H 3 N+ Glycine (Gly) H C C H O O– Serine (Ser) H 3 N+ C OH C C H O O– H 3 N+ CH 3 O– H 3 N+ C C H O H O Isoleucine Phenylalanine (Ile) (Phe) NH 2 CH 2 H O O– H 3 N+ C C H O NH 2 C C CH 2 H C O– H 3 N+ C C O O Glutamic acid (Glu) + HC NH CH C N H CH 2 O– O CH 2 H 3 N+ C NH 3+ CH 2 O– C CH 2 Threonine Asparagine Glutamine (Thr) (Asn) (Gln) Charged O OH O– H 3 N+ C Aspartic acid (Asp NH 2+ NH CH 2 CH 2 C O– H 3 N+ C C H O H O Histidine (His) Lysine (Lys) O– Arginine (Arg) H 3 N+ C C H O O– Tryptophan (Trp) C CH 2 C NH 2 C CH 2 O– H 3 N+ C O CH 2 H C C Polar uncharged OH C O– H 3 N+ C Leucine (Leu) O NH CH 2 CH CH 3 CH C CH 3 O– Tyrosine (Tyr)

Nucleic acids âStructurally as diverse as proteins âExtremely important: âContain genetic information âTwo basic types: âDeoxyribonucleic Acid (DNA) âRibonucleic Acid (RNA)

Nucleic Acids âMonomers: nucleotides âPolymers: âDNA: double stranded âRNA: single stranded âFunction: âInformation storage and retrieval âDNA stores genetic information âRNA is involved in expression of genetic information

