Schrdinger Heisenberg Model Recall Bohrs model of the

Schrödinger & Heisenberg Model
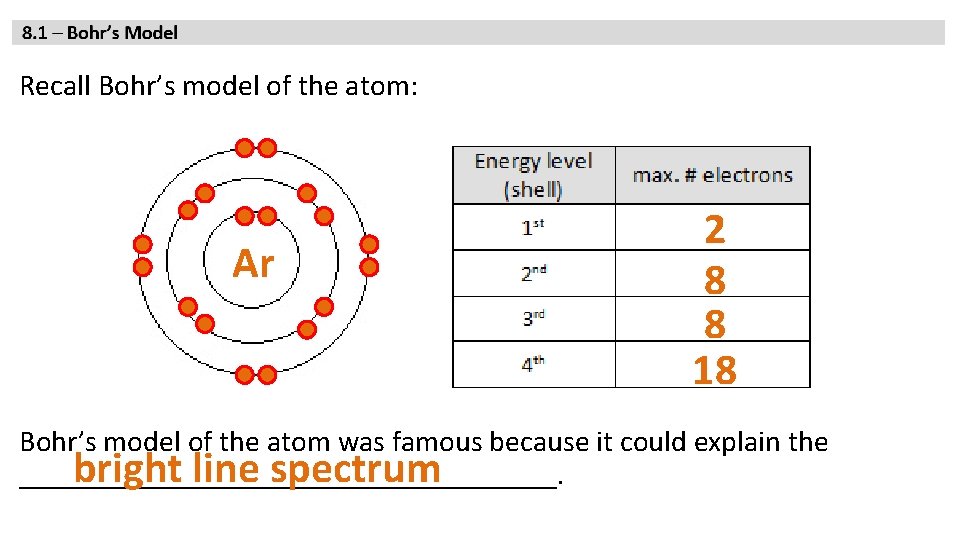
Recall Bohr’s model of the atom: Ar 2 8 8 18 Bohr’s model of the atom was famous because it could explain the bright line spectrum __________________.
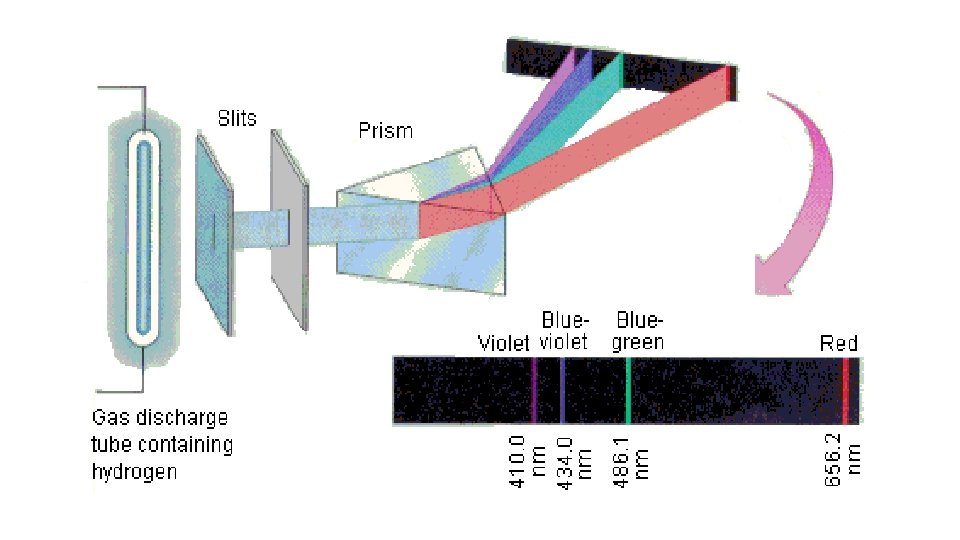

continuous The spectrum of white light is _________, it shows all the colours of the rainbow The spectrum of energized gas is _________, discontinuous it shows _________. discrete bands
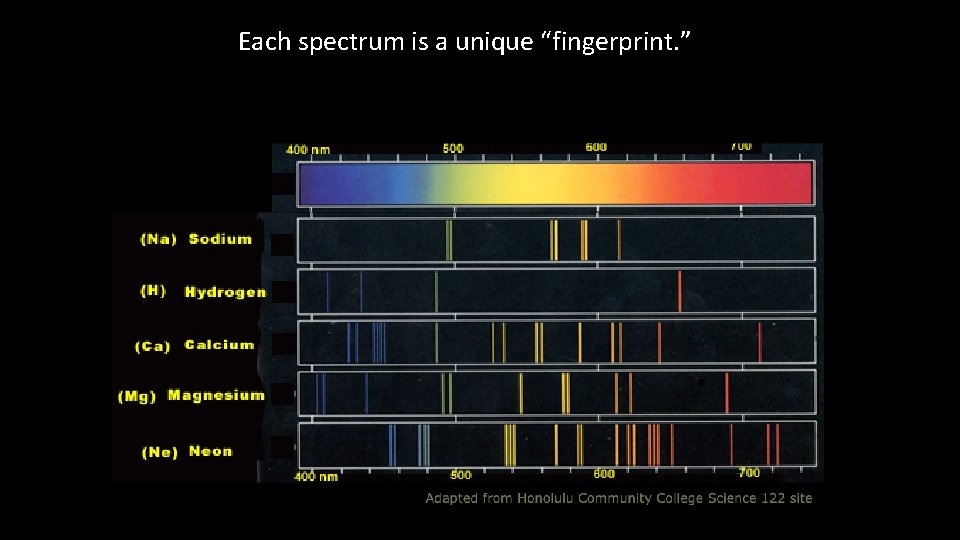
Each spectrum is a unique “fingerprint. ”


