Schoollocated Influenza Vaccination with Third Party Billing Outcomes

School-located Influenza Vaccination with Third Party Billing: Outcomes, Cost, and Reimbursement Children’s. Outcomes Research. Program The. Children’s. Hospital Aurora, Denver, CO CO Colorado Health Outcomes Program Univ. of Colorado Denver Aurora, CO 45 th National Immunization Conference March 31 st 2011 Allison Kempe, MD, MPH

Disclosures • The authors report no relevant financial interests regarding the work presented in this talk • This talk will not include any reference to products not licensed by the Food and Drug Administration

Acknowledgments Denver Public Health Judith C. Shlay, MD, MSPH (PI) Mette Riis, RN, MSW Sarah Rodgers, BA Denver Health and Hospitals Deborah Rinehart, MA Nicole Morgan, MHA Simon J. Hambidge, Ph. D, MD Anne Hammer, BSN, RN U. Colorado/Children’s Hospital Allison Kempe, MD, MPH Jennifer Pyrzanowski, MSPH Hai Fang, Ph. D, MPH Elizabeth Campagna, MS Karen Albright, Ph. D Miriam Dickinson, Ph. D Denver Public Schools Donna Shocks, MSN, CNS, RN Jean Lyons, BSN, RN Scott Romero, MS Linda Parker-Long, RN Bridget Beatty, MPH School paraprofessionals School nurses Local school administrations Kaiser Permanente Colorado Matthew F. Daley, MD Monica Buhlig, MPH Community Benefits Program Centers for Disease Control and Prevention Tara Vogt, Ph. D Emily Mc. Cormick, MPH

Background • Since 2008 influenza vaccination has been recommended annually for children ≥ 6 months of age • Given the increased number of visits required for universal vaccination, may not be realistic to accomplish in the medical home alone w/o additional delivery sites • School-located influenza vaccine programs may be instrumental in achieving universal influenza vaccine delivery in school-age children

Background • A variety of school-located influenza vaccine programs have been described: Hawaii, 2008; Minnesota, 2006; Tennessee, 2005, and Maryland, 2005 • However, previous school-located programs have either relied on donated influenza vaccine or focused efforts on VFC-eligible children alone Ref: Carpenter et al. , Pediatrics, 2007; Effler et al. , Emerg Infec Dis, 2010

Study Objectives • For a school-located influenza vaccine program with 3 rd party billing: – Assess feasibility and effectiveness • Percent of students receiving ≥ 1 doses of influenza vaccine • Percent fully vaccinated against seasonal influenza – Assess measures related to sustainability • Estimated cost of conducting the program • Reimbursement from 3 rd party payers for vaccine and administration costs

Study Methods • Time period: 11/2009 – 3/2010 • Setting: Urban public school district in Denver – 20 elementary schools targeted – Total enrolled 9, 814 students; range 305 – 655 students per school – Eligibility for free and reduced lunch: Median 87%; range 25% to 98%

School-Located Vaccine (SLV) Program • Community vaccinator (Denver Public Health) – Delivered vaccines to consented students at participating elementary schools – Billed 3 rd party payers for services delivered • Two clinics held per school – Seasonal influenza vaccine offered at both clinics • LAIV and TIV vaccines – H 1 N 1 injectable vaccine offered at 2 nd clinic only

School-located Vaccine (SLV) Program Pre-Clinic ØStudents brought Clinic ØStudents escorted Post Clinic ØBilled for vaccines consents to parents by grade to clinic given (separate consents during school hours ØBills resubmitted multiple for seasonal and ØStudents’ identity times if necessary H 1 N 1 vaccines) verified by ØReasons for denial Ø Health insurance & vaccinator captured vaccine records ØVaccine ØParents not billed for requested as part of administered vaccine or administration consent ØVaccines given fees if claim denied or ØIf consented, vaccine entered into not insured registries & insurance registry checked

Program Cost Estimates • Cost estimated by micro-costing approach: – Collected cost data on immunization-related activities • Pre-clinic, and post clinic activities • Costs of immunization-related supplies • Time to perform billing – Collected costs for each participating entity • Community vaccinator • School district