Nucleotides âHave three parts: âRibose sugar (deoxyribose or ribose) âPhosphate âNitrogenous bases âAdenine âGuanine âCytosine âThymine (Uracil) âA, G, C, T (U)
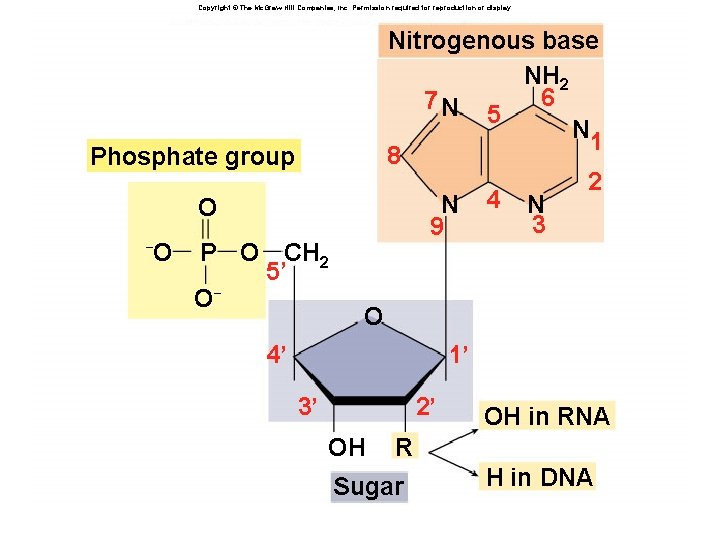
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Nitrogenous base NH 2 6 7 N 5 N 1 8 2 N 4 N 3 9 Fig. 3. 14(TE Art) Phosphate group O – O P O CH 2 5’ O– O 4’ 1’ 3’ 2’ OH OH in RNA R Sugar H in DNA
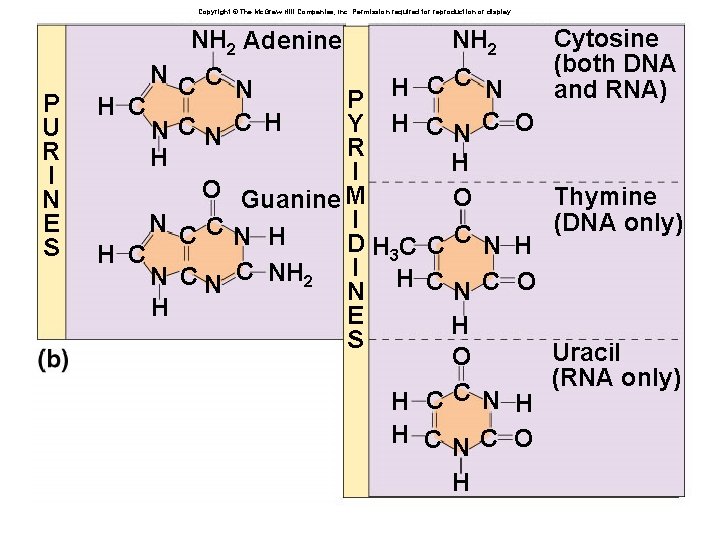
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. N P U R I N E S H C N H NH 2 Adenine CC N P H Y H CN C H R I O Guanine M I C C N H D H 3 C I H C NH 2 N E S NH 2 Cytosine (both DNA and RNA) CC N CN C O H Thymine O (DNA only) C C N H CN C O H Uracil O (RNA only) H CC N H H CN C O H
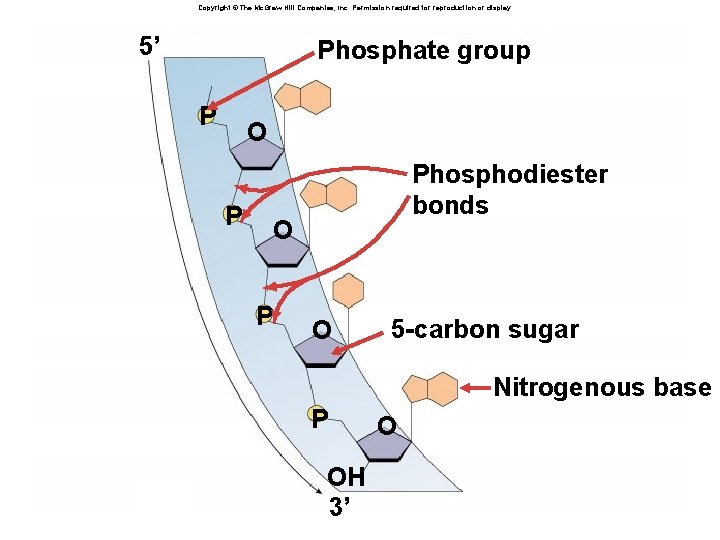
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 5’ Phosphate group Fig. 3. 15 a(TE Art) P O P Phosphodiester bonds O P O 5 -carbon sugar Nitrogenous base P OH 3’ O
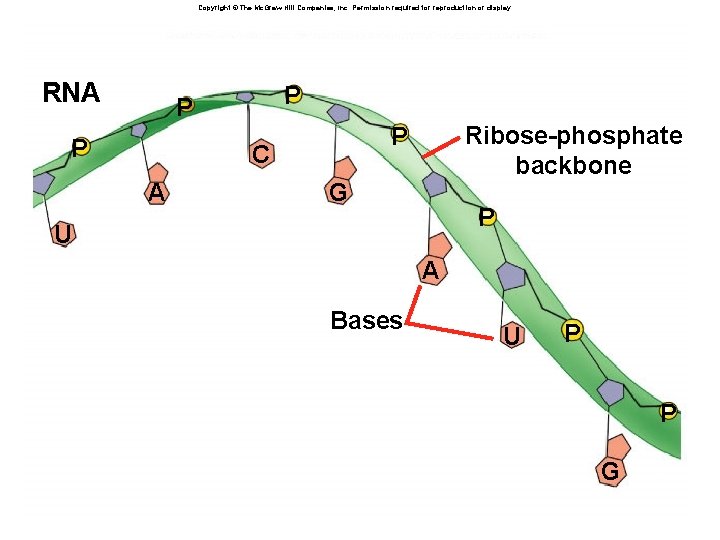
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Fig. 3. 17 b(TE Art) RNA P P P C A Ribose-phosphate backbone P G P U A Bases U P P G
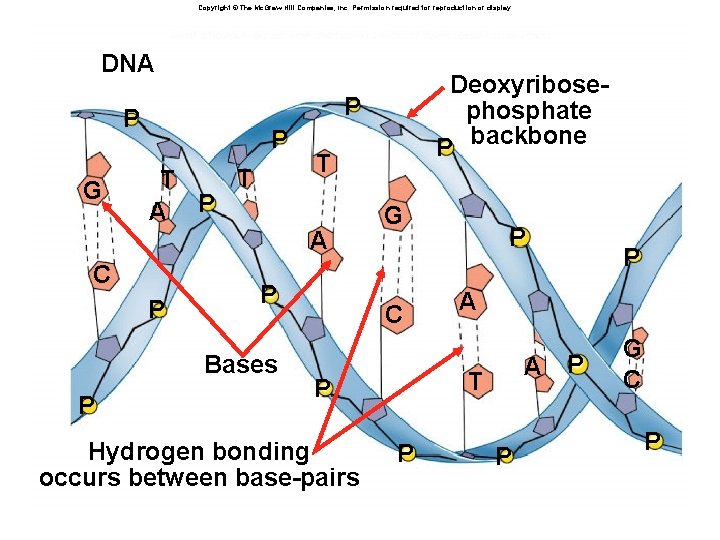
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. DNA Fig. 3. 17 a(TEDeoxyribose. Art) P P G P T T A P T A C P P Bases P phosphate P backbone G C P P A A T P Hydrogen bonding occurs between base-pairs P P P G C P