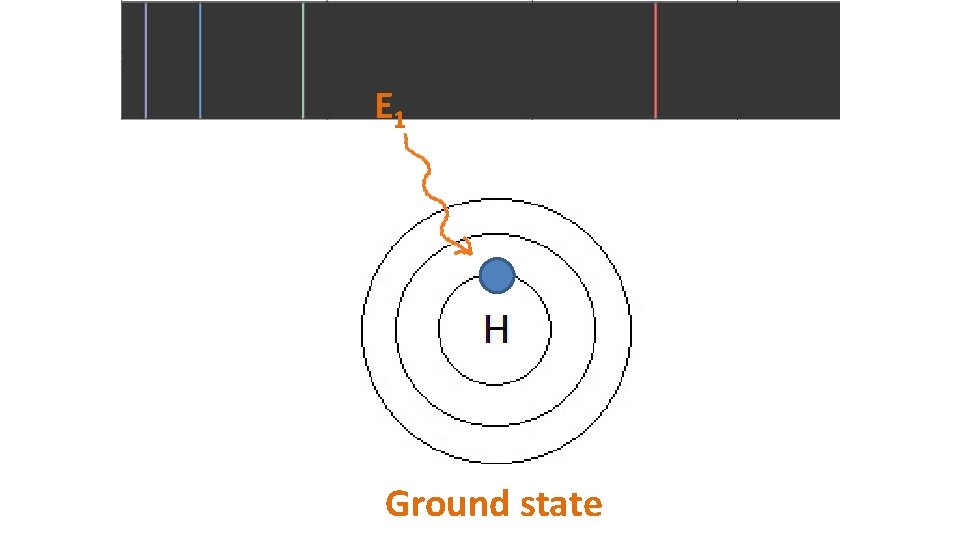
E 1 Ground state
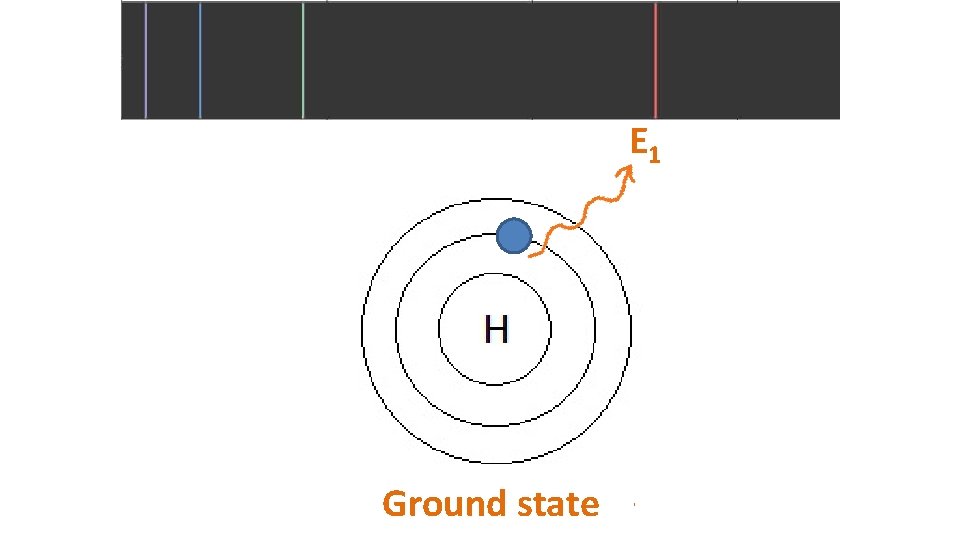
E 1 st excited 1 Ground state
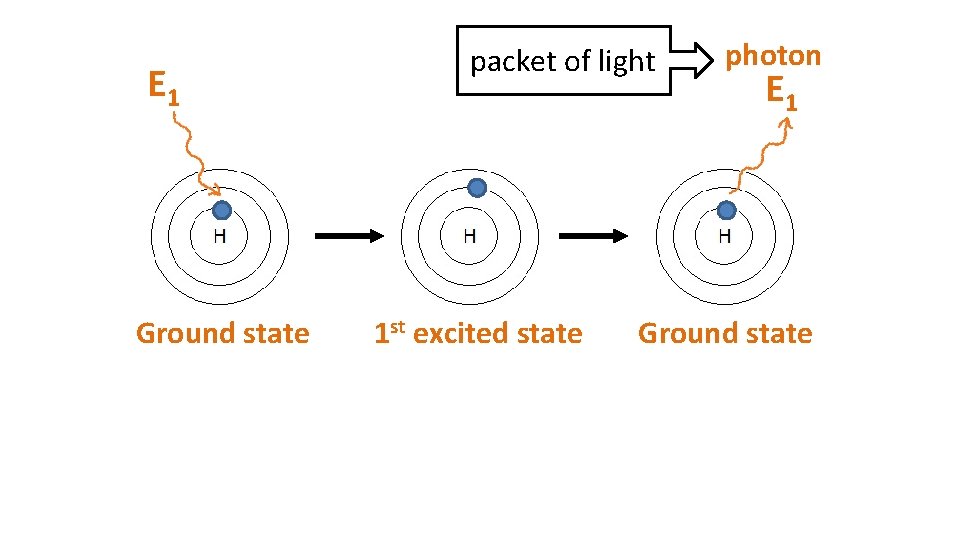
E 1 Ground state packet of light 1 st excited state photon E 1 Ground state
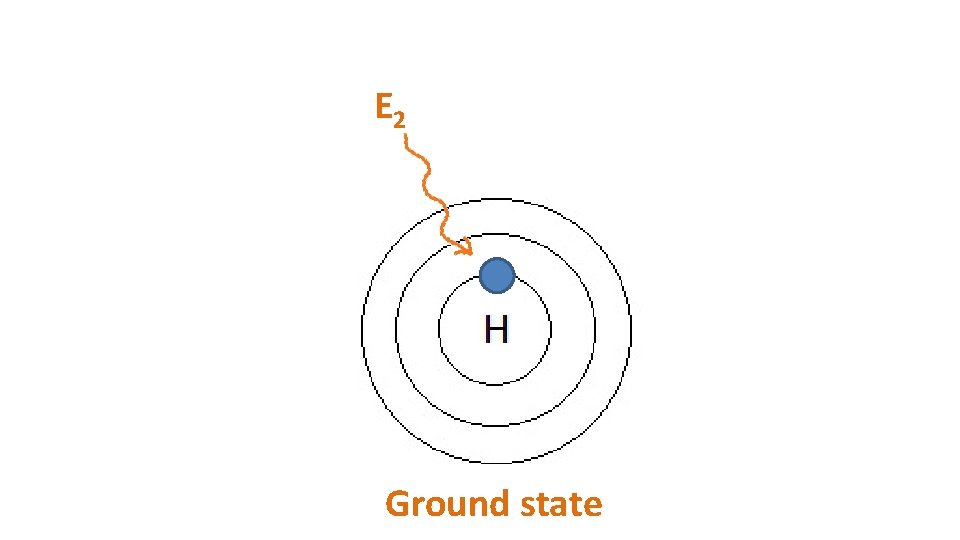
E 2 Ground state
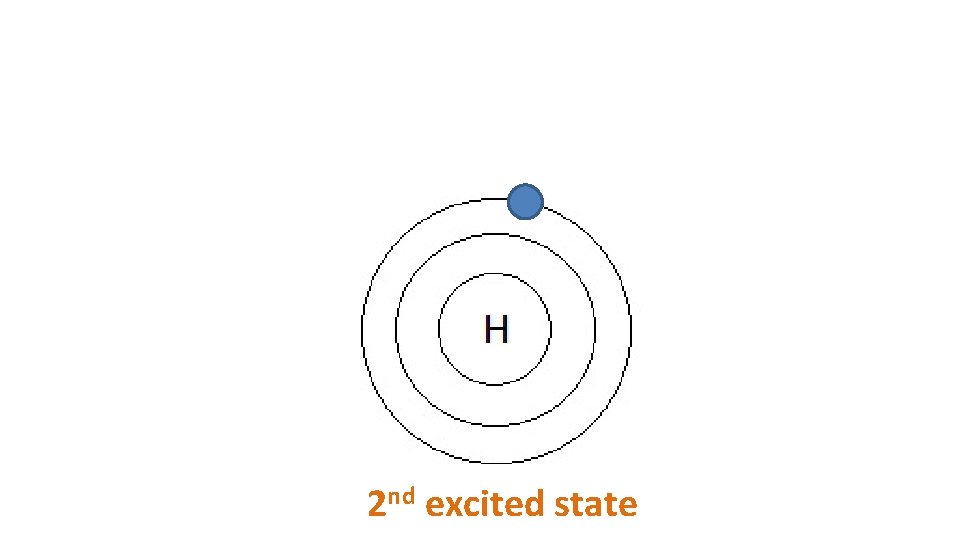
2 nd excited state
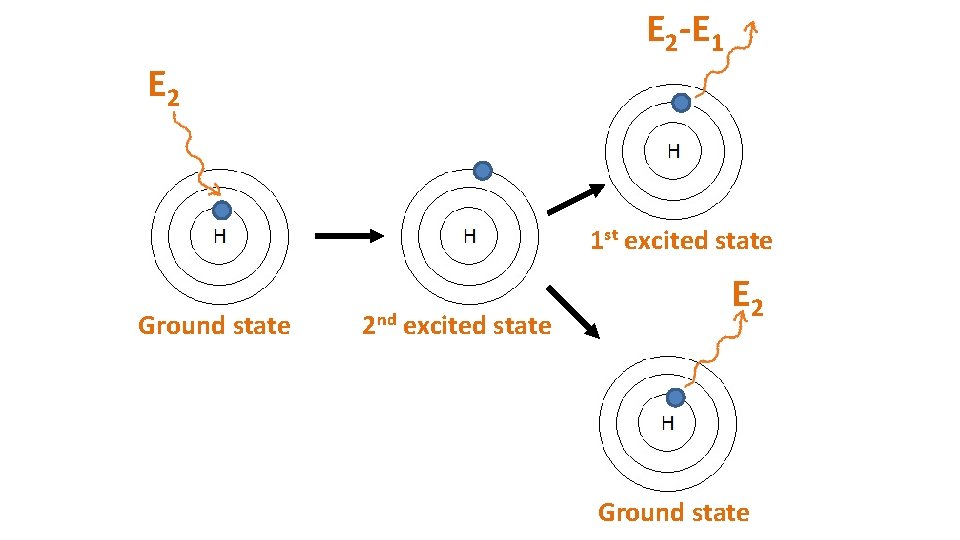
E 2 -E 1 E 2 1 st excited state Ground state 2 nd excited state E 2 Ground state


