Science 1206 Unit 2 Chemistry Periodic Table Videos

Science 1206 Unit 2: Chemistry Periodic Table Videos 1

Chemical Reactions l l l Chemistry: the study of matter, its properties and its changes Matter: anything that has mass and takes up space (energy is not matter). The 3 states of mater: – 2 Solid liquid gas

Pure Substances: have constant composition; all the particles that make up the substance are the same 1. Elements: - the simplest form of matter that can exist under normal conditions - composed of only one kind of atom - cannot be broken into simpler substances by chemical means (heat/electricity) - combine to form other substances 2. Compounds: substances composed of two or more different kinds of atoms can be broken down into simpler substances by chemical means 3

Mixtures l Mixtures have variable compositions – l Homogeneous Mixtures: solutions – have only one visible component l Heterogeneous Mixtures: mechanical mixtures – have 2 or more visible components – 4 Composed of 2 or more substances eg. sand in water, vegetable soup

Pure Substances: Elements and Compounds 5

Mixtures l 6 Homogeneous – uniform look

Mixtures l 7 Heterogeneous Mixtures

A closer look 8

Properties of Matter l Physical Property: – – 9 characteristics of matter, used to identify substances eg. state at room temperature, boiling and melting points, color, solubility, mass, electrical conductivity

Properties of Matter l Chemical Property: characteristic of matter that can be observed when matter undergoes a change in composition (chemical reaction): – – 10 describes "how it reacts“ butane reacts with oxygen to produce carbon dioxide and water
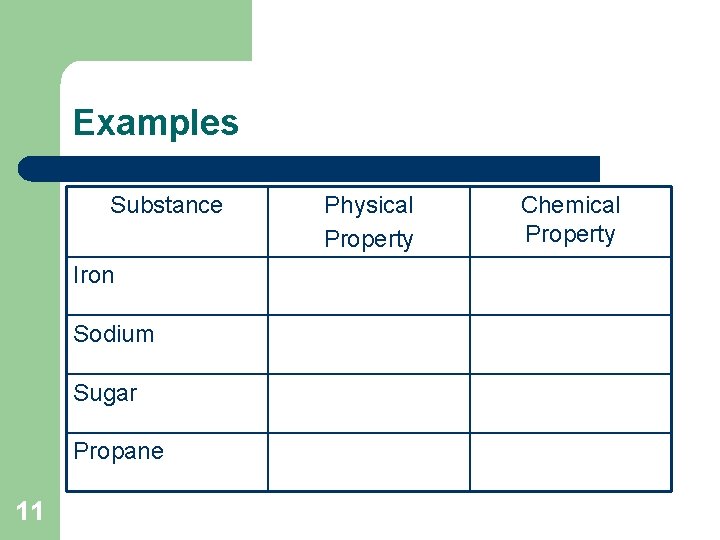
Examples Substance Iron Sodium Sugar Propane 11 Physical Property Chemical Property

Changes in Matter l Physical Change: a change in the size or form of a substance that does not change its composition – 12 cutting, bending, changes in state: boiling, melting, condensing, and solidifying

Changes in Matter l Chemical Change: a chemical reaction; a change in which at least one or more new substances – – 13 (products) are formed. The products have different properties from the starting substances (reactants). Fe(s) + O 2(g) Fe 2 O 3(s) The rust produced has completely different properties from iron and oxygen.

Change in Matter l Evidence of Chemical Change: – – 14 change in color, odor, energy (temperature change, light) bubbles = new gas produced precipitate = new solid produced Hard to reverse

Homework l Read pg. 172 -174 Questions: 1, 2, 4, 7 l Homework Check: l 15
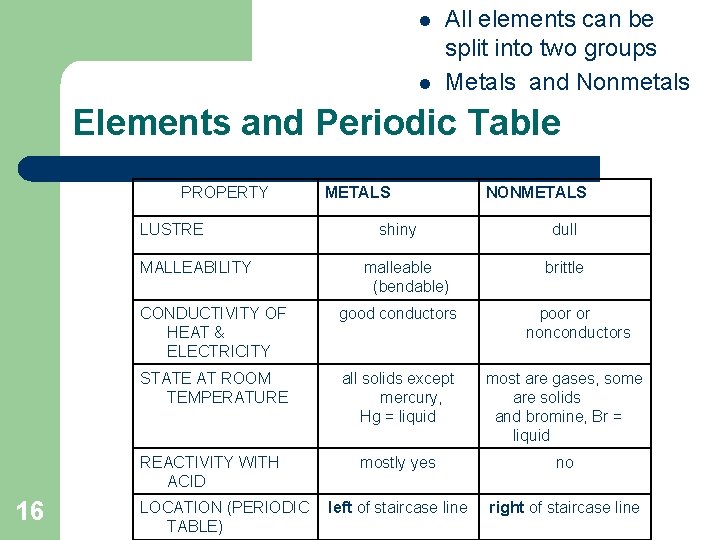
l l All elements can be split into two groups Metals and Nonmetals Elements and Periodic Table PROPERTY LUSTRE MALLEABILITY 16 METALS shiny malleable (bendable) NONMETALS dull brittle CONDUCTIVITY OF HEAT & ELECTRICITY good conductors STATE AT ROOM TEMPERATURE all solids except mercury, Hg = liquid most are gases, some are solids and bromine, Br = liquid REACTIVITY WITH ACID mostly yes no left of staircase line right of staircase line LOCATION (PERIODIC TABLE) poor or nonconductors

Metals and Nonmetals 17

Metalliods l METALLOIDS (Semimetals) – – – 18 elements that have some properties of metals and some properties of nonmetals includes all elements on either side of the staircase line except Al and At also includes one form of Carbon, graphite, which is dull and brittle (nonmetal), but is a good conductor of electricity (metal)

CHEMICAL FAMILIES (GROUPS): l l 19 Elements are grouped on the periodic table according to physical and chemical properties These are called Chemical Families or Groups
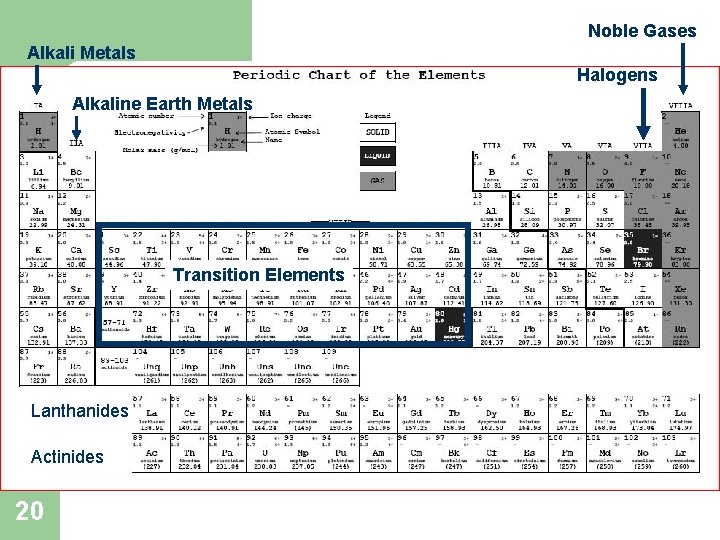
Noble Gases Alkali Metals Halogens Alkaline Earth Metals and Nonmetals Transition Elements Lanthanides Actinides 20

Hydrogen – Special Case l l 21 - the lightest element and most abundant element in the universe - doesn't really belong to any group - it sometimes behaves like an alkali metal, sometimes like a halogen and at other times in its own unique way ie. as an acid

Groups and Periods l Groups (Families) refer to the vertical columns Numbered on top l Periods refer to the horizontal rows l – 22 (Think school periods)

The Atom THE ATOM: The basic building block of all matter Electrically neutral: # of positive charges = # of negative charges composed of 3 types of subatomic particles: PARTICLE 23 SYMBOL RELATIVE CHARGE ACTUAL MASS (g) LOCATION Proton p+ 1+ 1. 67 x 10 -24 nucleus Neutron no 0 1. 67 x 10 -24 nucleus Electron e- 1 - 9. 11 x 10 -28 orbital

Finding the # of Protons • The Atomic Number IS the # of Protons Mass Number/ Finding the # of Electrons • If the element is NEUTRAL, then # protons = # electrons Finding the # of Neutrons • Mass Number = # p + # n • Therefore # n = Mass Number - # p 24
Examples 25

Quantum Mechanics l 26 According to this theory, an electron with a specific energy occupies a region in space (orbital) or electron energy level.

Electron Energy Diagrams l l 27 The number of occupied energy levels in any atom is normally the same as the period number in which the atom appears for the first 3 energy levels, the maximum number of electrons that can be present are 2, 8 and 8 in order of increasing energy (increasing distance from nucleus) a lower energy level is filled with electrons to its maximum before the next level is started. the electrons in the highest (outermost) occupied energy level = valence electrons, which is the same as the group number (for group A elements)
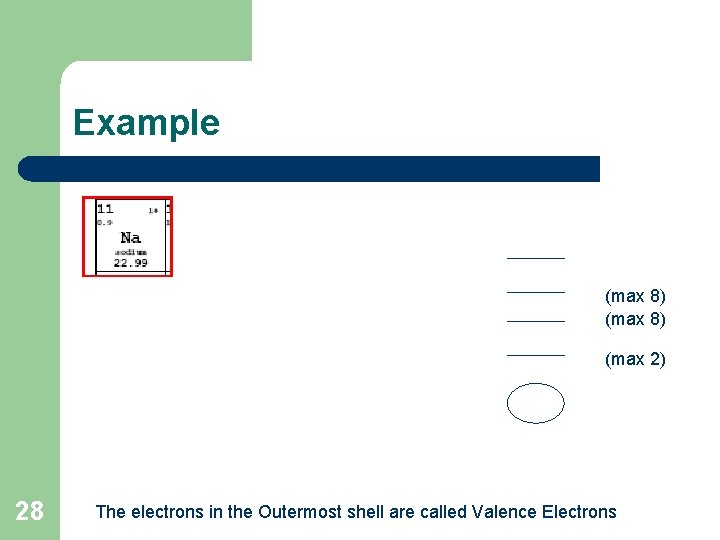
Example (max 8) (max 2) 28 The electrons in the Outermost shell are called Valence Electrons
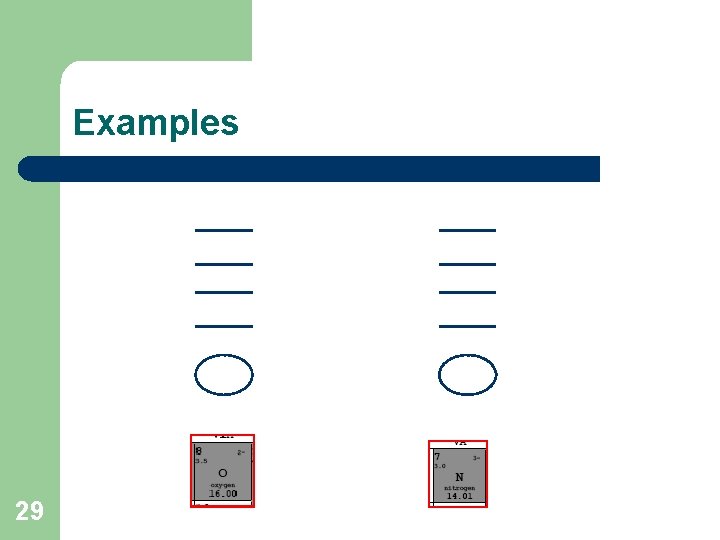
Examples 29

