Science 10 Unit 2 Elements are organized by

Science 10 Unit 2
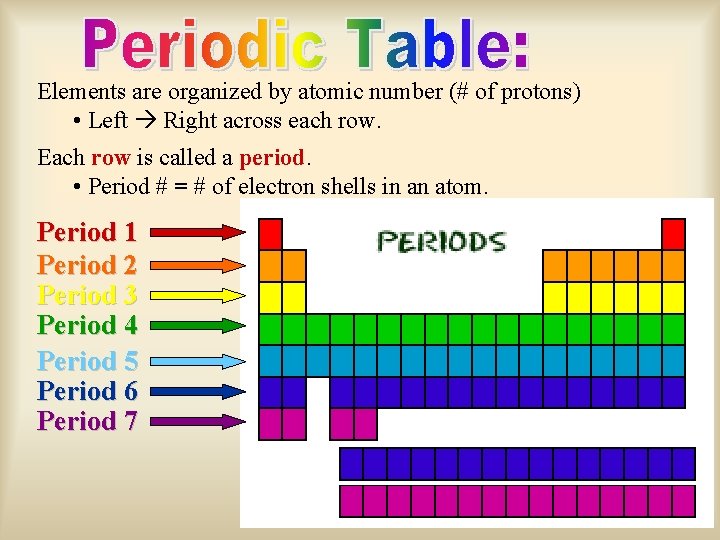
Elements are organized by atomic number (# of protons) • Left Right across each row. Each row is called a period. • Period # = # of electron shells in an atom. Period 1 Period 2 Period 3 Period 4 Period 5 Period 6 Period 7

Each column is called a group or family. Elements in the same group have similar chemical properties. Group 1: Alkali Metals Very reactive metals 2: Alkaline Earth Metals Somewhat reactive metals 3 -12: Transition Metals 17: Halogens Very reactive non-metals 18: Noble Gases Very unreactive gaseous non-metals Last digit of group # equals the # of valence electrons in the atom. (coloured groups only, not including transition elements) 1 17 2 13 14 3 4 5 6 7 8 9 10 11 12 15 16 18

All atoms want to have a full outer electron shell (valence). Atoms will gain or lose electrons to achieve a full valence. When atoms gain or lose electrons, they become electrically charged. v. The number of protons (+) is no longer equal to the number of electrons (-). v. E. g. : Sodium now has 11 protons (+) and only 10 electrons (-) giving it an overall charge of 1+

Metals tend to lose electrons to become postive ions (cations). + 1 elost Lithium ion (Li+) Non-metals tend to gain electrons to become negative ions (anions). -2 2 egained Oxygen ion (O 2 -)

v Note: to tell at a glance what kind of ion an element will form, look at the periodic table. Group 1: 2: 13: 14: 15: 16: 17: (Alkali Metals) (Alkaline Earth Metals) (Aluminum, etc. ) (Carbon, etc. ) (Nitrogen, etc. ) (Oxygen, etc. ) (Halogens) +1 +2 +3 ± 4 -3 -2 -1

v Some metals are multivalent (can form ions in more than one way) i. e. : Iron can lose 2 or 3 electrons to become a 2+ or 3+ ion. v On your periodic table, the most common charge is listed on top. Ion Charges Most Common Ion Charges

In Class: Periodic Table Scavenger Hunt Homework: Workbook pages 60 -61 (leave Q 4 on P 61 until after next class)
- Slides: 8