Science Matter Energy Systems Chapter 2 System A





















- Slides: 31

Science, Matter, Energy, & Systems Chapter 2

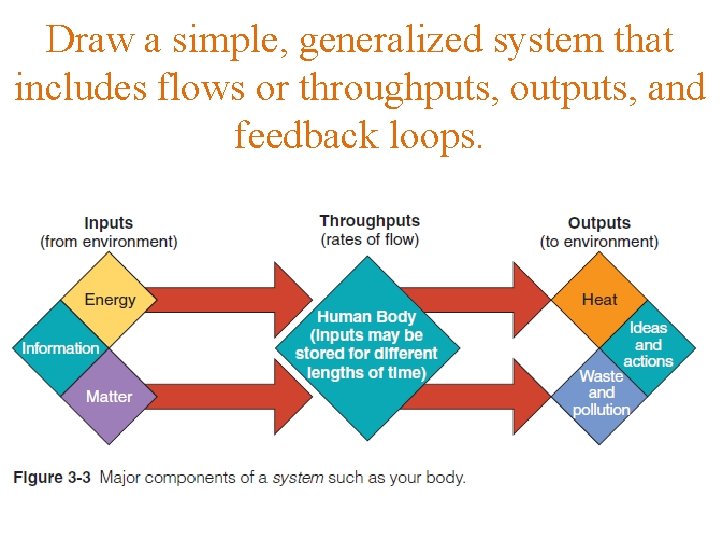
System – A set of components that function and interact in some regular and theoretically predictable manner. Inputs - Matter, energy, or information entering a system. Compare output, throughput. Flows /Throughputs - Rate of flow of matter, energy, or information through a system. Feedback loop Circuit - of sensing, evaluating, and reacting to changes in environmental conditions as a result of information fed back into a system; it occurs when one change leads to some other change, which eventually reinforces or slows the original change. Positive feedback - loop Situation in which a change in a certain direction provides information that causes a system to change further in the same direction. Negative feedback loop - Situation in which a change in a certain direction provides information that causes a system to change less in that direction. Matter - Anything that has mass (the amount of material in an object) and takes up space. On the earth, where gravity is present, we weigh an object to determine its mass. Element - Chemical, such as hydrogen (H), iron (Fe), sodium (Na), carbon (C), nitrogen (N), or oxygen (O), whose distinctly different atoms serve as the basic building blocks of all matter. Two or more elements combine to form compounds that make up most of the world’s matter. Compounds - Combination of atoms, or oppositely charged ions, of two or more different elements held together by attractive forces called chemical bonds.

Atoms - Minute unit made of subatomic particles that is the basic building block of all chemical elements and thus all matter; the smallest unit of an element that can exist and still have the unique characteristics of that element. Ions - Atom or group of atoms with one or more positive () or negative () electrical charges. Molecules - Combination of two or more atoms of the same chemical element (such as O 2) or different chemical elements (such as H 2 O) held together by chemical bonds. Protons - Positively charged particle in the nuclei of all atoms. Each proton has a relative mass of 1 and a single positive charge. Neutrons - Extremely tiny center of an atom, making up most of the atom’s mass. It contains one or more positively charged protons and one or more neutrons with no electrical charge (except for a hydrogen-1 atom, which has one proton and no neutrons in its nucleus). Electrons - Tiny particle moving around outside the nucleus of an atom. Each electron has one unit of negative charge and almost no mass. Organic compound - Compounds containing carbon atoms combined with each other inorganic compound and with atoms of one or more other elements such as hydrogen, oxygen, nitrogen, sulfur, phosphorus, chlorine, and fluorine. All other compounds are called inorganic compounds. Energy - Capacity to do work by performing mechanical, physical, chemical, or electrical tasks or to cause a heat transfer between two objects at different temperatures.