When particles are exposed to energy • ground state (lowest energy state) atoms absorb specific energy and become “excited” – an electron moves to a higher shell • excited atoms then release the energy at specific frequencies to get back to ground state – the electron moves back to its original shell, emitting a photon (packet of light) • the difference between two particular energy levels is called the quantum • the lines in the spectrum are produced when electrons de-excite
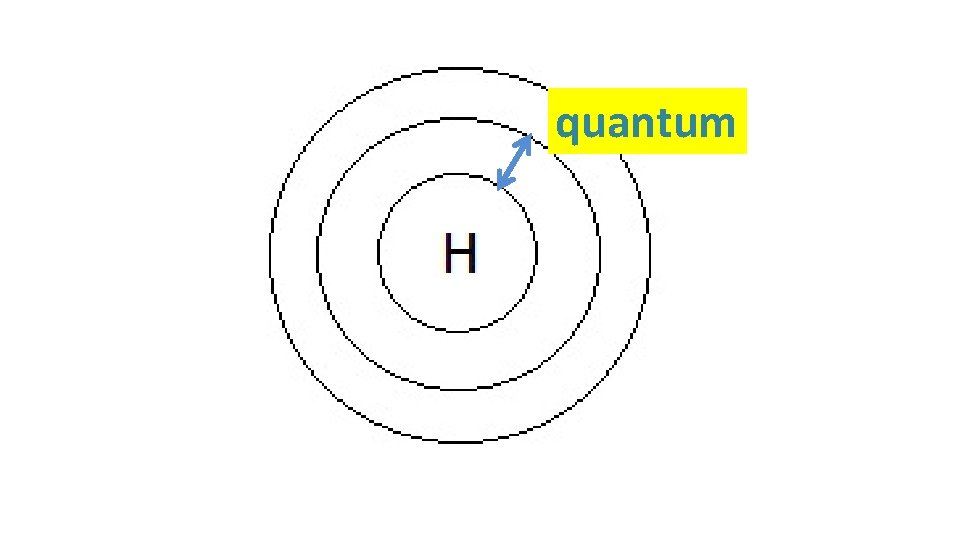
quantum

Bohr’s model was proven significantly wrong in 2 ways: – electrons don’t revolve around the nucleus in circles – the model can only predict energies in H

Recap What does “ground state” refer to? A. B. C. D. A hydrogen atom Lowest energy state of an atom Highest energy state of an atom When the atom is on the ground

Recap How does an electron become “excited? ” A. B. C. D. The atom it resides in reacts with another atom It absorbs energy and jumps shells It absorbs specific amounts of energy and jumps shells When it sees a particle of opposite charge

Recap What is a photon? A. B. C. D. A packet of light An excited proton An excited electron A Chinese wonton stuffed with Vietnamese noodles
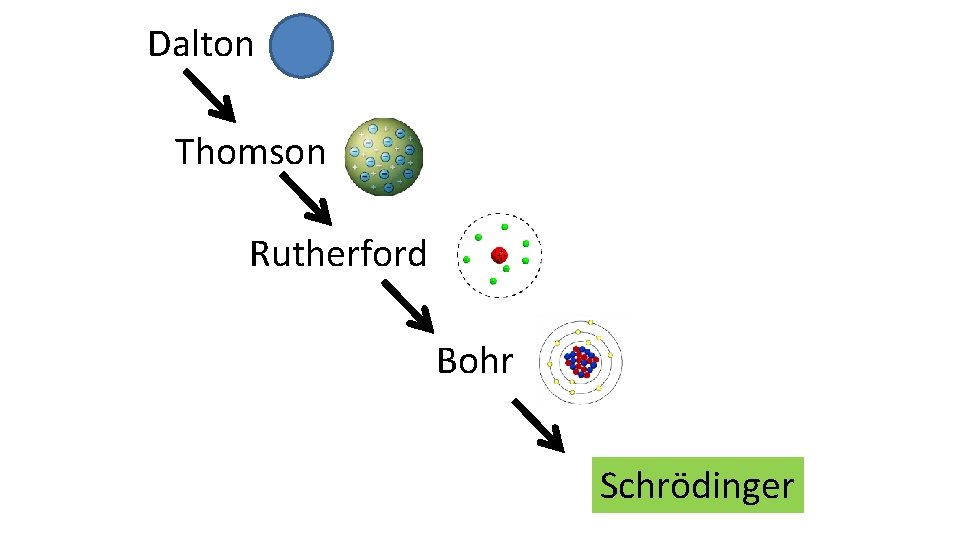
Dalton Thomson Rutherford Bohr Schrödinger


Schrödinger’s model describes the probability of where to find an electron in an atom • orbital: the region of space around a nucleus where an electron can be found • orbitals are described by quantum numbers
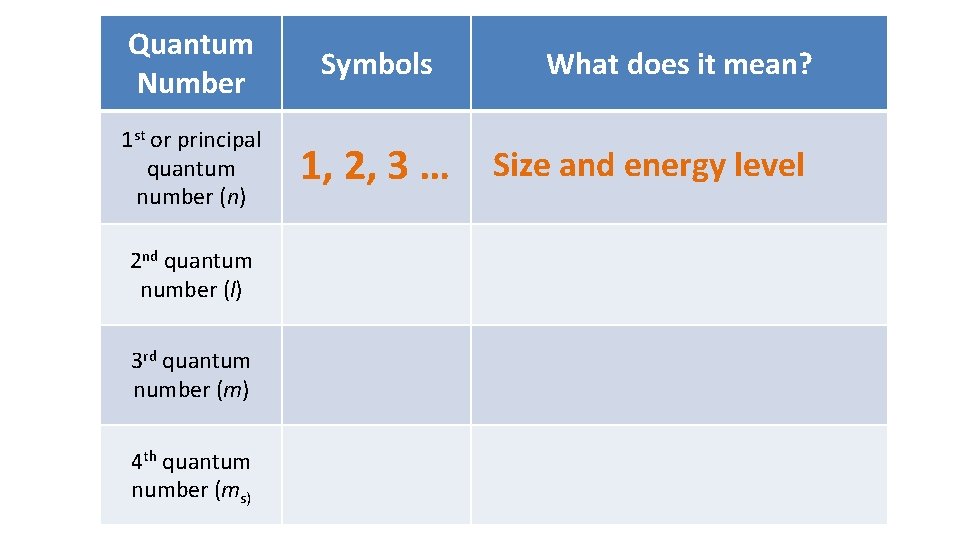
Quantum Number 1 st or principal quantum number (n) 2 nd quantum number (l) 3 rd quantum number (m) 4 th quantum number (ms) Symbols What does it mean? 1, 2, 3 … Size and energy level
2 s