Worksheet l l 30 Complete the worksheet # 1 on pg 12 Checked as homework tomorrow
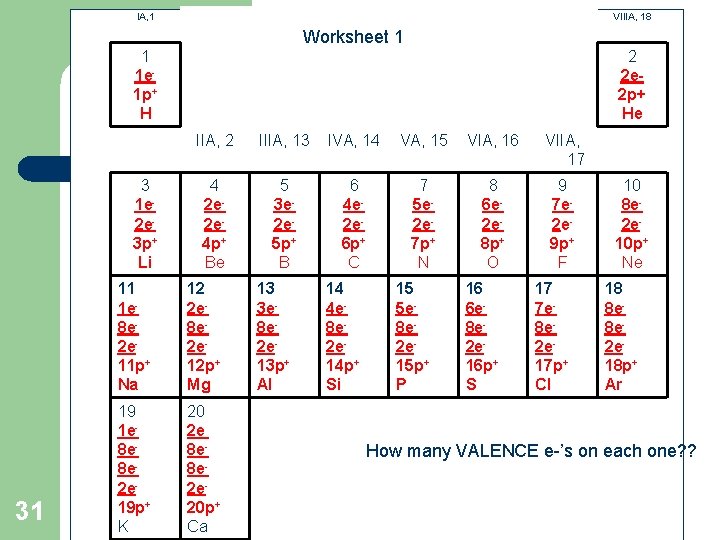
IA, 1 VIIIA, 18 Worksheet 1 1 1 e 1 p+ H 3 1 e 2 e 3 p+ Li 31 2 2 e 2 p+ He IIA, 2 IIIA, 13 IVA, 14 VA, 15 VIA, 16 VIIA, 17 4 2 e 2 e 4 p+ Be 5 3 e 2 e 5 p+ B 6 4 e 2 e 6 p+ C 7 5 e 2 e 7 p+ N 8 6 e 2 e 8 p+ O 9 7 e 2 e 9 p+ F 11 1 e 8 e 2 e 11 p+ Na 12 2 e 8 e 2 e 12 p+ Mg 19 1 e 8 e 8 e 2 e 19 p+ K 20 2 e 8 e 8 e 2 e 20 p+ Ca 13 3 e 8 e 2 e 13 p+ Al 14 4 e 8 e 2 e 14 p+ Si 15 5 e 8 e 2 e 15 p+ P 16 6 e 8 e 2 e 16 p+ S 17 7 e 8 e 2 e 17 p+ Cl 10 8 e 2 e 10 p+ Ne 18 8 e 8 e 2 e 18 p+ Ar How many VALENCE e-’s on each one? ?

STABLE ATOMS (pg. 4 – notes) (pg 188 – Book) l The noble gases are very stable (unreactive) – They all have 8 valence electrons l l Other elements are unstable (reactive) – They have to lose OR gain electrons to find stability – a) Octet Rule: - atoms attempt to obtain 8 valence electrons b) Duet Rule: - atoms attempt to obtain 2 valence electrons - includes H, Li and Be – 32 Valence electrons = electrons in the outermost shell

Ions l When an element loses or gains electrons, they become charged – Sngle atoms: form simple ions (monatomic ions) Na = sodium atom become: l Na+ = sodium ion l – and Cl = chlorine atom Cl- = chloride ion group of atoms: form complex ions (polyatomic ions) l 33 and Ex. N and O can form Atoms = No charge NO 3 - - nitrate ion Ions = Charged

Sodium metal and chlorine gas react to produce Na. Cl, a very stable and unreactive substance, compared to Na (alkali metal) or Cl (halogen). They do so by first forming ions. 34

Finding the charge of an ions 35 1. Determine how many electrons an element must lose/gain 2. Use the periodic table of elements

Positive and Negative Ions Elements that lose electrons form positive ions called Cations l l 36 Elements that gain electrons form negative ions called Anions
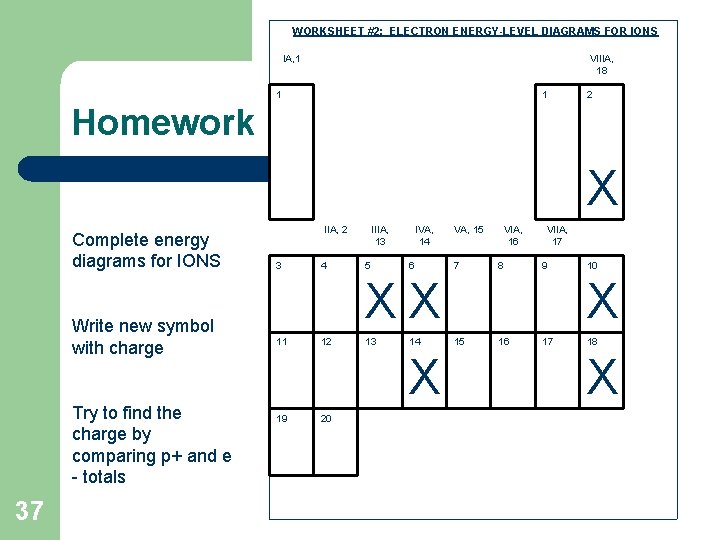
WORKSHEET #2: ELECTRON ENERGY-LEVEL DIAGRAMS FOR IONS IA, 1 VIIIA, 18 1 1 2 Homework X Complete energy diagrams for IONS Write new symbol with charge Try to find the charge by comparing p+ and e - totals 37 IIA, 2 3 4 IIIA, 13 5 IVA, 14 6 VA, 15 7 VIA, 16 8 VIIA, 17 9 XX 11 12 13 14 X 19 20 10 X 15 16 17 18 X

Homework Check l Worksheet # 1 – Energy Diagrams of Atoms Worksheet # 2 – Energy Diagrams of Ions l Today’s Class l – – – 38 Quick recap Naming Ions Biomes Test
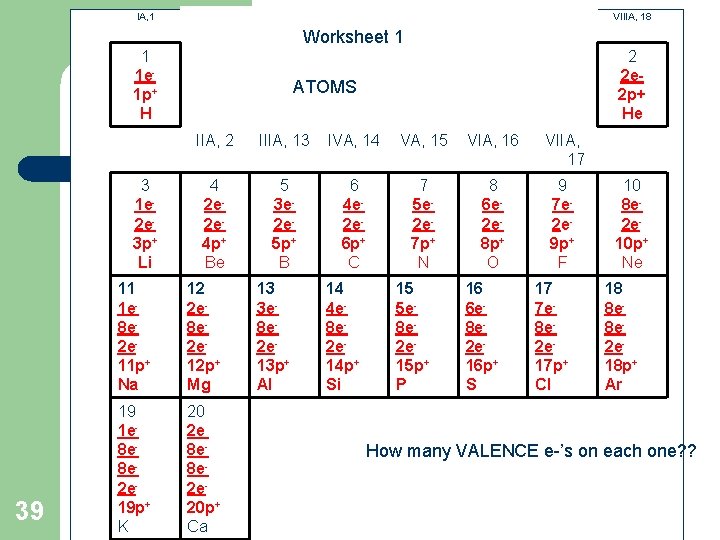
IA, 1 VIIIA, 18 Worksheet 1 1 1 e 1 p+ H 3 1 e 2 e 3 p+ Li 39 2 2 e 2 p+ He ATOMS IIA, 2 IIIA, 13 IVA, 14 VA, 15 VIA, 16 VIIA, 17 4 2 e 2 e 4 p+ Be 5 3 e 2 e 5 p+ B 6 4 e 2 e 6 p+ C 7 5 e 2 e 7 p+ N 8 6 e 2 e 8 p+ O 9 7 e 2 e 9 p+ F 11 1 e 8 e 2 e 11 p+ Na 12 2 e 8 e 2 e 12 p+ Mg 19 1 e 8 e 8 e 2 e 19 p+ K 20 2 e 8 e 8 e 2 e 20 p+ Ca 13 3 e 8 e 2 e 13 p+ Al 14 4 e 8 e 2 e 14 p+ Si 15 5 e 8 e 2 e 15 p+ P 16 6 e 8 e 2 e 16 p+ S 17 7 e 8 e 2 e 17 p+ Cl 10 8 e 2 e 10 p+ Ne 18 8 e 8 e 2 e 18 p+ Ar How many VALENCE e-’s on each one? ?
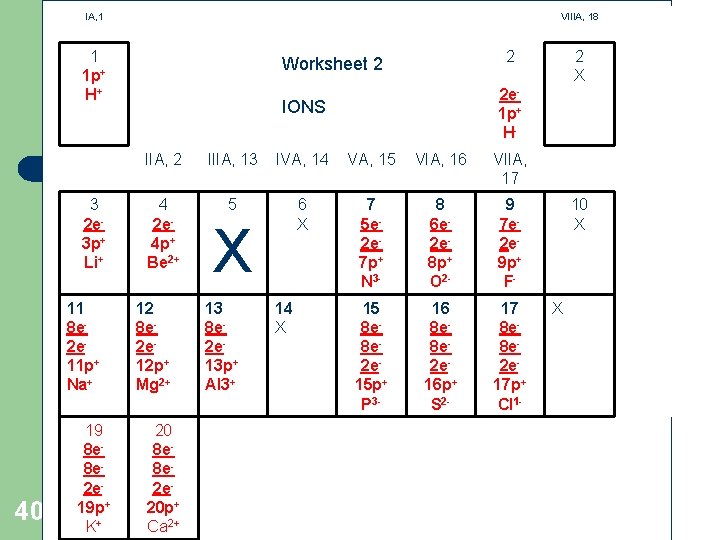
IA, 1 VIIIA, 18 1 1 p+ H+ 3 2 e 3 p+ Li+ 11 8 e 2 e 11 p+ Na+ 40 19 8 e 8 e 2 e 19 p+ K+ 2 Worksheet 2 2 e 1 p+ H- IONS IIA, 2 IIIA, 13 IVA, 14 VA, 15 VIA, 16 VIIA, 17 4 2 e 4 p+ Be 2+ 5 6 X 7 5 e 2 e 7 p+ N 3 - 8 6 e 2 e 8 p+ O 2 - 9 7 e 2 e 9 p+ F- 15 8 e 8 e 2 e 15 p+ P 3 - 16 8 e 8 e 2 e 16 p+ S 2 - 17 8 e 8 e 2 e 17 p+ Cl 1 - 12 8 e 2 e 12 p+ Mg 2+ 20 8 e 8 e 2 e 20 p+ Ca 2+ X 13 8 e 2 e 13 p+ Al 3+ 14 X 2 X 10 X X