Program Cost Estimates • Cost outcomes – Cost/vaccine – Percent of costs reimbursed within each insurance type for: • Vaccine purchasing costs • Vaccine administration costs • Total = Vaccine and administration costs combined

Results: Participants vs. Non-Participants • Age: Slightly younger – 68% participants were <9 years of age vs. 66% of non-participants ≥ 9 years of age (p=0. 025) • Race/ethnicity: Higher participation among self -identified Hispanic – 76% participants vs. 61% non-participants were Hispanic (p <0. 001) • Correspondence language more likely Spanish – 56% participants vs. 34% non-participants reported a correspondence language of Spanish (p < 0. 001)
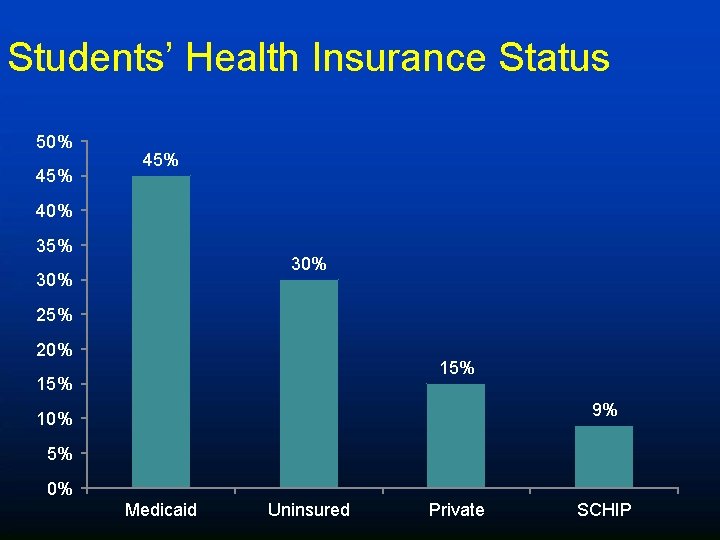
Students’ Health Insurance Status 50% 45% 40% 35% 30% 25% 20% 15% 9% 10% 5% 0% Medicaid Uninsured Private SCHIP

Results: Consent Process • 31% (3, 007) of enrolled students returned a consent form – Seasonal: overall 23% (2, 248); range 0% - 46% – H 1 N 1: overall 18% (1, 725); range 4% - 34% • 93% (2, 787) of consented students were vaccinated with one or more influenza vaccines
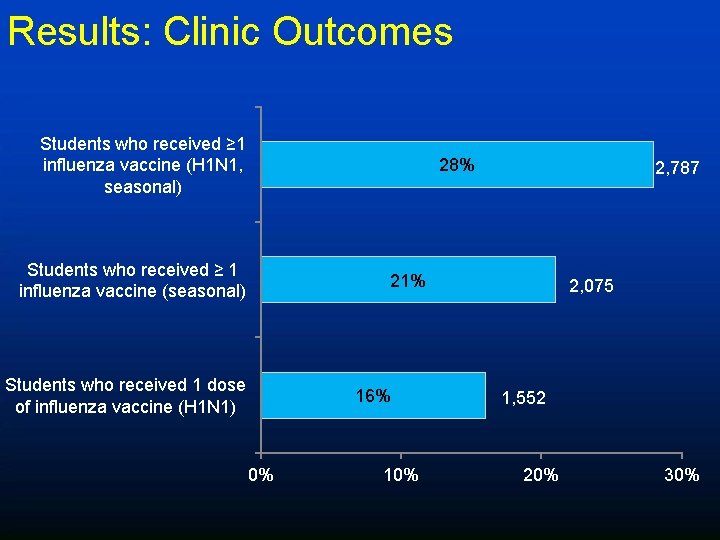
Results: Clinic Outcomes Students who received ≥ 1 influenza vaccine (H 1 N 1, seasonal) 28% Students who received ≥ 1 influenza vaccine (seasonal) 2, 787 21% Students who received 1 dose of influenza vaccine (H 1 N 1) 16% 0% 10% 2, 075 1, 552 20% 30%