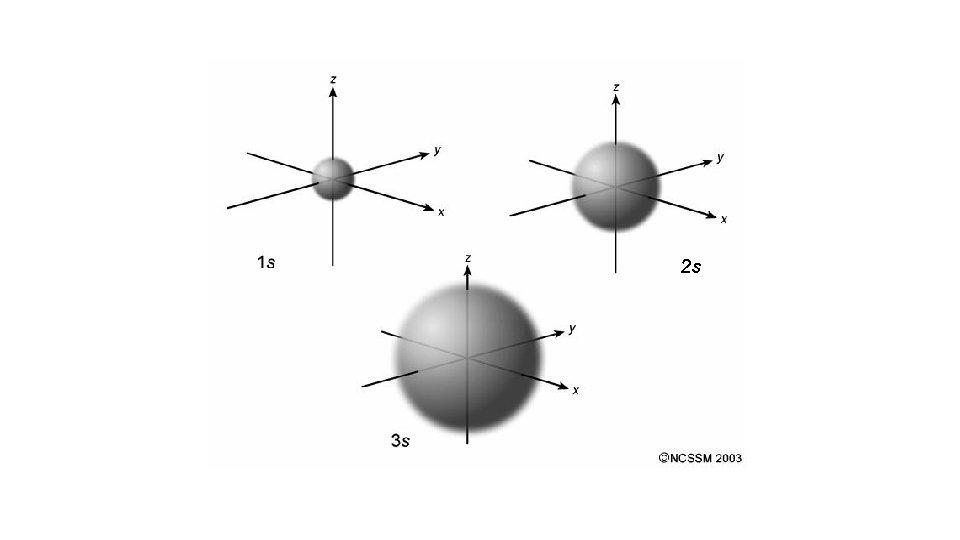
Quantum Number 1 st or principal quantum number (n) 2 nd quantum number (l) 3 rd quantum number (m) 4 th quantum number (ms) Symbols What does it mean? 1, 2, 3 … Size and energy level s, p, d, f Orbital shape
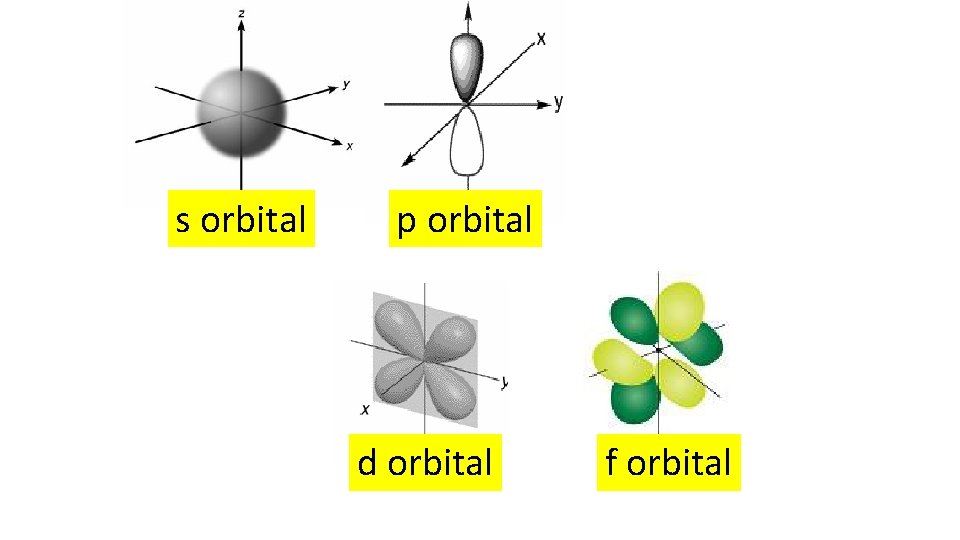
s orbital p orbital d orbital f orbital
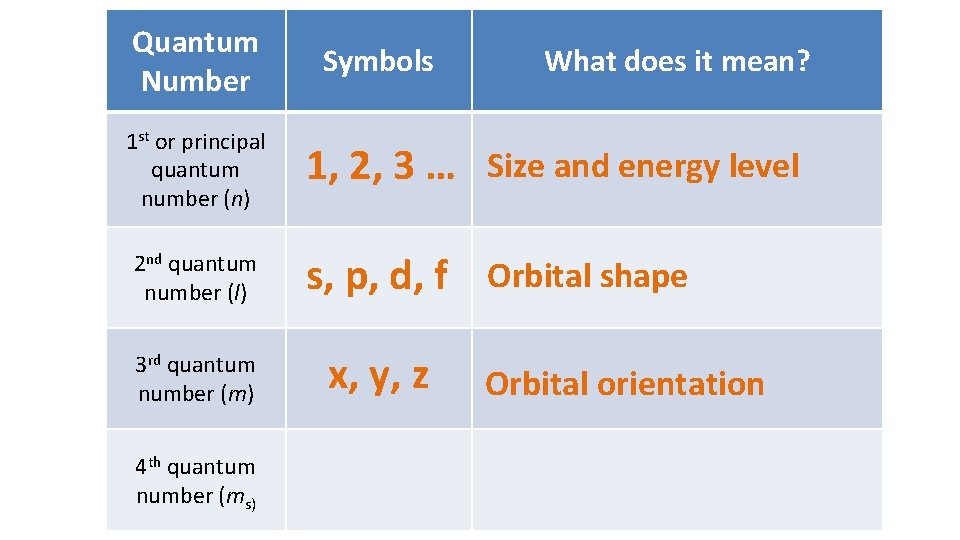