Lipids âAre polymers, but not like we have been seeing âInclude triglycerides: nonpolar water insoluble (long CH 2 chains) âEnergy storage âFats and oils âSteroids: nonpolar water insoluble âHormones âVitamins âPhospholipids: have charged group

Fatty Acids âFatty acids âLong chains of CH 2 groups with a carboxylic acid at the end âCan be saturated or unsaturated âSaturated have no C=C double bonds: long straight chains âUnsaturated have C=C double bonds: no rotation about double bond âleads to “kinks” in the chain

Triglycerides: Fats and Oils âForm of stored energy for plants and animals âConsists of glycerol with three fatty acids attached âDegree of saturation/unsaturation important âAffects whether they are liquid or solid at room temp âSaturated tend to pack well: solid (fats) âUnsaturated tend not to pack well: liquid (oils)
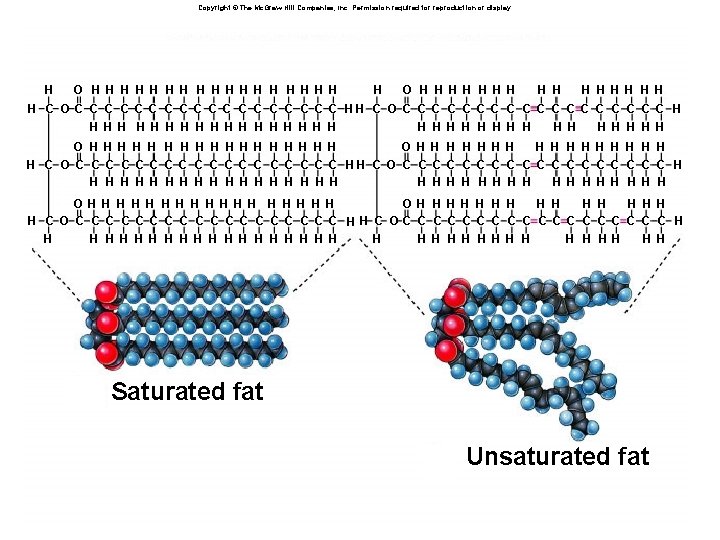
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H O H H H H H H H H C O C C C C C HH C O C C C C C H H H H H H H H H O H H H H H H H H H C O C C C C C C C C C H H H H H H H H H OH H H H H O H H H H C O C C C C C H H C OC C C C C H H H H H H H HH H H Saturated fat Unsaturated fat