Results: Clinic Outcomes • Among participants who received their 1 st dose at clinic 1 and required 2 nd dose, 82% received 2 nd dose • 93% of 1 st Clinic participants received LAIV vaccine, 7% received TIV

Costs to Administer Clinics Entity Description Community Vaccinator $16. 41 ØPre-clinic: printing, distributing, collecting, and reviewing consent forms, updating vaccinators’ vaccine registry, office/administration supplies, communication with families, translation resources (46%) ØDay of clinic: RN hours, clerical hours, transportation, medical supplies (39%) ØPost –clinic: registering patients, submitting claims, follow-up of denied claims (15%) School Total Cost/vaccine $5. 30 ØPre-clinic: distributing, collecting, and reviewing consent forms (86%) ØDay of clinic: support staff to escort students, school nurse support (14%) $21. 72

Costs to Administer Clinics Entity Description Community Vaccinator $16. 41 ØPre-clinic: printing, distributing, collecting, and reviewing consent forms, updating vaccinators’ vaccine registry, office/administration supplies, communication with families, translation resources 46% ØDay of clinic: RN hours, clerical hours, transportation, medical supplies 39% ØPost –clinic: registering patients, submitting claims, follow-up of denied claims 15% School Total Cost/vaccine $5. 30 ØPre-clinic: distributing, collecting, and reviewing consent forms, communication with parents 86% ØDay of clinic: support staff to escort students, school nurse support 14% $21. 72
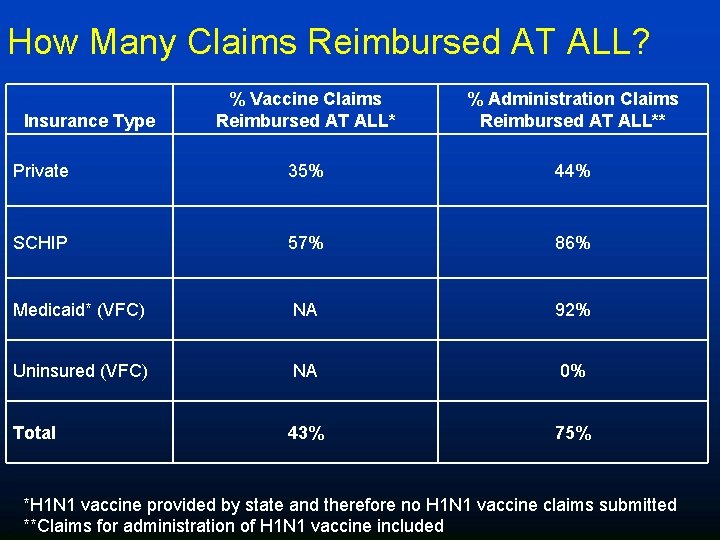
How Many Claims Reimbursed AT ALL? % Vaccine Claims Reimbursed AT ALL* % Administration Claims Reimbursed AT ALL** Private 35% 44% SCHIP 57% 86% Medicaid* (VFC) NA 92% Uninsured (VFC) NA 0% 43% 75% Insurance Type Total *H 1 N 1 vaccine provided by state and therefore no H 1 N 1 vaccine claims submitted **Claims for administration of H 1 N 1 vaccine included
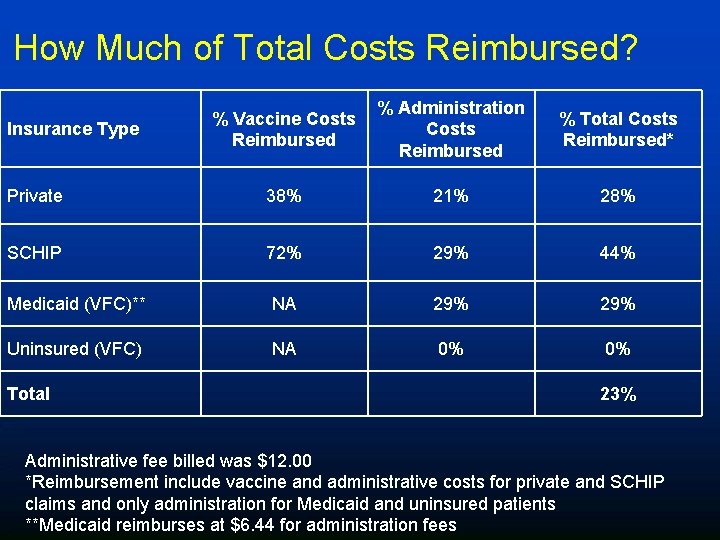
How Much of Total Costs Reimbursed? % Vaccine Costs Reimbursed % Administration Costs Reimbursed % Total Costs Reimbursed* Private 38% 21% 28% SCHIP 72% 29% 44% Medicaid (VFC)** NA 29% Uninsured (VFC) NA 0% 0% Insurance Type Total 23% Administrative fee billed was $12. 00 *Reimbursement include vaccine and administrative costs for private and SCHIP claims and only administration for Medicaid and uninsured patients **Medicaid reimburses at $6. 44 for administration fees