Quantum Number 1 st or principal quantum number (n) 2 nd quantum number (l) 3 rd quantum number (m) 4 th quantum number (ms) Symbols What does it mean? 1, 2, 3 … Size and energy level s, p, d, f Orbital shape x, y, z Orbital orientation
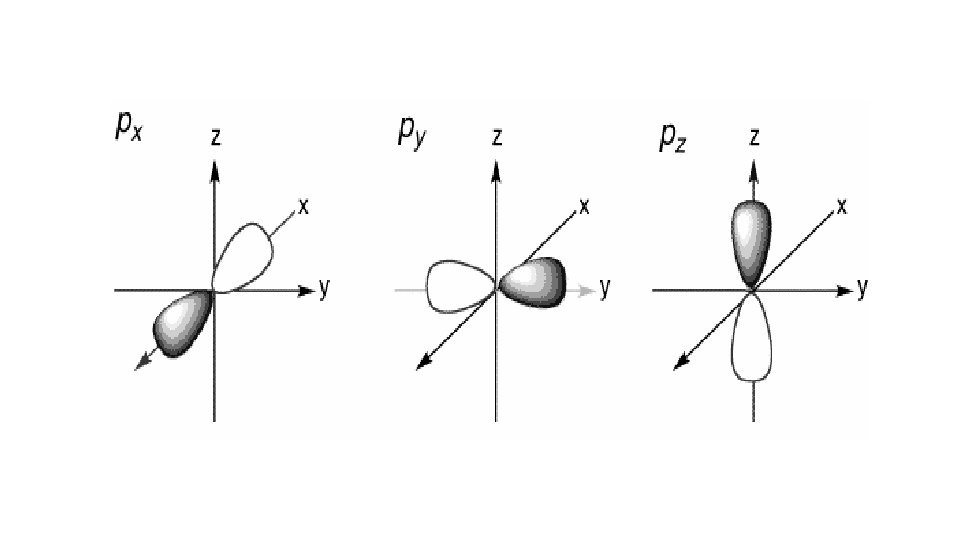
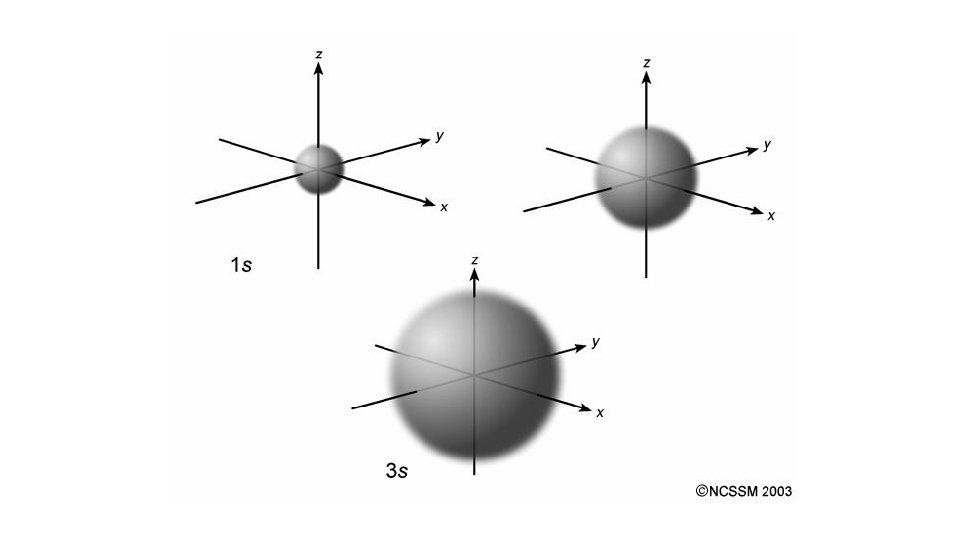
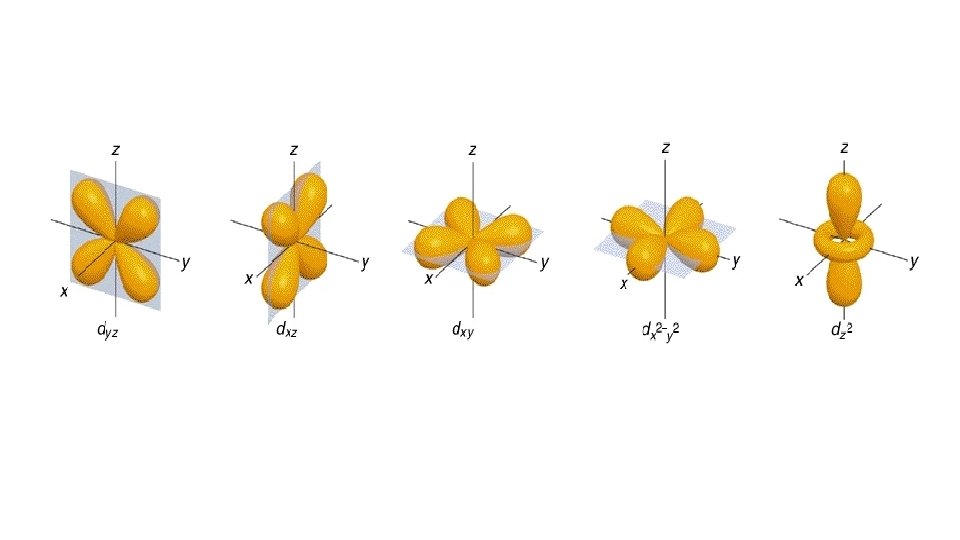
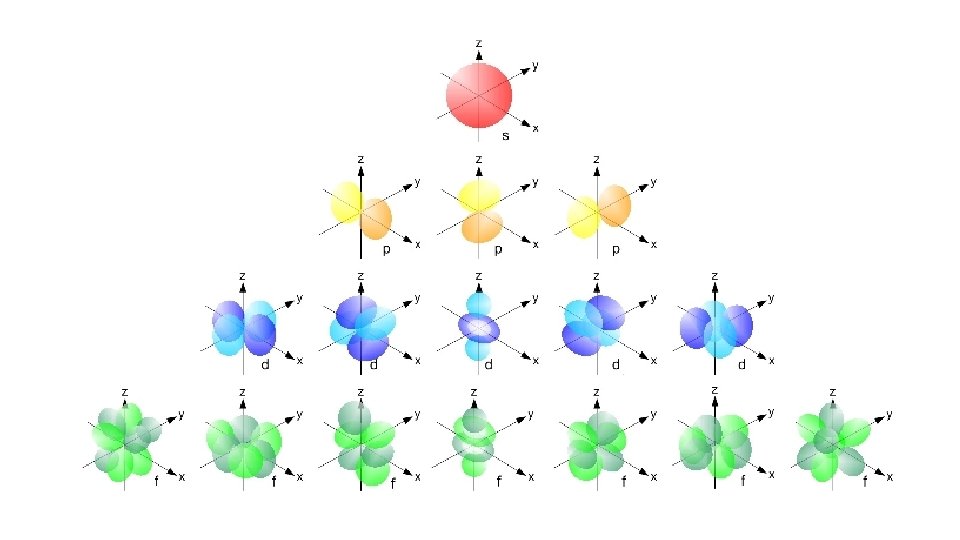