A quick review l Elements – – l Compounds – l Pure substances that have more then 1 type of atom Mixtures – 41 Pure substances composed of 1 type of atom Everything on the periodic table Substances with made of more then one compound

A quick review l l 42 Physical properties Chemical properties Physical Changes Chemical Changes
Previously l The periodic table – – – l The Atom – – – 43 Metals and Nonmetals Groups/Families Periods] Protons Electrons Neutrons Mass Number/

Last Class l Energy Levels (2, 8, 8 rules) l Ions: elements that do not have the same number of protons and electrons – – They have a charge Ex: 2+ Ca 44

Ions l Cations : Positive Ions – l Anions : Negative Ions – 45 Formed when elements lose electrons Formed with elements gain electrons

Naming Ions (Notes: pg 5) Write this down!! l To name ions: – Cations l l element name + the word “ion” Ex: Try: Na = sodium atom – Na+ = sodium ion – 46 Mg Mg 2+

Naming Ions (Notes: pg 5) l To name ions: – Anions l l stem of element name + “ide” + the word “ion” Ex: Try: Cl = chlorine atom – Cl- = chloride ion F F- – More Practice: S 47 O N P
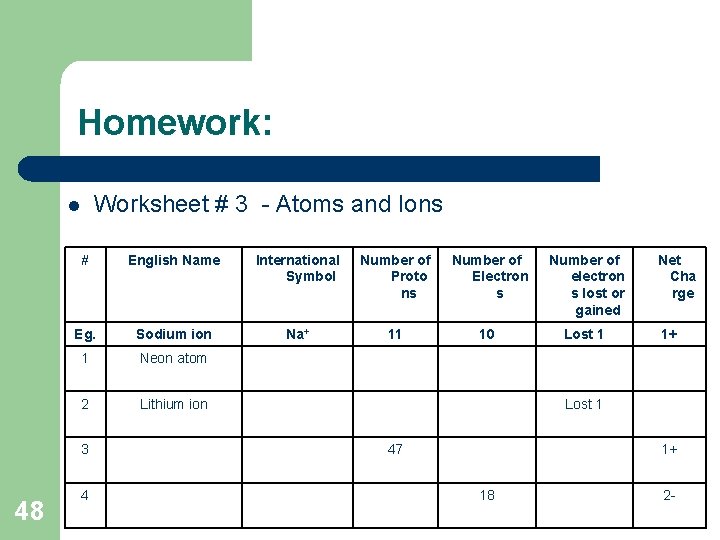
Homework: Worksheet # 3 - Atoms and Ions l # English Name International Symbol Number of Proto ns Number of Electron s Number of electron s lost or gained Eg. Sodium ion Na+ 11 10 Lost 1 1 Neon atom 2 Lithium ion 3 48 4 Net Cha rge 1+ Lost 1 47 1+ 18 2 -

Homework Check and Review l Worksheets # 1, 2, and 3 should be done l Today: – – – 49 Review Worksheet #3 – step by step Introduce Naming Ionic Compounds Review Ecology Test #2
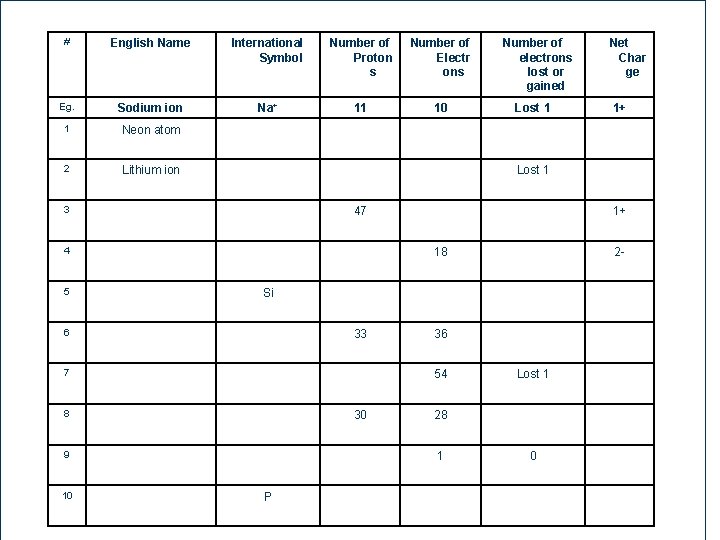
# English Name International Symbol Number of Proton s Number of Electr ons Eg. Sodium ion Na+ 11 10 1 Neon atom 2 Lithium ion 47 4 2 - Si 33 7 36 54 8 30 9 50 1+ 1+ 18 6 10 Lost 1 Net Char ge Lost 1 3 5 Number of electrons lost or gained 28 1 P Lost 1 0
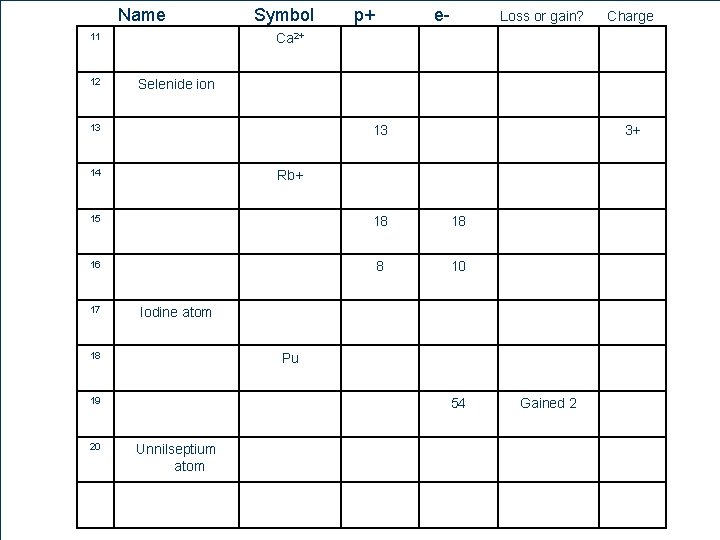
Name Symbol 11 p+ 13 14 17 Intern ationa l Symb ol Nu mbe r of Prot ons Numb er of electr ons lost or gaine d Net Ch arg e 18 18 8 10 Pu 19 51 Nu mbe r of Elec tron s Iodine atom 18 20 3+ Rb+ English Name 16 Charge Selenide ion 13 15 Loss or gain? Ca 2+ 12 # e- 54 Unnilseptium atom Gained 2

Ionic Compounds (Notes: pg. 5) (Text: pg 188 -195) l Ionic Compounds are composed of a cation and an anion – Cation l l – Anion l l 52 Usually a metal (Na+, Mg 2+, Li+, Ca 2+, Fe 3+, etc) Exception - Ammonium, NH 4+ Can be a nonmetal (Cl-, N 3 -, O 2 -) Polyatomic Ions (NO 3 -, PO 43 -, CH 3 COO-)

Ionic Compounds l l All are solids at SATP (Standard Ambient Temperature and Pressure) of 25 o. C and 100 k. Pa. When they dissolve in water, they form aqueous solutions that conduct electricity – l 53 they are electrolytes These compounds form after an electron transfer:

Ionic Compounds l These compounds form after an electron transfer: – 54 Ex: Calcium and Chlorine form Calcium Chloride

l Homework Check: – – 55 Read pg 188 – 189 Answer questions #1, 2, 3

Ionic Compounds (metals with nonmetals) l the resulting ions (cations and anions) are attracted to each other (since they are oppositely charged) and they form ionic bonds l Together all of the ions present form an ionic crystal lattice in which the net charge is zero – – 56 Eg: (1) in a sample of sodium chloride, Na. Cl, for every Na+ ion there is one Cl- ion Eg: (2) in a sample of calclium chloride, Ca. Cl 2, for every Ca 2+ ion there are 2 Cl- ions

Crystal Lattice Na. Cl : A Formula Unit expression of the simplest whole number ratio of cations to anions 57

Types of Ionic Compounds l Monatomic Ions (Simple Ions) – – – l Polyatomic Ions (Complex Ions) – – – 58 Single atoms that have lost or gained one or more electrons Form Binary Ionic Compounds (2 simple ions) Eg. Na+ Cl- Cations or anions composed of a group of atoms with a net positive or negative charge NH 4+ NO 2 NO 3 CO 32 Ammonium ion Nitrite ion Nitrate ion Carbonate ion

Types of Ions and Compounds l Multivalent Ions – – l Hydrated Ionic Compounds – – 59 certain transition metals can form more than one type of ion, each with a different charge Eg. Fe 3+ Fe 2+ The more commonly occurring is listed on top, thus Fe 3+ is more common than Fe 2+ Others: Water molecules are loosely held within the ionic compound Eg. Zn. Cl 2 • 6 H 2 O Cu. SO 4 • 5 H 2 O

Binary Ionic Compounds (Write this Down) l 60 Composed of two different ions l. Only Name Formula sodium chloride Na. Cl magnesium oxide Mg. O lithium nitride Li 2 N aluminum oxide Al 2 O 3 two different types of elements!