High-throughput economy - The situation in most advanced industrialized countries, in which everincreasing economic growth is sustained by maximizing the rate at which matter and energy resources are used, with little emphasis on pollution prevention, recycling, reuse, reduction of unnecessary waste, and other forms of resource conservation. Low-throughput economy – Economy based on working with nature by recycling and reusing discarded matter, preventing pollution, conserving matter and energy resources by reducing unnecessary waste and use, not degrading renewable resources, building things that are easy to recycle, reuse, and repair, not allowing population size to exceed the carrying capacity of the environment, and preserving biodiversity and ecological integrity.

s a t m e r a Jus sa ta s a rem inder s u J rem er ind Just a in r e d

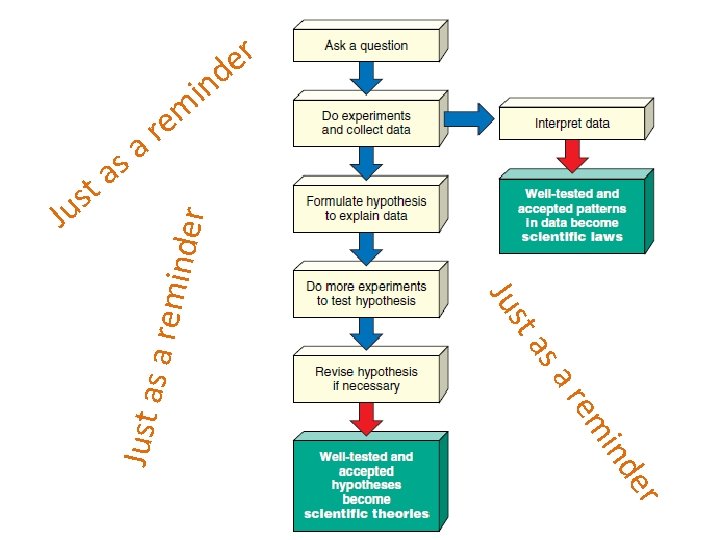
Suppose a scientist observes an abnormality in the growth of robin embryos in a certain area. She knows the area has been sprayed with a pesticide and suspects the chemical may be causing the abnormalities she has observed. To test this hypothesis, the scientist carries out a controlled experiment. She maintains two groups of robin embryos of the same age in the laboratory. Each group is exposed to exactly the same conditions of light, temperature, food supply, and so on, except the embryos in the experimental group are exposed to a known amount of the pesticide in question. The embryos in both groups are then examined over an identical period of time for the (Hint If she finds a significantly larger number of the abnormalities in the abnormality. : Wh at othan experimental group the control group, the results support the idea that the embr ther in facto pesticide is the culprit. yos rs in na ture— elves coprocedure, t thoriginal To be sure no errors occur during the researcher should repeat uld p nothe e l a o borat should repeat the ssiblyother scientists the experiment several times. Ideally one or more ory— expla in the and i experiment independently. n the resul s? )scientist’s Can you find flaws in this experiment that might lead you to question tthe them s conclusions?

Define matter. Anything that has mass (the amount of material in an object) and takes up space. On the earth, where gravity is present, we weigh an object to determine its mass.

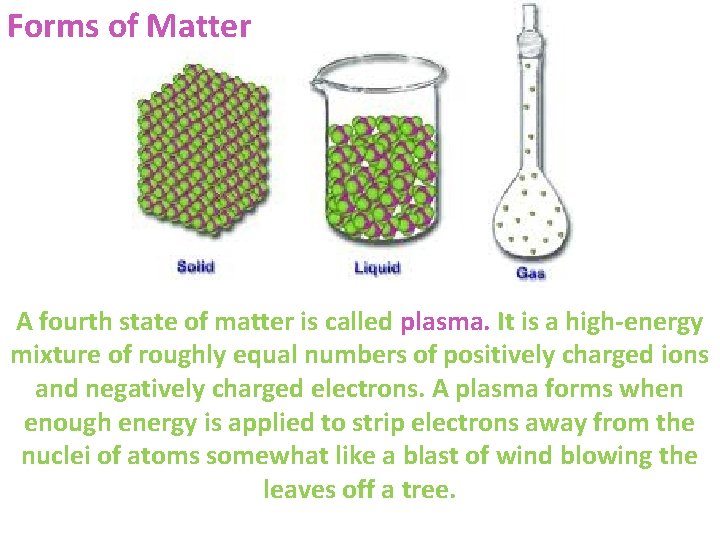
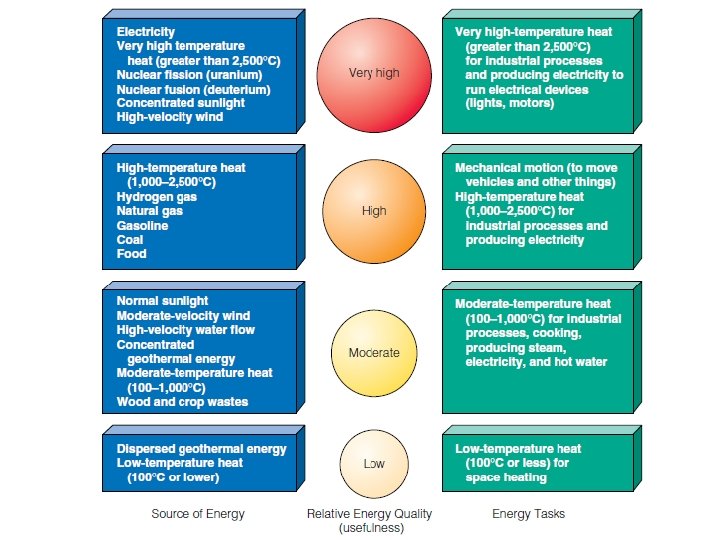
Distinguish between forms & quality of matter Matter quality is a measure of how useful a form of matter is to us as a resource, based on its availability and concentration. The form of matter is based on its structure.