Quantum Number 1 st or principal quantum number (n) 2 nd quantum number (l) Symbols What does it mean? 1, 2, 3 … Size and energy level s, p, d, f Orbital shape 3 rd quantum number (m) x, y, z 4 th quantum number (ms) ½ or -½ Orbital orientation Electron spin

Each energy level has a specific set of orbitals and each one represents where a maximum of 2 electrons can be found. Orbital Type Begins at n= # of Orbitals in a Subshell s p d f 1 2 3 4 1 3 5 7 Maximum # of Electrons in Subshell 2 6 10 14

Recap What is an orbital? A. B. C. D. The path an electron takes around the nucleus Lobes The shape of the nucleus Mathematical description (probability) of where to find an electron in an atom
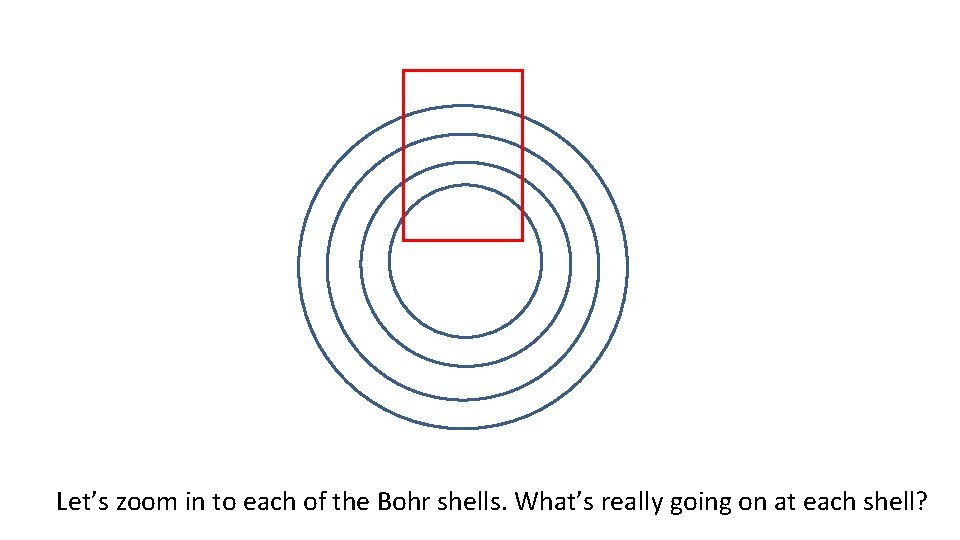
Let’s zoom in to each of the Bohr shells. What’s really going on at each shell?
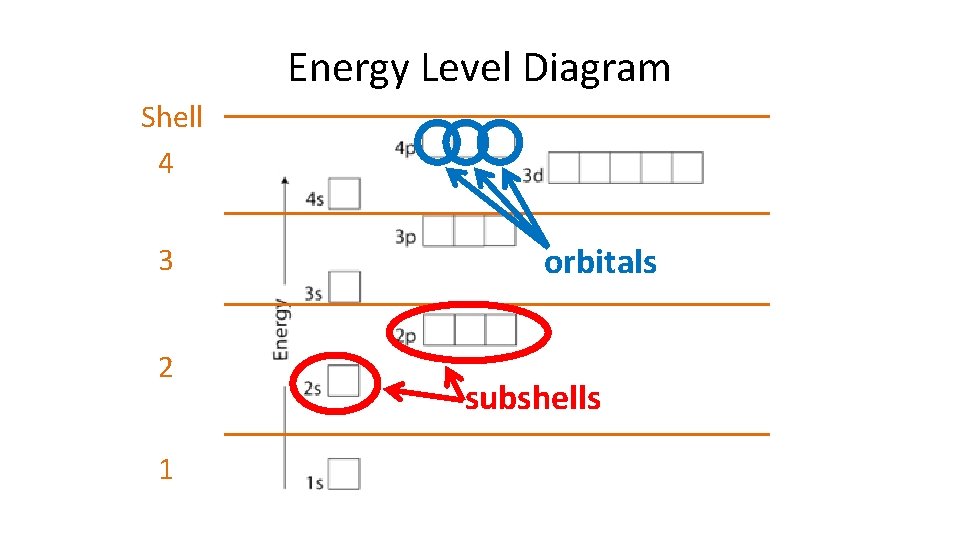
Energy Level Diagram Shell 4 3 2 1 orbitals subshells

Rules to follow when filling orbitals: 1. Fill orbitals from lowest to highest energy (Aufbau Principle) 2. Place one electron in each orbital of a sub-shell 3. When each orbital of a sub-shell has one electron, go back and pair the electrons (Hund’s Rule) 4. If two electrons are in a orbital, they must have opposite spin (Pauli Exclusion Principle) These rules ensure that the electron configuration gives the ______ stable atom by reducing lowest energy, most _____________________. electron-electron repulsion
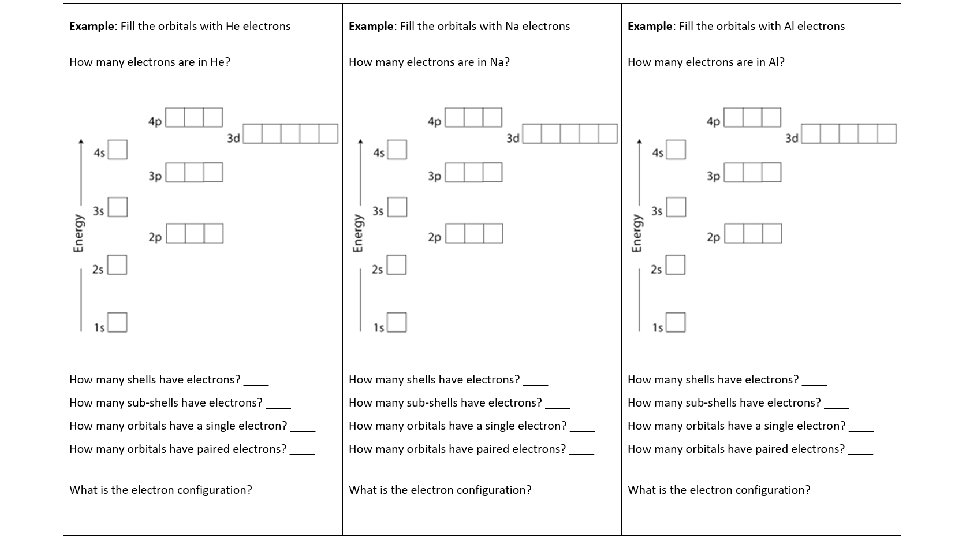