Binary ionic formulas l. Given the name, write the formula: Steps 1. Write the symbols for the ions 2. Balance the charges 3. Write the chemical formula Ex: potassium iodide 1. 2. 3. Remember: Ba. Cl 2 not Ba. Cl 2 Ex: calcium iodide 1. 2. 3. 61

Example: silver oxide 62 beryllium bromide

What about these? aluminum oxide 63 zinc nitride

Nomenclature (Naming) l Name the cation and anion and put them together Na. I Ca. I 2 Al 2 S 3 64
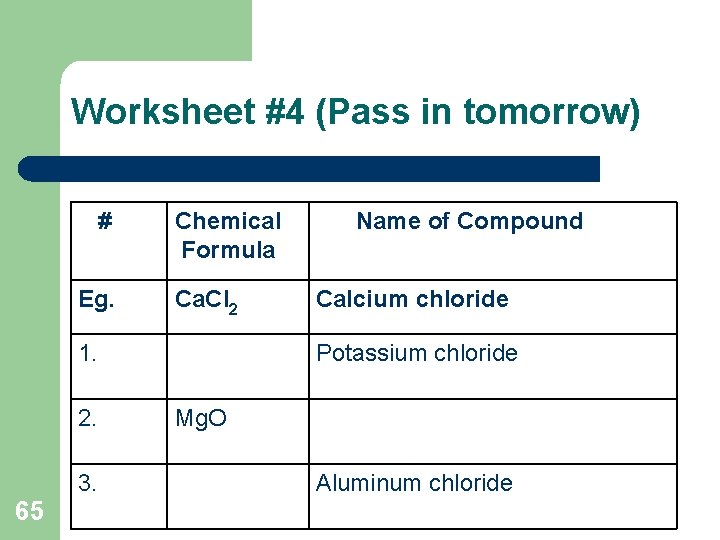
Worksheet #4 (Pass in tomorrow) # Eg. Chemical Formula Ca. Cl 2 1. 2. 3. 65 Name of Compound Calcium chloride Potassium chloride Mg. O Aluminum chloride

Science 1206 Complex Ionic Compounds 66

Write this Down!! Complex Ions l l 67 More then one type of atom is in the ion Listed on the back of periodic table
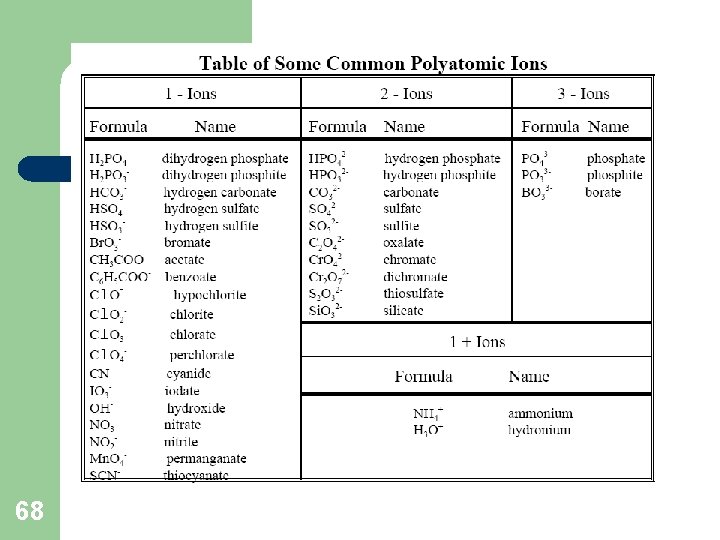
68

Write this Down!! Give formulas for the following: 69 l potassium bromate l silver nitrate l aluminum cyanide

l l 70 ammonium carbonate (2 complex ions) calcium hydrogen carbonate

Name the following formulas l 71 Identify cation and anion first (Look at the tables) – Na. CH 3 COO – KMn. O 4

Continued, , , – Na 3 BO 3 Man, this is COMPLEX !!!! – NH 4 NO 3 If you see ‘lots of letters’ -- 72

Worksheets l Worksheet # 4 – Binary Ionic Compounds Worksheet # 5 – Complex Ions l Both due to be passed in on Tuesday l 73

Science 1206 Multivalent Ions 74

Worksheets Due: l Worksheet # 4 – Binary Ionic Compounds Worksheet # 5 – Complex Ions l Both due to be passed in Today l 75

Quick Review 76 l Binary Ionic Compounds l Complex Ions

Write this Down!! Multivalent Ions l 77 Some elements can form more then one ion

Multivalent Ions l Ex: Iron – Can form Fe 3+ and Fe 2+ the most common is listed on top – Fe 3+ and Cl- – 78 Fe 2+ and Cl-

Naming Rules l l Named the same as other ionic compounds, but we specify the charge using Roman Numerals 1=I 2 = II Ex: – Iron (III) chloride 3 = III 4 = IV 5=V – Iron (II) chloride 6 = VI 7 = VII 79 8 = VIII

Names Formulas Ex: copper (I) nitrate Ex: copper (II) nitrate 80

Names Formulas continued Ex: ruthenium (IV) oxide Ex: bismuth (V) carbonate 81

Formulas Names l Need to identify which ion is present Use the anion (-) to find the charge of the cation (+) l Ex: l – 82 1. Cr. F 3 2. Cr. S
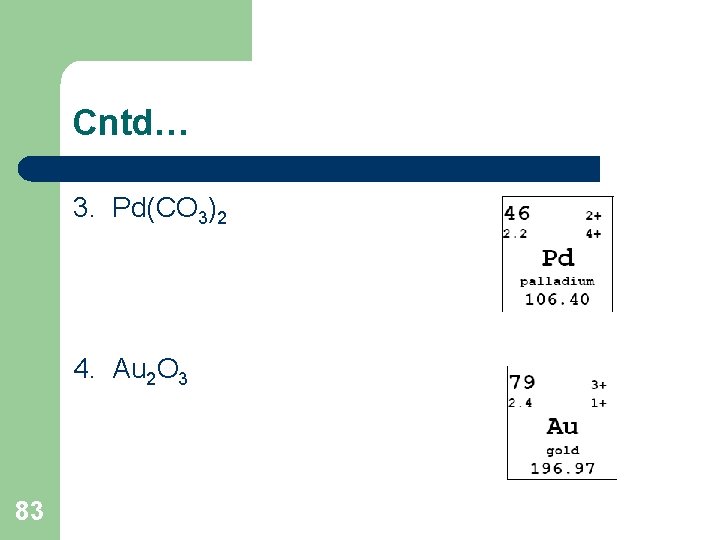
Cntd… 3. Pd(CO 3)2 4. Au 2 O 3 83

Worksheets 84 l Worksheet # 6 - Multivalent Ions l Test: Next week

Science 1206 Hydrated Compounds 85

Homework Check l Worksheet #6 – Multivalent Ions l Return Worksheets #4 & #5 – 86 Fix mistakes are return to me for ½ marks!!
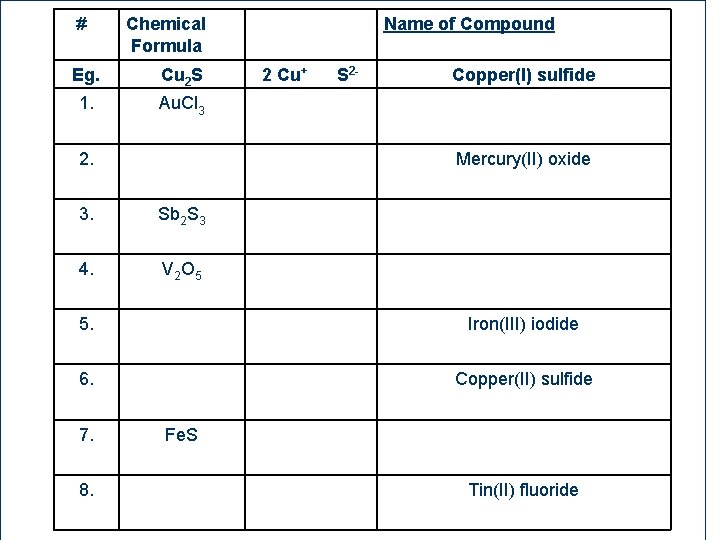
# Chemical Formula Eg. Cu 2 S 1. Au. Cl 3 2. 2 Cu+ S 2 - Copper(I) sulfide Mercury(II) oxide 3. Sb 2 S 3 4. V 2 O 5 5. Iron(III) iodide 6. Copper(II) sulfide 7. 87 Name of Compound 8. Fe. S Tin(II) fluoride
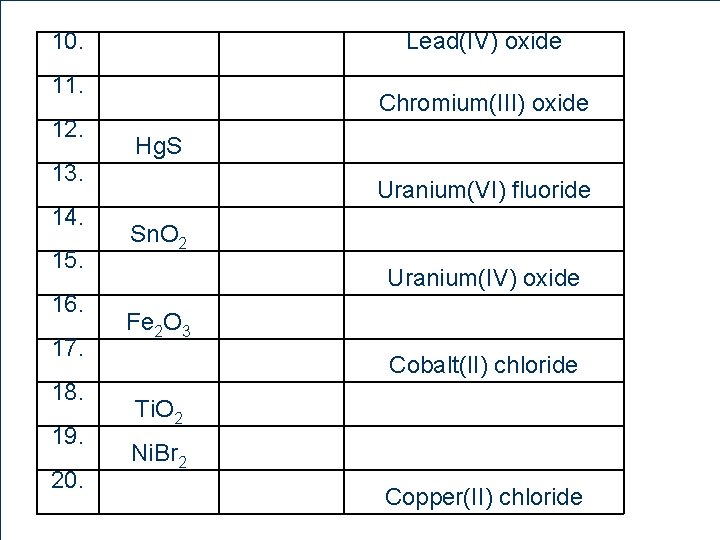
10. Lead(IV) oxide 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 88 Chromium(III) oxide Hg. S Uranium(VI) fluoride Sn. O 2 Uranium(IV) oxide Fe 2 O 3 Cobalt(II) chloride Ti. O 2 Ni. Br 2 Copper(II) chloride

Write this Down!!! Hydrated Compounds (pg. 6 notes, not in textbook) l Ionic compounds sometime have water molecules held loosely Ex: Copper(II) sulfate pentahydrate Cobalt chloride dihydrate 89 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca

Name Formula 1. 2. Give the formula as usual Add “ • #H 2 O ” on the end Ex. Zinc chloride hexahydrate 90 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca

continued… Ex: copper(II) sulfate pentahydrate 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca 91

Formula Name l 1. Name the ionic compound 2. Add prefixhydrate on the end l Ex: Ba(OH)2 • 8 H 2 O l 92 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca

Continued… Ex: Ni. Cl 2 • H 2 O Don’t forget to check if the cation is MULTIVALENT!!! 93 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca

Homework l Worksheet #7 Fix/Finish Worksheets 4 & 5 l Remember: l – – 94 Lunchtime tutorial today Friday @ 3: 00 pm

Test Outline l Classifications of Matter – – – l l Properties and Changes of Matter Elements and the Periodic Table – – 95 Pure substances Compounds Mixtures Groups/Families Periods

l The Atom – – – l Naming and Formula Writing – – 96 # of protons, electrons, neutrons Energy diagrams Ions Binary Ionic Compounds Complex Ions Multivalent cations Hydrated compounds