Forms of Matter A fourth state of matter is called plasma. It is a high-energy mixture of roughly equal numbers of positively charged ions and negatively charged electrons. A plasma forms when enough energy is applied to strip electrons away from the nuclei of atoms somewhat like a blast of wind blowing the leaves off a tree.

High-quality matter is concentrated, usually is found near the earth’s surface, and has great potential for use as a matter resource. Low-quality matter is dilute, often is deep underground or dispersed in the ocean or the atmosphere, and usually has little potential for use as a material resource.

Define energy. Capacity to do work by performing mechanical, physical, chemical, or electrical tasks or to cause a heat transfer between two objects at different temperatures.


Distinguish between forms of energy & quality of energy.

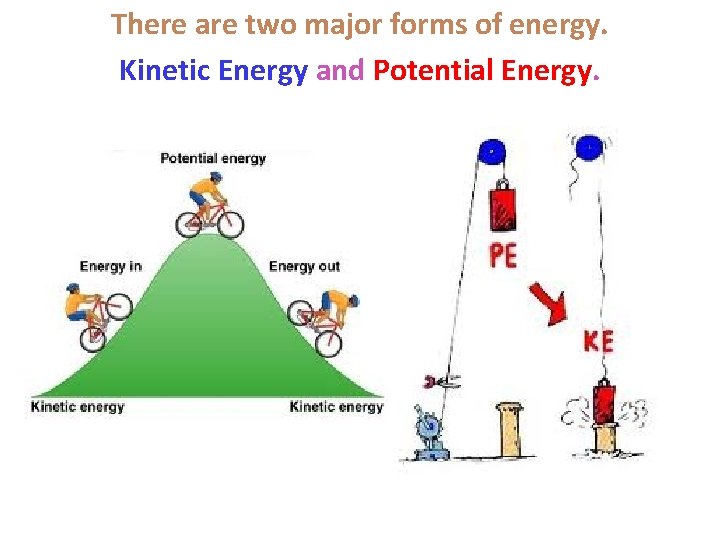
There are two major forms of energy. Kinetic Energy and Potential Energy.

Energy quality is a measure of an energy source’s ability to do useful work. There are two types of energy quality: Ø High quality energy is concentrated and can perform much useful work. Ø By contrast, low-quality energy is dispersed and has little ability to do useful work.


Draw a simple, generalized system that includes flows or throughputs, outputs, and feedback loops.