More About Shells Shell 4 3 2 1 2 2 6 2 10 6 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 2 8 8 18
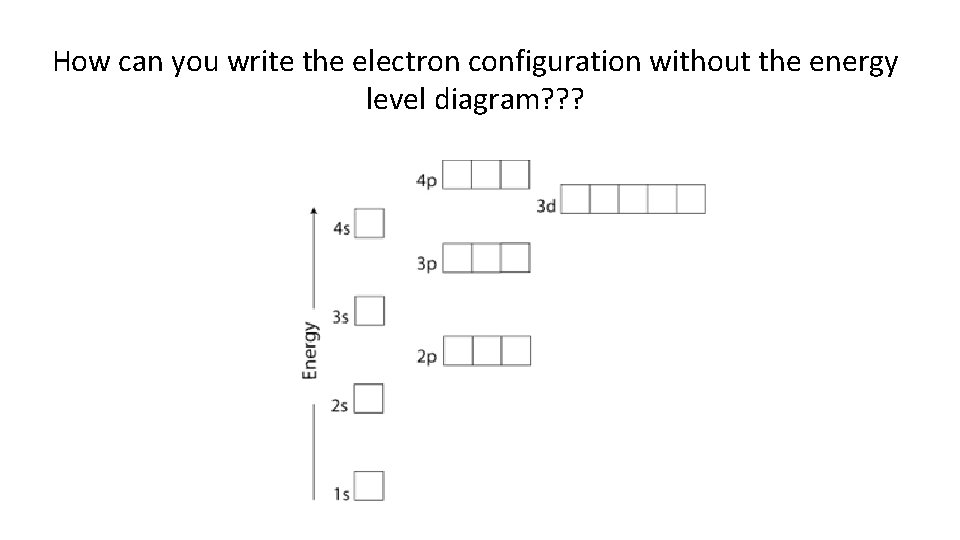
How can you write the electron configuration without the energy level diagram? ? ?
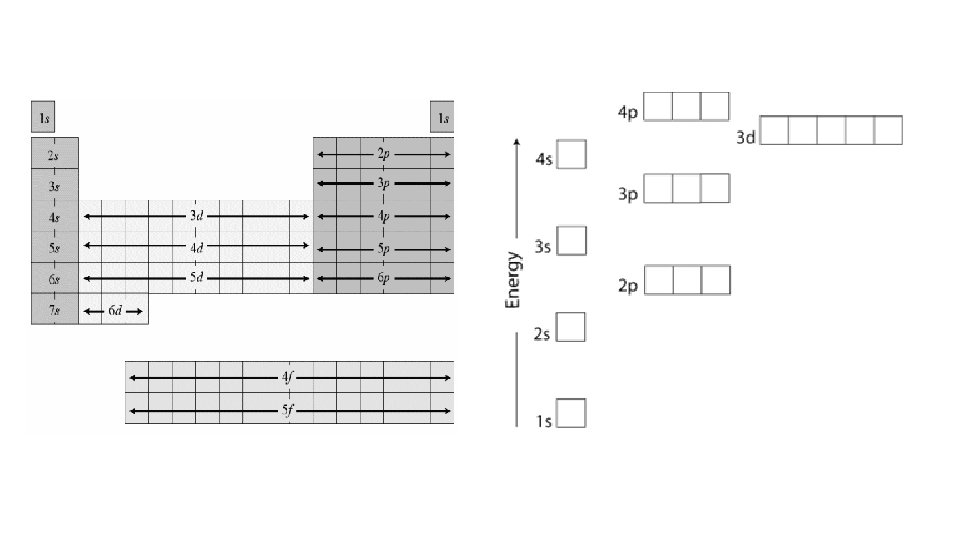
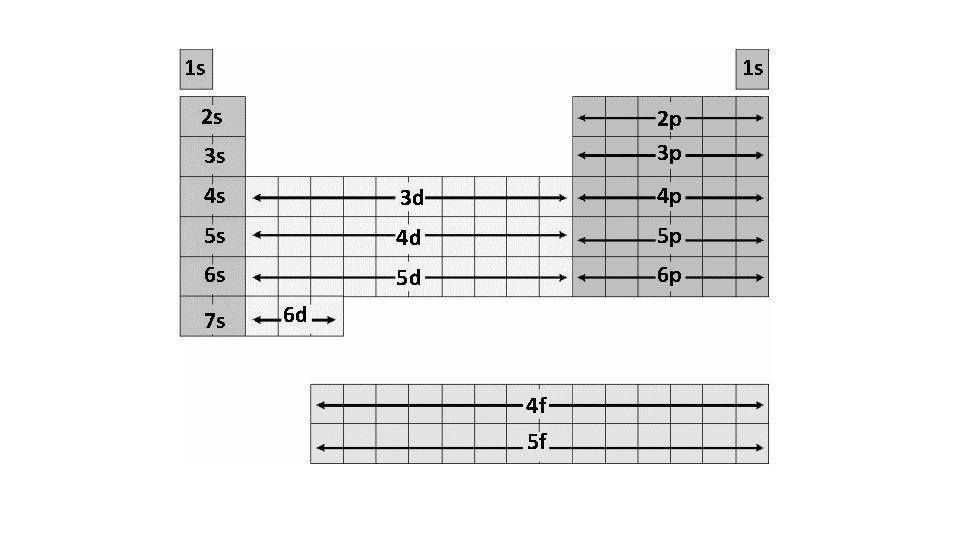
1 s 1 s 2 s 2 p 3 p 3 s 4 s 3 d 4 p 5 s 4 d 5 p 6 s 5 d 6 p 7 s 6 d 4 f 5 f
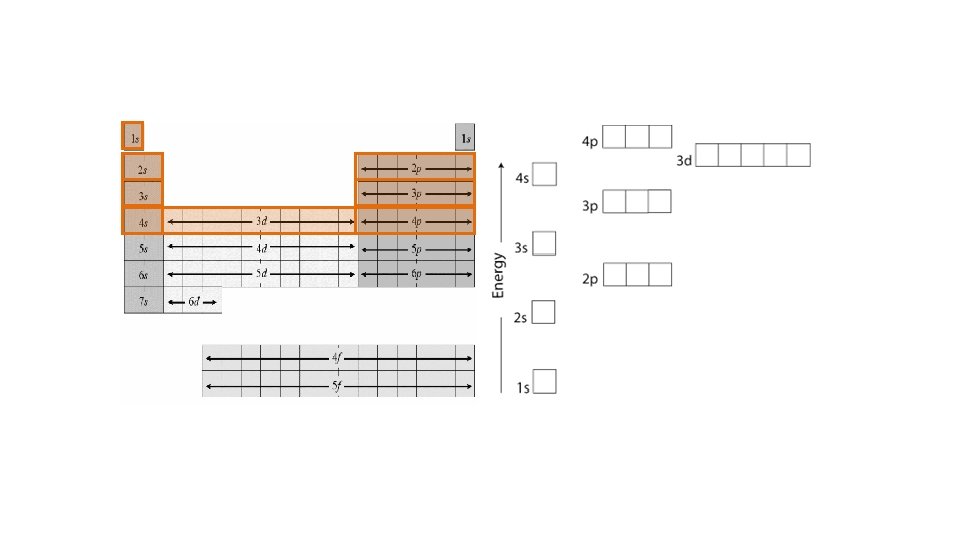
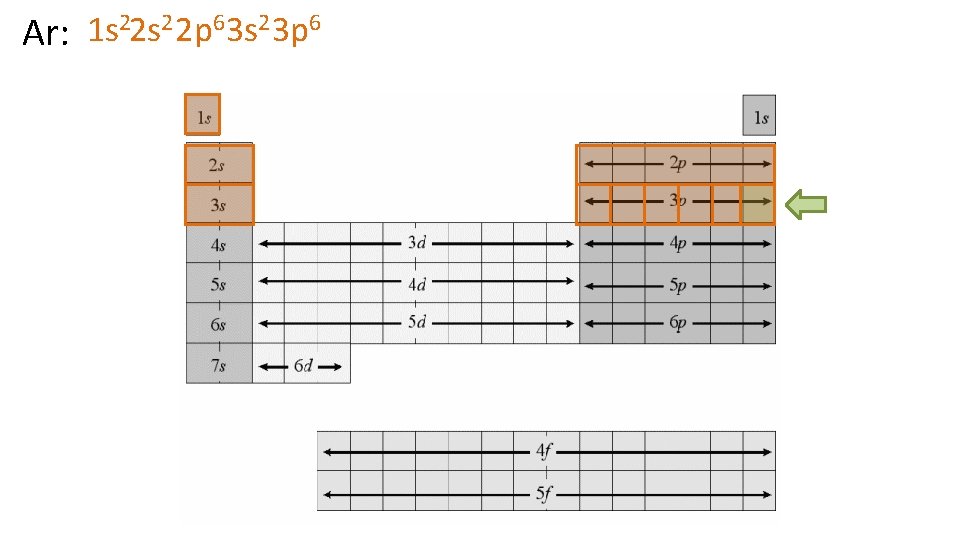
22 s 2 2 p 6 3 s 2 3 p 6 14 2 3 5 1 s Ar:
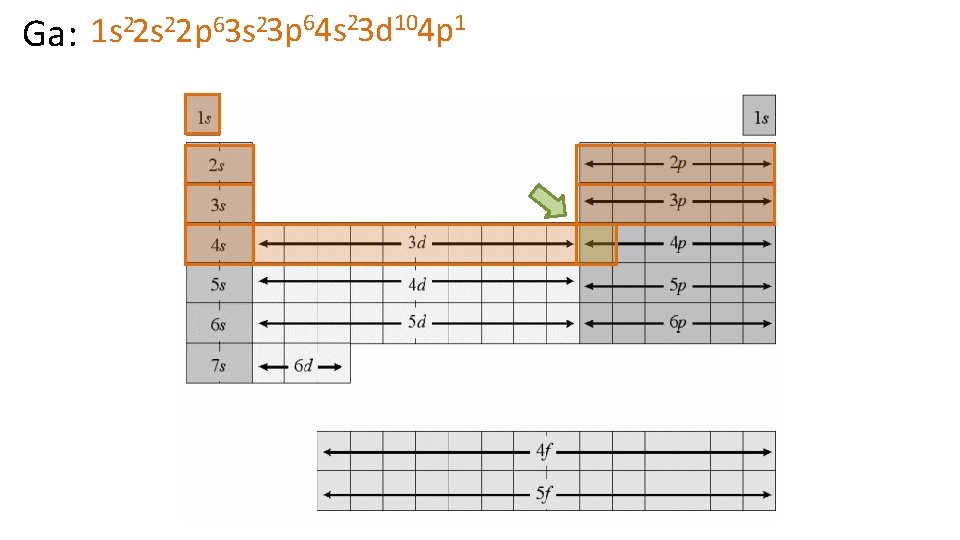
22 s 22 p 63 s 23 p 64 s 23 d 104 p 1 1 s Ga:
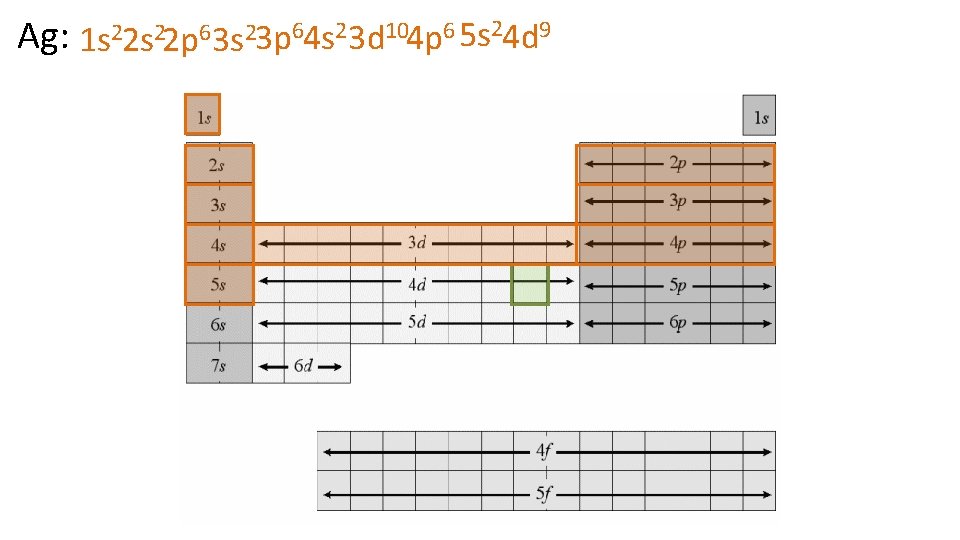
Ag: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 6 5 s 24 d 9
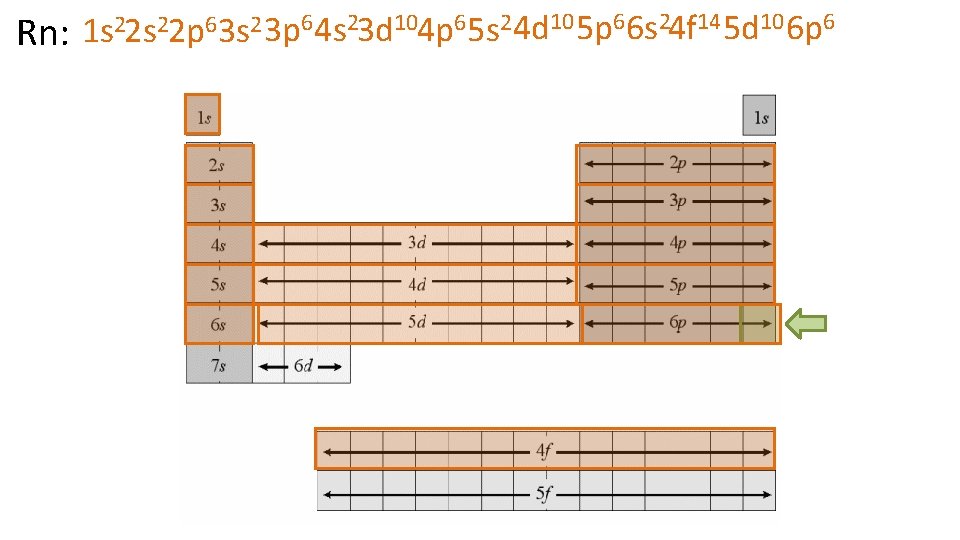
22 s 22 p 6 3 s 2 3 p 6 4 s 23 d 104 p 6 5 s 2 4 d 10 5 p 6 6 s 24 f 14 5 d 10 6 p 6 1 s Rn:
![K Ti Ni Ga Kr 1 2 2 6 [Ar] 4 s 1 s K Ti Ni Ga Kr 1 2 2 6 [Ar] 4 s 1 s](http://slidetodoc.com/presentation_image_h2/60a43cef807b82e79d87ecee5d906de8/image-51.jpg)