Test l l 97 25 Multiple Choice Short Answer Naming Formula Writing

Homework Check l Worksheet #6 – Multivalent Ions Worksheet #7 – Hydrated Compounds l Return Worksheets #4 & #5 l – 98 Fix mistakes are return to me for ½ marks!!
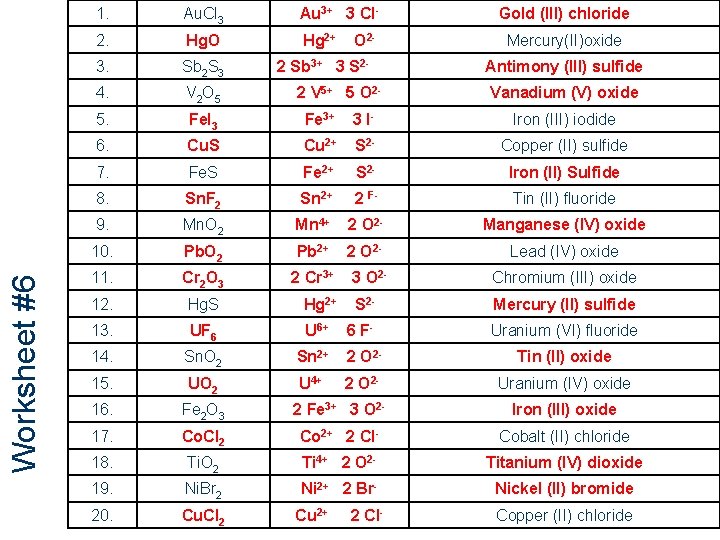
Worksheet #6 99 1. Au. Cl 3 Au 3+ 3 Cl- 2. Hg. O Hg 2+ 3. Sb 2 S 3 2 Sb 3+ 3 S 2 - 4. V 2 O 5 5. Fe. I 3 Fe 3+ 3 I- Iron (III) iodide 6. Cu. S Cu 2+ S 2 - Copper (II) sulfide 7. Fe. S Fe 2+ S 2 - Iron (II) Sulfide 8. Sn. F 2 Sn 2+ 2 F- Tin (II) fluoride 9. Mn. O 2 Mn 4+ 2 O 2 - Manganese (IV) oxide 10. Pb. O 2 Pb 2+ 2 O 2 - Lead (IV) oxide 11. Cr 2 O 3 2 Cr 3+ 12. Hg. S Hg 2+ 13. UF 6 14. O 2 - 2 V 5+ 5 O 2 - Gold (III) chloride Mercury(II)oxide Antimony (III) sulfide Vanadium (V) oxide 3 O 2 - Chromium (III) oxide S 2 - Mercury (II) sulfide U 6+ 6 F- Uranium (VI) fluoride Sn. O 2 Sn 2+ 2 O 2 - Tin (II) oxide 15. UO 2 U 4+ 2 O 2 - Uranium (IV) oxide 16. Fe 2 O 3 2 Fe 3+ 3 O 2 - Iron (III) oxide 17. Co. Cl 2 Co 2+ 2 Cl- Cobalt (II) chloride 18. Ti. O 2 Ti 4+ 2 O 2 - Titanium (IV) dioxide 19. Ni. Br 2 Ni 2+ 2 Br- Nickel (II) bromide 20. Cu. Cl 2 Cu 2+ 2 Cl- Copper (II) chloride
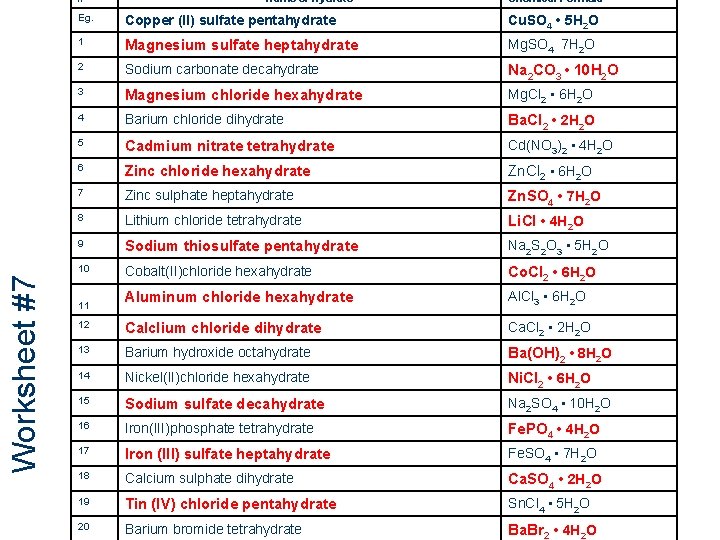
Worksheet #7 # 10 0 Name of Hydrate Chemical Formula Eg. Copper (II) sulfate pentahydrate Cu. SO 4 • 5 H 2 O 1 Magnesium sulfate heptahydrate Mg. SO 4 7 H 2 O 2 Sodium carbonate decahydrate Na 2 CO 3 • 10 H 2 O 3 Magnesium chloride hexahydrate Mg. Cl 2 • 6 H 2 O 4 Barium chloride dihydrate Ba. Cl 2 • 2 H 2 O 5 Cadmium nitrate tetrahydrate Cd(NO 3)2 • 4 H 2 O 6 Zinc chloride hexahydrate Zn. Cl 2 • 6 H 2 O 7 Zinc sulphate heptahydrate Zn. SO 4 • 7 H 2 O 8 Lithium chloride tetrahydrate Li. Cl • 4 H 2 O 9 Sodium thiosulfate pentahydrate Na 2 S 2 O 3 • 5 H 2 O 10 Cobalt(II)chloride hexahydrate Co. Cl 2 • 6 H 2 O Aluminum chloride hexahydrate Al. Cl 3 • 6 H 2 O 12 Calclium chloride dihydrate Ca. Cl 2 • 2 H 2 O 13 Barium hydroxide octahydrate Ba(OH)2 • 8 H 2 O 14 Nickel(II)chloride hexahydrate Ni. Cl 2 • 6 H 2 O 15 Sodium sulfate decahydrate Na 2 SO 4 • 10 H 2 O 16 Iron(III)phosphate tetrahydrate Fe. PO 4 • 4 H 2 O 17 Iron (III) sulfate heptahydrate Fe. SO 4 • 7 H 2 O 18 Calcium sulphate dihydrate Ca. SO 4 • 2 H 2 O 19 Tin (IV) chloride pentahydrate Sn. Cl 4 • 5 H 2 O 20 Barium bromide tetrahydrate Ba. Br 2 • 4 H 2 O 11

Review: l 10 1 Worksheets

Science 1206 Molecular Compounds 10 2

Molecular Compounds (Handouts pg 8, Text pg 201) l l l MOLECULAR SUBSTANCES: are solids, liquids or gases at SATP if soluble, dissolve in water to form colorless aqueous solutions that do not conduct electricity – l 10 3 they are non-electrolytes they contain only nonmetal atoms

Molecular Compounds l Molecule: – l Covalent Bond: – 10 4 a particle of a molecular substance that contains a fixed number of covalently-bonded nonmetal atoms formed from the sharing of valence electrons between nonmetal atoms, which results in an electron structure that is the same as a noble gas, for each atom in the molecule

Covalent Bonding 10 5 l Hydrogen l Chlorine l Carbon dioxide
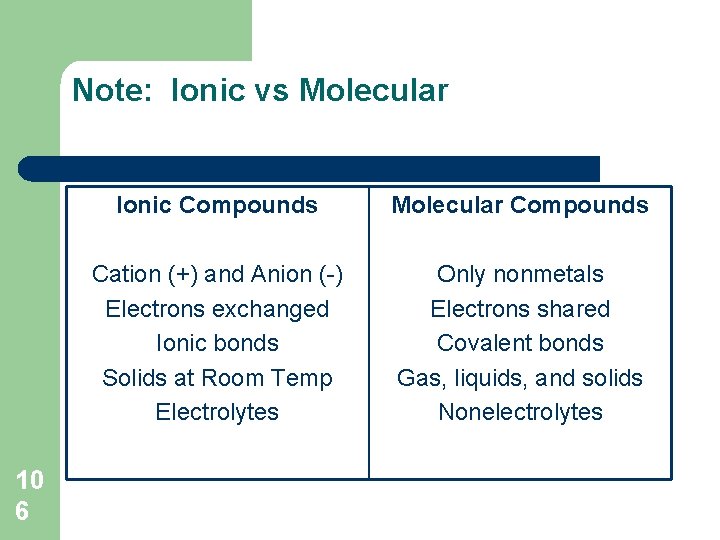
Note: Ionic vs Molecular 10 6 Ionic Compounds Molecular Compounds Cation (+) and Anion (-) Electrons exchanged Ionic bonds Solids at Room Temp Electrolytes Only nonmetals Electrons shared Covalent bonds Gas, liquids, and solids Nonelectrolytes
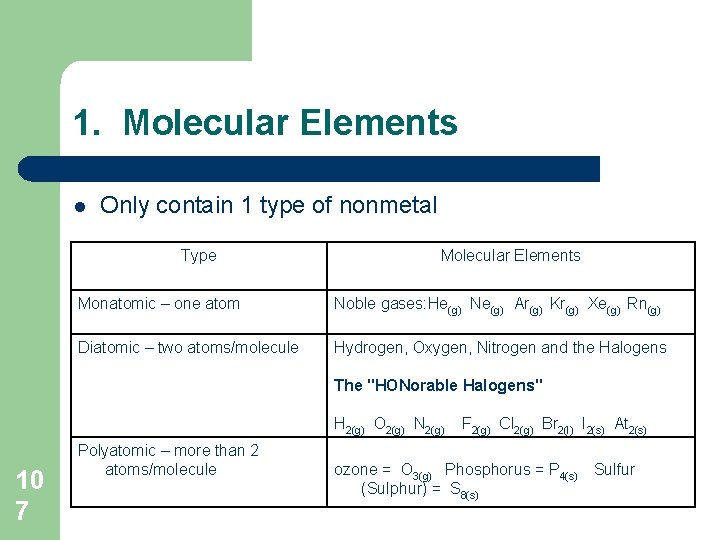
1. Molecular Elements l Only contain 1 type of nonmetal Type Molecular Elements Monatomic – one atom Noble gases: He(g) Ne(g) Ar(g) Kr(g) Xe(g) Rn(g) Diatomic – two atoms/molecule Hydrogen, Oxygen, Nitrogen and the Halogens The "HONorable Halogens" H 2(g) O 2(g) N 2(g) 10 7 Polyatomic – more than 2 atoms/molecule F 2(g) Cl 2(g) Br 2(l) I 2(s) At 2(s) ozone = O 3(g) Phosphorus = P 4(s) (Sulphur) = S 8(s) Sulfur