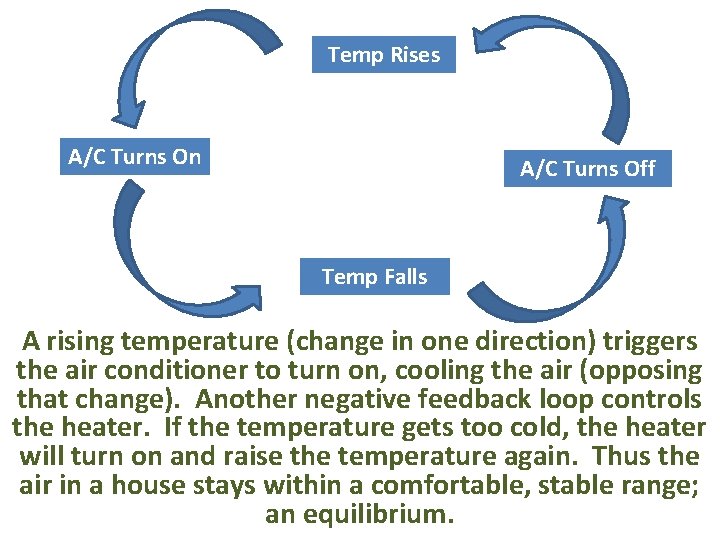
Distinguish between positive and negative feedback loops. A positive feedback loop causes a system to change further in the same direction. A negative, or corrective, feedback loop causes a system to change in the opposite direction. So which feedback loop is more desirable?

Ice Melts Temp Rises If you roll a small snowball down a snow-covered hill, it gets bigger and bigger as it rolls. This kind of self-magnification is aptly called the "snowball effect. " Ironically, the snowball effect encapsulates the urgency with which we must respond to global warming.

Temp Rises A/C Turns On A/C Turns Off Temp Falls A rising temperature (change in one direction) triggers the air conditioner to turn on, cooling the air (opposing that change). Another negative feedback loop controls the heater. If the temperature gets too cold, the heater will turn on and raise the temperature again. Thus the air in a house stays within a comfortable, stable range; an equilibrium.

Define organic compound, and list important organic compounds and their building blocks. Organic compounds - Compounds containing carbon atoms combined with each other and with atoms of one or more other elements such as hydrogen, oxygen, nitrogen, sulfur, phosphorus, chlorine, and fluorine.

■ Hydrocarbons: compounds of carbon and hydrogen atoms. An example is methane (CH 4), the main component of natural gas, and the simplest organic compound. ■ Chlorinated hydrocarbons: compounds of carbon, hydrogen, and chlorine atoms. An example is the insecticide DDT (C 14 H 9 Cl 5). ■ Simple carbohydrates (simple sugars): certain types of compounds of carbon, hydrogen, and oxygen atoms. An example is glucose (C 6 H 12 O 6), which most plants and animals break down in their cells to obtain energy.

Contrast inorganic compounds with organic compounds. Inorganic compounds do not have carbon–carbon or carbon–hydrogen bonds. Some of the inorganic compounds discussed in this book are sodium chloride (Na. Cl), water (H 2 O), nitrous oxide (N 2 O), nitric oxide (NO), carbon monoxide (CO), carbon dioxide (CO 2), nitrogen dioxide (NO 2), sulfur dioxide (SO 2), ammonia (NH 3), hydrogen sulfide (H 2 S), sulfuric acid (H 2 SO 4), and nitric acid (HNO 3).

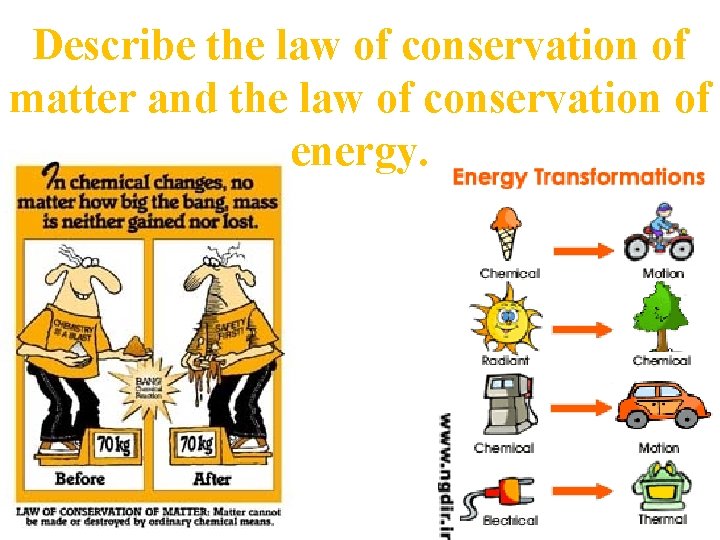
Describe the law of conservation of matter and the law of conservation of energy.