K Ti Ni Ga Kr 1 2 2 6 [Ar] 4 s 1 s 2 s 2 p 3 s 3 p 2 2 6 2 6 [Ar] 1 s 2 s 2 p 3 s 3 p 4 s 3 d 2 8 2 2 6 [Ar] 1 s 2 s 2 p 3 s 3 p 4 s 3 d 1 s 2 2 p 6 3 s 2[Ar] 3 p 6 4 s 2 3 d 10 4 p 1 2 10 6 2 2 6 [Ar] 4 s 3 d 4 p 1 s 2 s 2 p 3 s 3 p Ar
![5 2 [Ar]4 s 3 d 5 2 [Ar]4 s 3 d](http://slidetodoc.com/presentation_image_h2/60a43cef807b82e79d87ecee5d906de8/image-52.jpg)
5 2 [Ar]4 s 3 d

Example: Write the electron configuration for Ga using core notation. Closest previous noble gas element: ______ Ar 23 d 104 p 1 [Ar] 4 s Core notation: __________
![Practice: Write the electron configuration for the following elements. 23 d 10 [Ar]4 s Practice: Write the electron configuration for the following elements. 23 d 10 [Ar]4 s](http://slidetodoc.com/presentation_image_h2/60a43cef807b82e79d87ecee5d906de8/image-54.jpg)
Practice: Write the electron configuration for the following elements. 23 d 10 [Ar]4 s a) Zn ________________ 1 [Ar]4 s b) K ________________ 23 d 104 p 6 [Ar]4 s c) Kr ________________
- Slides: 54