2. Molecular Compounds l 10 8 Molecular Compounds a) Common (to memorize): H 2 O(l) = water CH 4(g) = methane CH 3 OH(l) = methanol H 2 O 2(l) = hydrogen peroxide C 3 H 8(g) = propane C 2 H 5 OH(l) = ethanol NH 3(g) = ammonia C 6 H 12 O 6(s) = glucose C 12 H 22 O 11(s) = sucrose

2. Molecular Compounds l 10 9 B. Binary Molecular Compounds – 2 different nonmetals – CO CO 2 CCl 4

Writing Molecular Formulas General Rules 1. Write each atom symbol. 2. Each prefix indicates the subscript for the nonmetal atom that precedes it (# of atoms present). 3. If no prefix is present, then there is only one atom of that nonmetal present. Monoxide = one oxygen atom present. l Examples: 11 0 Carbon monoxide Carbon dioxide Carbon tetrachloride Suphur trioxide Dinitrogen monoxide

Naming Binary Molecular Compounds Naming Molecular Substances General Rules 1. First element is named in full. 2. Second element name is shortened and given an ide ending. 3. Use prefixes (same as for hydrates) to indicate the number of each kind of atom. – – – 11 1 The prefix mono is usually only used for molecules with 1 atom of oxygen (monoxides). Certain Hydrogen compounds (those with H first in the formula) do not use prefixes. H 2 S(g) = hydrogen sulfide, not dihydrogen sulfide

Examples l 11 2 Examples: NO(g) P 4 O 6(s) SO 2(g) SO 3(g) N 2 O(g) PCl 3(s) NI 3(s)

Homework l 11 3 Worksheet #8

Science 1206 Acids and Bases 11 4

Homework Check l 11 5 Worksheet 8 – Molecular compounds
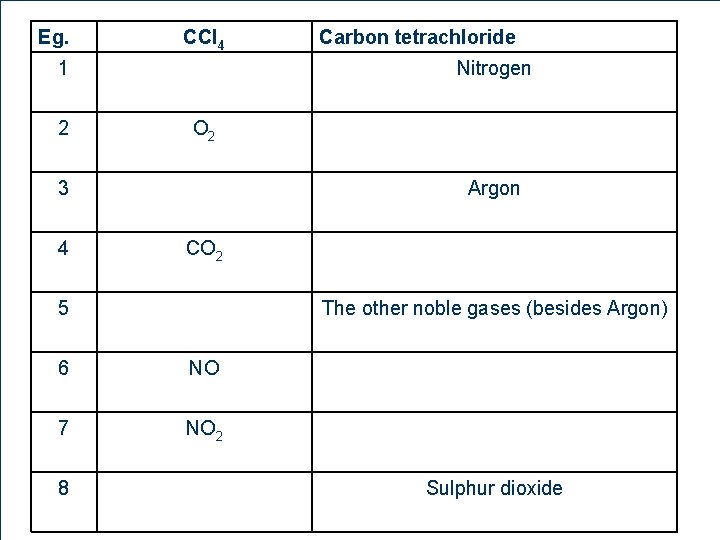
Eg. CCl 4 1 2 Nitrogen O 2 3 4 Argon CO 2 5 11 6 The other noble gases (besides Argon) 6 NO 7 NO 2 8 Carbon tetrachloride Sulphur dioxide
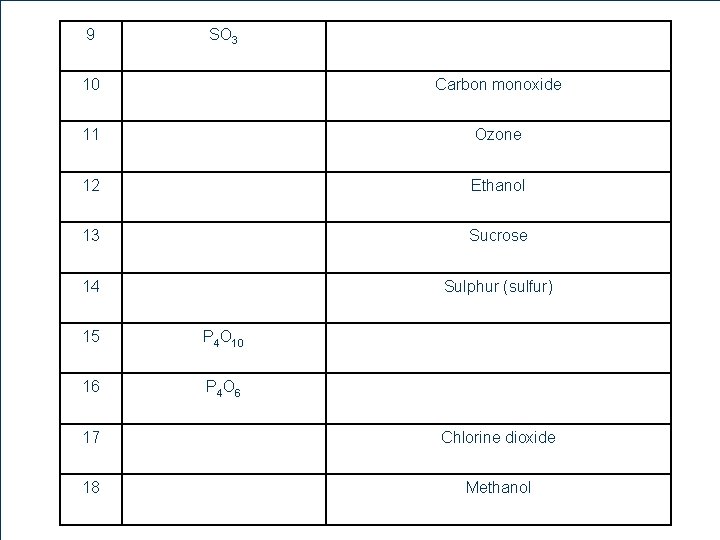
9 11 7 SO 3 10 Carbon monoxide 11 Ozone 12 Ethanol 13 Sucrose 14 Sulphur (sulfur) 15 P 4 O 10 16 P 4 O 6 17 Chlorine dioxide 18 Methanol
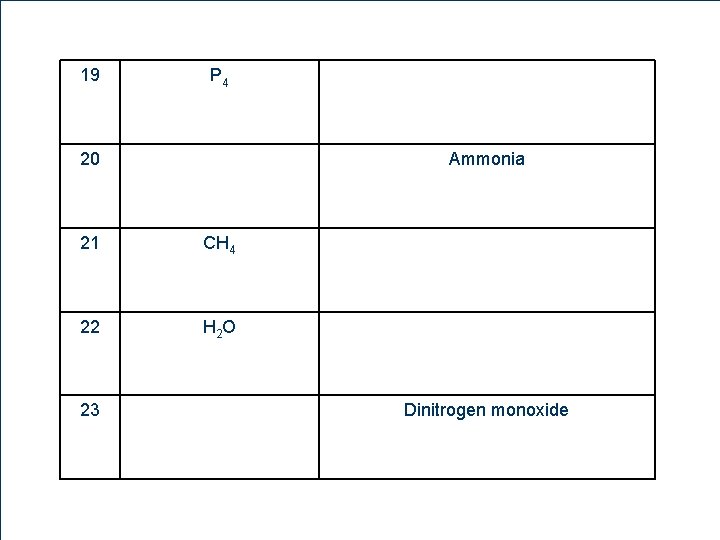
19 P 4 20 21 CH 4 22 H 2 O 23 11 8 Ammonia Dinitrogen monoxide

Acids (p. 10 notes) l ACIDS – l Properties of acids: – – – 11 9 Molecules that ionize in water to produce hydrogen ions, H+(aq), ions which give acids their properties Conduct electricity Turn blue litmus paper red Taste sour React with many metals to produce hydrogen gas, H 2(g) Have a p. H value of less than 7 Neutralize or partially neutralize bases

Acids l General Formula: H____ (aq) or l Note: not all hydrogen containing compounds are acids – 12 0 ____COOH (aq) Eg: NH 3 CH 4 CH 3 OH C 2 H 5 OH

Naming Acids l 1. 2. General Rules Name the hydrogen compound like an ionic compound Then convert the ionic name to the acid name hydrogen _______ide becomes hydro______ic acid hydrogen____ite becomes _____ous acid hydrogen____ate becomes _____ic acid 12 1
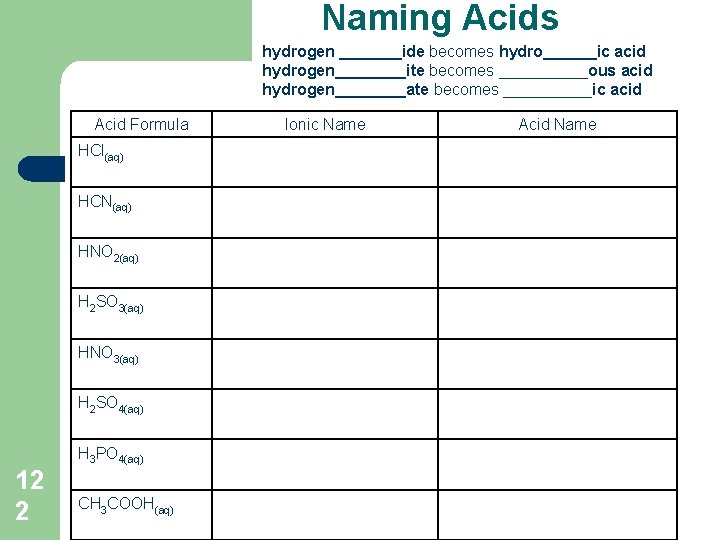
Naming Acids hydrogen _______ide becomes hydro______ic acid hydrogen____ite becomes _____ous acid hydrogen____ate becomes _____ic acid Acid Formula HCl(aq) HCN(aq) HNO 2(aq) H 2 SO 3(aq) HNO 3(aq) H 2 SO 4(aq) H 3 PO 4(aq) 12 2 CH 3 COOH(aq) Ionic Name Acid Name

Writing Acid Formulas General Rules: 1. Translate acid name into ionic name: hydro___ic acid hydrogen ___ide ___ous acid hydrogen ___ite ___ic acid hydrogen ___ate 12 3 2. Write chemical formulas for each ion, using rules for writing formulas for ionic compounds. 3. Hydrogen symbol is written first (cation), except for carboxylic acids (those with COO group), in which case hydrogen is placed at the end eg: CH 3 COOH 4. Give the state as aqueous = (aq).
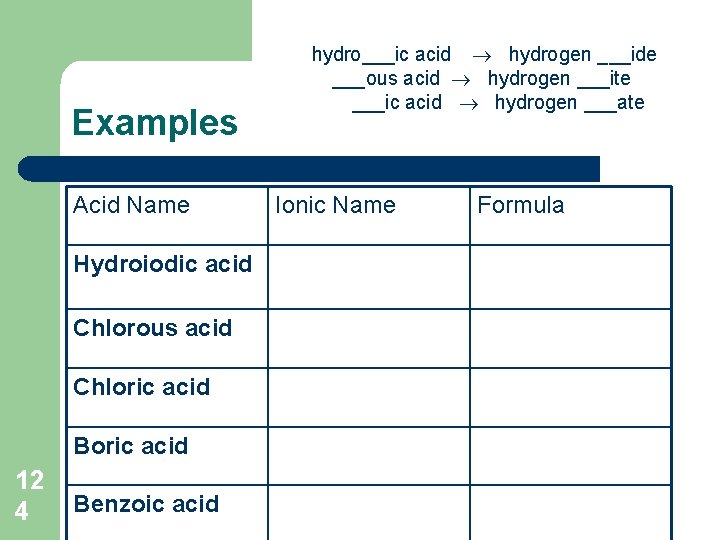
Examples Acid Name Hydroiodic acid Chlorous acid Chloric acid Boric acid 12 4 Benzoic acid hydro___ic acid hydrogen ___ide ___ous acid hydrogen ___ite ___ic acid hydrogen ___ate Ionic Name Formula