The law of conservation of matter states mater cannot be created nor destroyed. It only changes form. The law of conservation of energy, also known as the first law of thermodynamics: In all physical and chemical changes, energy is neither created nor destroyed, but it may be converted from one form to another. So what is the second law?

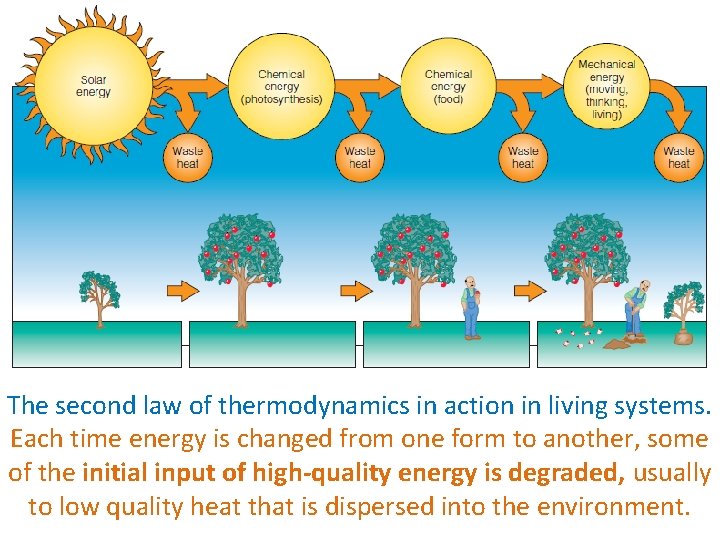
The second law of thermodynamics in action in living systems. Each time energy is changed from one form to another, some of the initial input of high-quality energy is degraded, usually to low quality heat that is dispersed into the environment.

Explain the phrase: “There is no away. ” The law of conservation of matter means there is no “away” as in “to throw away. ” Everything we think we have thrown away is still here somewhere on the planet with us in one form or another.

With your row partner… A tree grows and increases its mass. Explain why this is not a violation of the law of conservation of matter

On your white board… O 2 s a y l l a r Methane (CH 4) gas is the major component of u t a n s natural gas. Write and balance the chemical t s i x e equation for the burning of methane when it n e g y x combines with oxygen gas in the atmosphere to O : t n Hi form carbon dioxide and water. CH 4 + 202 CO 2 + 2 H 2 O

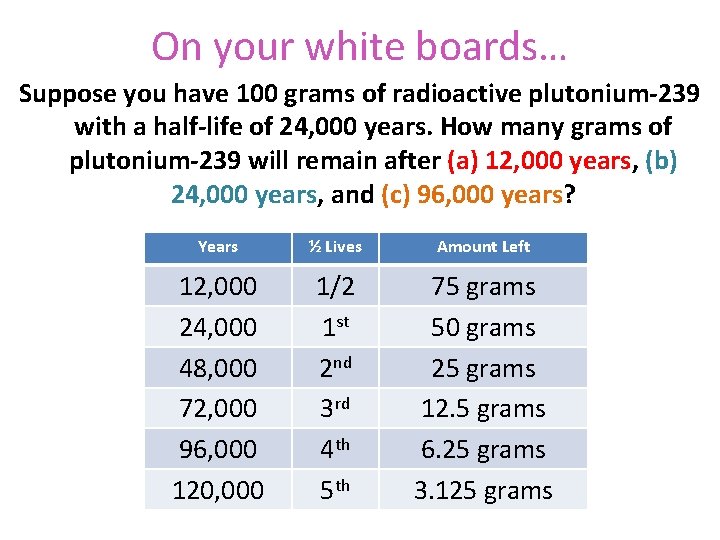
On your white boards… Suppose you have 100 grams of radioactive plutonium-239 with a half-life of 24, 000 years. How many grams of plutonium-239 will remain after (a) 12, 000 years, (b) 24, 000 years, and (c) 96, 000 years? Years ½ Lives Amount Left 12, 000 24, 000 48, 000 72, 000 96, 000 120, 000 1/2 1 st 2 nd 3 rd 4 th 5 th 75 grams 50 grams 25 grams 12. 5 grams 6. 25 grams 3. 125 grams

Summary… Someone wants you to invest money in an automobile engine that will produce more energy than the energy in the fuel you use to run the motor. What is your response? Explain.