Phospholipid: Structure âLike triglyceride but replace fatty acid with phosphate or modified phosphate âPhosphate is charged âLeads to structure with hydrophilic and hydrophobic domains âHow will this behave in water? ? ? âSpontaneously forms bilayers âHydrophilic regions from each layer face water, hydrophobic regions in interior
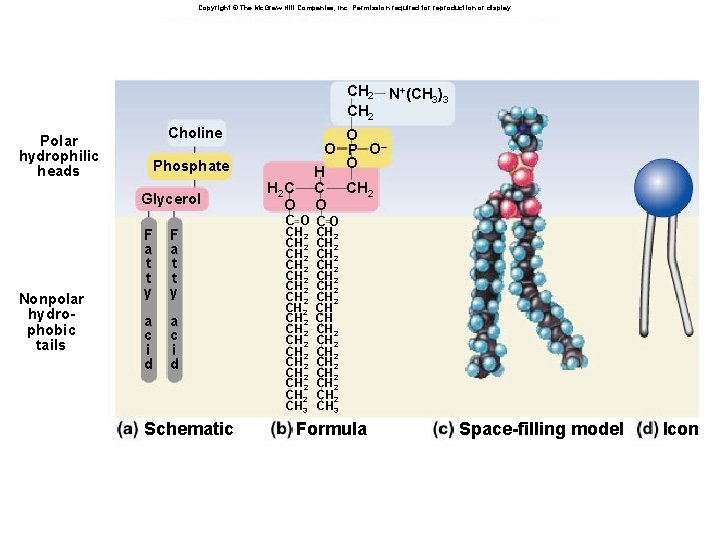
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. CH 2 Polar hydrophilic heads Choline Nonpolar hydrophobic tails O O P O– O H C CH 2 O Phosphate Glycerol F a t t y a c i d Schematic N+(CH 3)3 H 2 C O CO CH 2 CH 2 CH 2 CH 2 CH 3 CH 2 CH 2 CH CH CH 2 CH 2 CH 3 Formula Space-filling model Icon
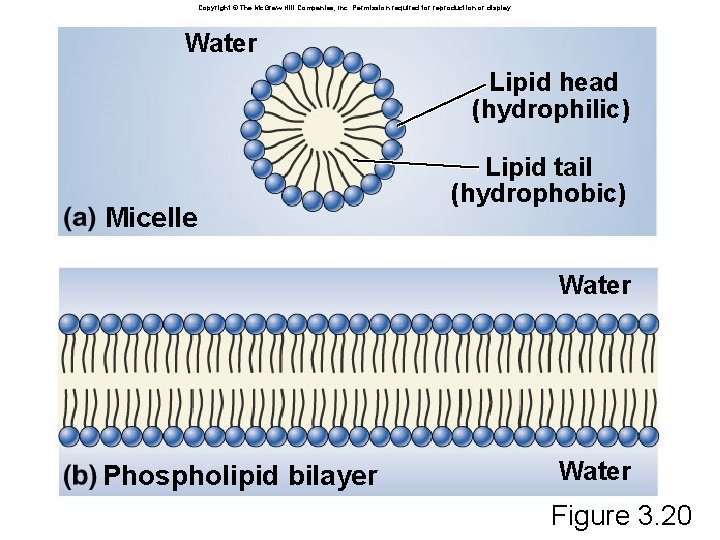
Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Water Lipid head (hydrophilic) Micelle Lipid tail (hydrophobic) Water Phospholipid bilayer Water Figure 3. 20

Phospholipids: Function âBasis for biological membranes âForm spontaneously âLook at function in detail as membrane transport âNow need to build cells out of these macromolecules âBut first, lets relate all of this to FOOD. . .

How Does this Relate to Food? ? ? Starch Cellulose Protein Tryglicerides Glycogen Mayo: more triglycerides

- Slides: 33