Homework l 12 5 Worksheet #9
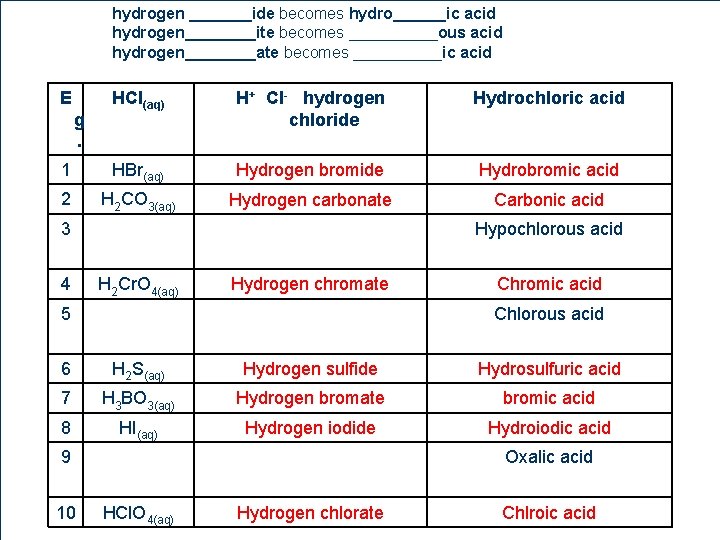
hydrogen _______ide becomes hydro______ic acid hydrogen____ite becomes _____ous acid hydrogen____ate becomes _____ic acid E HCl(aq) H+ Cl- hydrogen chloride Hydrochloric acid 1 HBr(aq) Hydrogen bromide Hydrobromic acid 2 H 2 CO 3(aq) Hydrogen carbonate Carbonic acid g. 3 4 Hypochlorous acid H 2 Cr. O 4(aq) Hydrogen chromate 5 12 6 Chromic acid Chlorous acid 6 H 2 S(aq) Hydrogen sulfide Hydrosulfuric acid 7 H 3 BO 3(aq) Hydrogen bromate bromic acid 8 HI(aq) Hydrogen iodide Hydroiodic acid 9 10 Oxalic acid HCl. O 4(aq) Hydrogen chlorate Chlroic acid
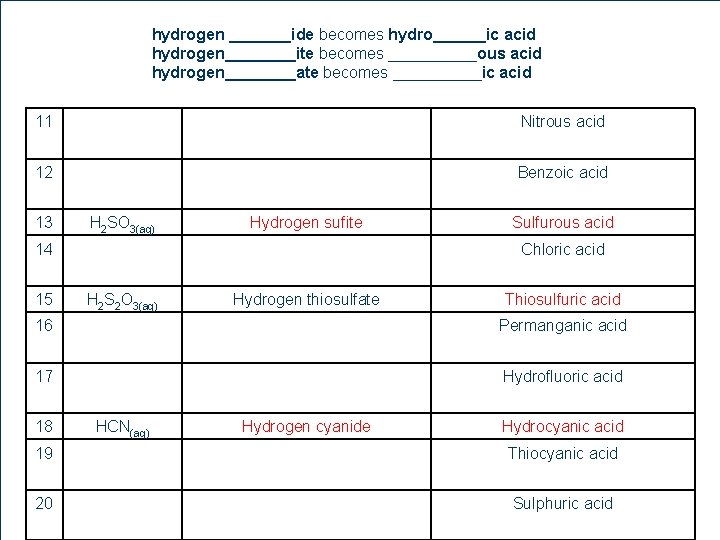
hydrogen _______ide becomes hydro______ic acid hydrogen____ite becomes _____ous acid hydrogen____ate becomes _____ic acid 11 Nitrous acid 12 Benzoic acid 13 H 2 SO 3(aq) Hydrogen sufite 14 15 Sulfurous acid Chloric acid H 2 S 2 O 3(aq) Hydrogen thiosulfate Thiosulfuric acid 16 Permanganic acid 17 Hydrofluoric acid 18 19 12 7 20 HCN(aq) Hydrogen cyanide Hydrocyanic acid Thiocyanic acid Sulphuric acid

Base l l Most are ionic compounds with OH- and (aq) Properties of bases: – – – 12 8 – Conduct electricity Turn red litmus paper blue Taste bitter Feel slippery Have a p. H value greater than 7 Neutralize or partially neutralize acids

Bases l Naming Bases – Follow the general rules given for ionic compounds Na. OH(aq) NH 4 OH(aq) l Writing Base Formulas – follow the general rules given for ionic compounds lithium hydroxide 12 9 Calcium hydroxide

WHMIS l Workplace and Hazardous Materials Information System MSDS l 13 0 Material Safety Data Sheet

Homework l WHMIS and MSDS Worksheet – – l 13 1 Passed in on Monday Very short assignment Naming and Formula Review Sheet

Science 1206 Chemical Equations 13 2

Pass in Assignments l WHMIS and MSDS Worksheet – – 13 3 Passed in on Monday Very short assignment l Naming and Formula Review Sheet l Get Homework Checklist

Chemical Equations l Show chemicals react to form new compounds and molecules 2 Mg(s) + O 2(g) 2 Mg. O(s) Reactants Products l 13 4 l The number of atoms must be conserved!!! We use Coefficients to balance

Before we start: l Learn to count the atoms: l Ca(NO 3)2 Ca = N= O= 13 5 (NH 4)2 SO 4 N= H= S= O=

Balancing Chemical Equations Start by balancing the atom with the highest numbers: Mg + O 2 13 6 Mg. O
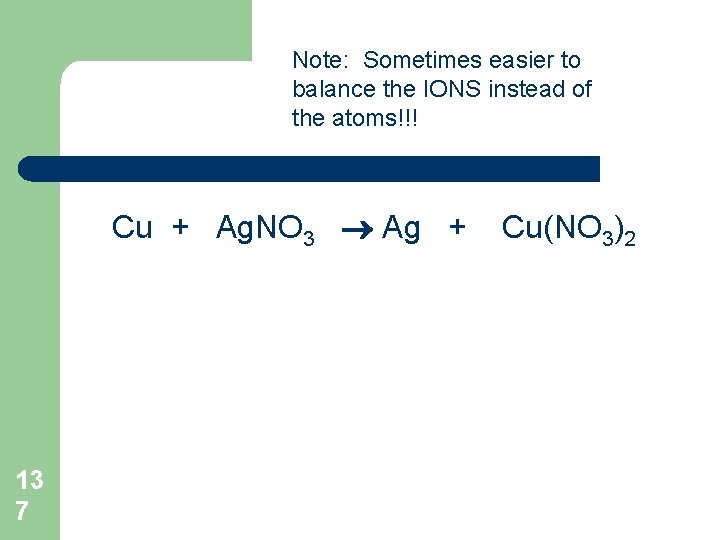
Note: Sometimes easier to balance the IONS instead of the atoms!!! Cu + Ag. NO 3 Ag + 13 7 Cu(NO 3)2
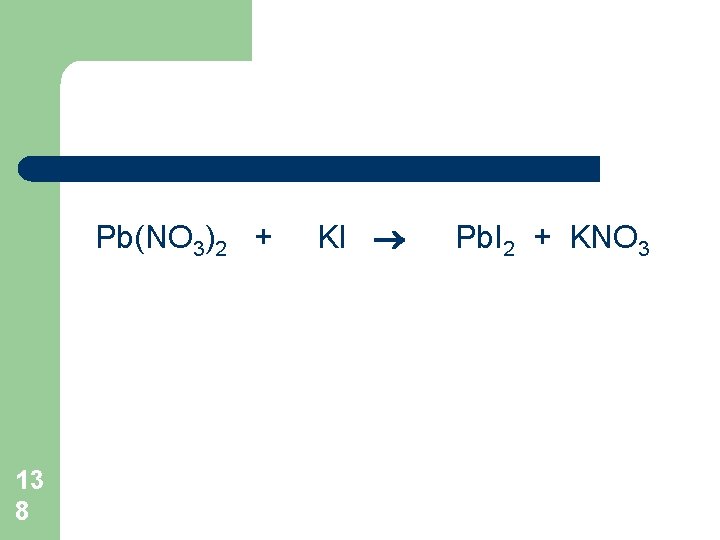
Pb(NO 3)2 + 13 8 KI Pb. I 2 + KNO 3
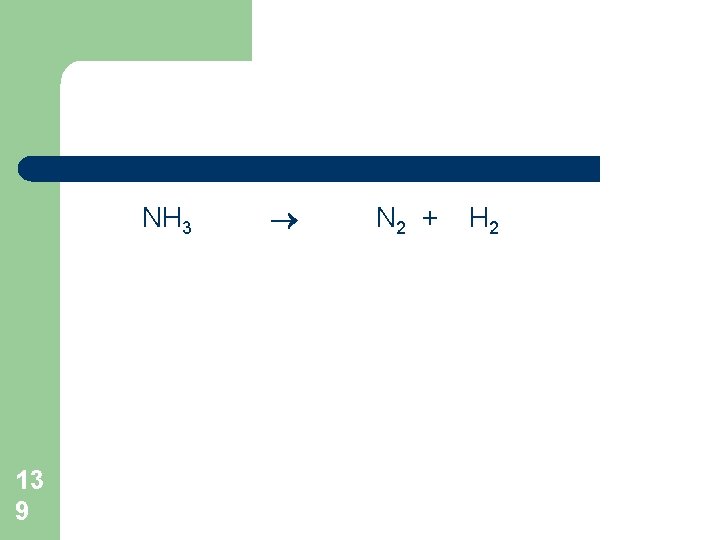
NH 3 13 9 N 2 + H 2
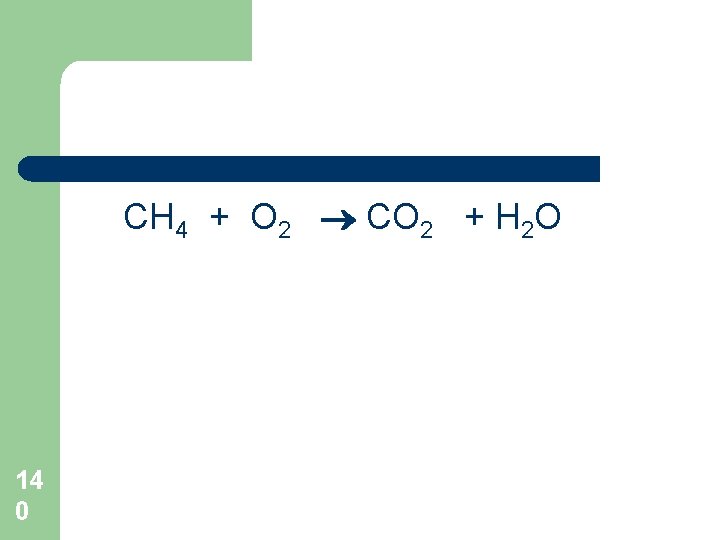
CH 4 + O 2 CO 2 + H 2 O 14 0

Try These: 1. Fe 14 1 + O 2 Fe 2 O 3 2. Na + Cl 2 Na. Cl 3. As. Cl 3 + H 2 S As 2 S 3 + HCl
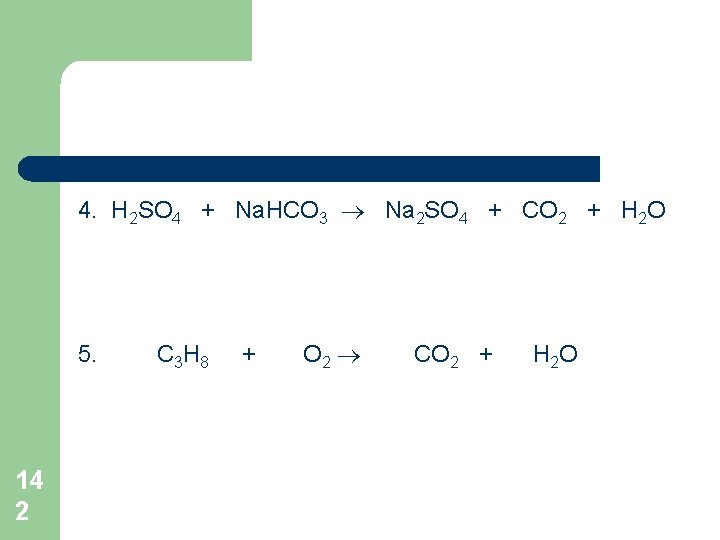
4. H 2 SO 4 + Na. HCO 3 Na 2 SO 4 + CO 2 + H 2 O 5. 14 2 C 3 H 8 + O 2 CO 2 + H 2 O

Homework l Worksheet #10 Balancing – – We will correct this at the beginning of next class Have it done!!!! Test Review: 14 3

14 4
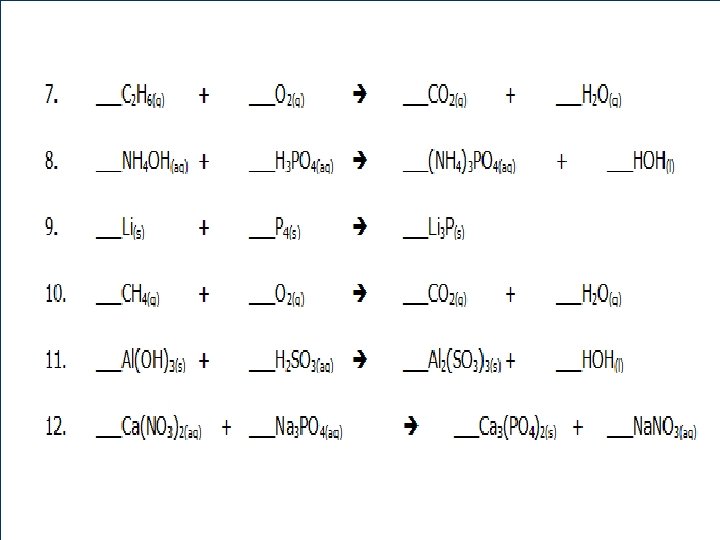
14 5

WRITING BALANCED CHEMICAL EQUATIONS (pg. 2, Part II notes) l To write a balanced chemical equation from a statement or word equation: 1. 2. 14 6 Write the chemical formulas for all reactants and products involved Arrange as Reactants Products

Example Hydrogen and chlorine react to produce hydrogen chloride gas. 1. Word Equation: 2. Chemical Equation: 14 7

Example 2 (pg. 6, Balancing Worksheet) Solid potassium and aqueous magnesium chloride react to produce solid magnesium and aqueous potassium chloride. 1. Word Equation: 2. Chemical Equation: 14 8

Remainder of Class (Homework) l Worksheet #11 – Balancing Chemical Reactions – 14 9 Don’t forget: WHMIS Worksheet and Formula Review

Science 1206 Reaction Types 15 0

Homework l Worksheet #11 – Balancing Chemical Reactions – Don’t forget: WHMIS Worksheet and Formula Review l l 15 1 Both have to be passed in today Quiz on Monday – Balancing and Reaction Types

Worksheet #11 2. Solid aluminum combines with oxygen gas to produce solid aluminum oxide. Aluminum + oxygen aluminum oxide Al 15 2 + O 2 Al 2 O 3

3. Hydrogen peroxide decomposes (breaks down) into water and oxygen gas. Hydrogen peroxide water + oxygen 2 H 2 O 2 15 3 2 H 2 O + O 2

4. The combustion (burning) of ethyne gas, C 2 H 2(g) in the presence of oxygen gas produces carbon dioxide gas and water vapor. ethyne + oxygen carbon dioxide + water 2 C 2 H 2 15 4 + 5 O 2 4 CO 2 + 2 H 2 O

Types of Chemical Reactions (pg 2 -4 notes) l There are 5 types of reactions that you need to recognize: – – – 15 5 Simple Composition Simple Decomposition Single Replacement Double Replacement Hydrocarbon Combustion

1. Formation (Simple Composition) 2 elements or compounds react to form 1 new compound Example: General: 2 Mg(s) + O 2(g) 2 Mg. O A + B AB Word: element + element compound 15 6

Formation: A + B AB 15 7 l magnesium reacts with oxygen from the air l Magnesium + oxygen l Mg + O 2

Formation: A + B AB 15 8 l hydrogen and oxygen react to produce water l Hydrogen + oxygen water l 2 H 2 + O 2 2 H 2 O

2. Decomposition 1 compound breaks down into 2 elements or compounds General: Word: 15 9 AB A + B compound element + element OR compound + compound Example: 2 Cu 2 O(s) 4 Cu(s) + O 2(g)

AB A + B water is broken down into its elements Water H 2 O 16 0

AB A + B mercury(II) oxide decomposes Mercury(II) oxide Hg. O 16 1

3. Single Replacement (Single Displacement) l A element replaces the cation/anion in a compound General: A + BC B + AC Word: element + compound Example: 16 2 Zn(s) + Pb(NO 3)2(aq) Pb(s) + Zn(NO 3)2(aq)

A + BC B + AC 16 3 l Barium and zinc chloride react l Barium + zinc chloride l Ba + Zn. Cl 2
A + BC B + AC l chlorine reacts with sodium bromide solution l Chlorine + sodium bromide l 16 4 Cl 2 + Na. Br

4. Double Replacement (Double Displacement) l Two compounds react and switch ions General: AB + CD AD + CB Word: compound + compound Example: Ba. Cl 2(aq) + Ag. NO 3(aq) Ba(NO 3)2(aq) + Ag. Cl(s) 16 5

16 6 l solutions of barium chloride and potassium carbonate react l barium chloride + potassium carbonate l Ba. Cl 2 + K 2 CO 3

5. Hydrocarbon Combustion (Not on midterm) l A hydrocarbon (Cx. Hy) reacts with oxygen to produce CO 2 and H 2 O General: Cx. Hy + O 2(g) CO 2(g) + H 2 O(g) Word: hydrocarbon + oxygen carbon dioxide + water Example: 16 7 C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(g)

16 8 l Butane, C 4 H 10(g) is burned as fuel in a lighter l Butane + oxygen
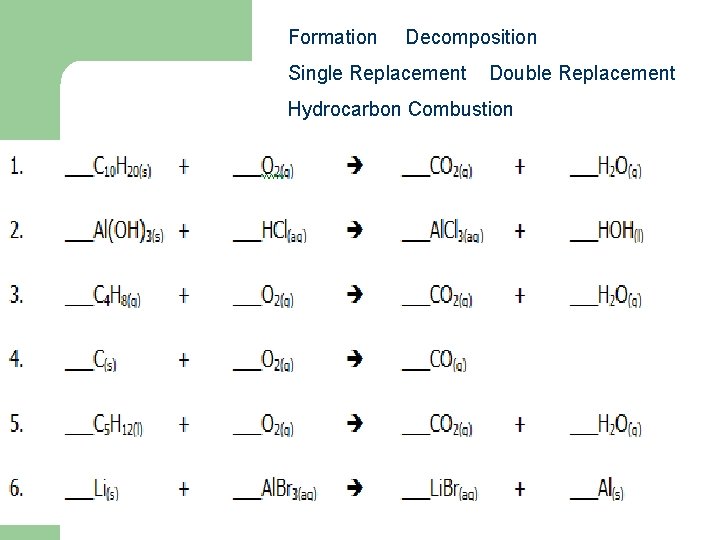
Formation Decomposition Single Replacement Double Replacement Hydrocarbon Combustion 16 9
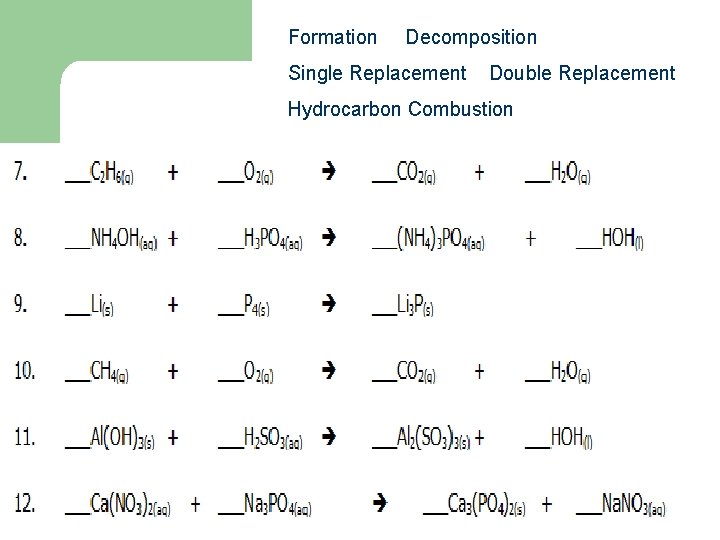
Formation Decomposition Single Replacement Double Replacement Hydrocarbon Combustion 17 0

Homework l Worksheets – – – l 17 1 12 13 14 (Not Combustion) Monday – Short Quiz – Balancing – Give equations from names – Identify reaction types
- Slides: 171