Reasons for Denial of Private Claims Administration Claim Denials (n = 212) • Patient’s deductible (34%) • Service provided out of network (17%) • Service not covered under benefit plan (15%) • Claim not submitted properly (14%) • Charges exceed fee schedule (8%) • Benefit for this service included with payment for another routine procedure (6%) • Patient not covered (5%)

Limitations • No vaccine history available for non-participants; unknown whether program increased vaccine uptake or only shifted where vaccine received • H 1 N 1 pandemic occurred during implementation of program and may have influenced participation and cost of the program • Program began in November; late start may have influenced participation

Limitations • Some costs estimated not based on observation • Program costs collected during start-up year are likely higher than in subsequent years • Billing database collected limited data related to denial of claims

Challenges to SLV with 3 rd Party Billing • Competing demands placed on schools – Importance of “seat time” • Insurance – Lack of recognition by insurers of SLV clinics as acceptable venue for provision of vaccination services – Difficult to obtain insurance information from families – Time consuming to verify insurance information – Limited reimbursement by insurers • Required versus recommended vaccines • Resistance to billing families for unpaid claims in a public school setting

Challenges to School-Located Vaccination • Family Educational Rights and Privacy Act (FERPA) – Increased costs of conducting program • Limited ability to access student contact information and immunization records • Hindered ability to perform a more targeted intervention – Complicated process for obtaining and completing consents

Conclusions • School-located influenza vaccine program was feasible and effective reaching 30% in year 1 • Uptake of services greater for uninsured or Medicaid-insured students • Medicaid reimbursement rates ($6. 44) not adequate to cover administration costs • Reimbursement from private insurers for vaccine and administration inadequate (year 1) • Reimbursement will need to be improved and efficiency increased in order to make this approach financially sustainable

Funding This investigation was supported by Cooperative Agreement #1 U 01 P 000199 -01 from the Centers for Disease Control and Prevention. The findings and conclusions are solely the responsibility of the authors, and do not necessarily represent the official views of the Centers for Disease Control and Prevention.

Questions?

Summary of Findings • In its first year, over 30% of students consented to participate in the program and almost all received ≥ 1 influenza vaccine at school • Among participants in need of 2 doses, 80% received both vaccines • Almost half of those who participated were insured by Medicaid and nearly 1/3 of participants had no insurance

Summary of Findings • Total cost in this 1 st year was $21. 72 per vaccine • Vaccine reimbursement from private and SCHIP 3 rd party payers was less than cost of purchasing vaccines • Administration reimbursement – Payment from private insurers and SCHIP occurred for < ½ of claims submitted resulting in coverage <30% of administration costs – Medicaid paid $6. 44 for almost all claims submitted resulting in coverage of ≈30% administration costs
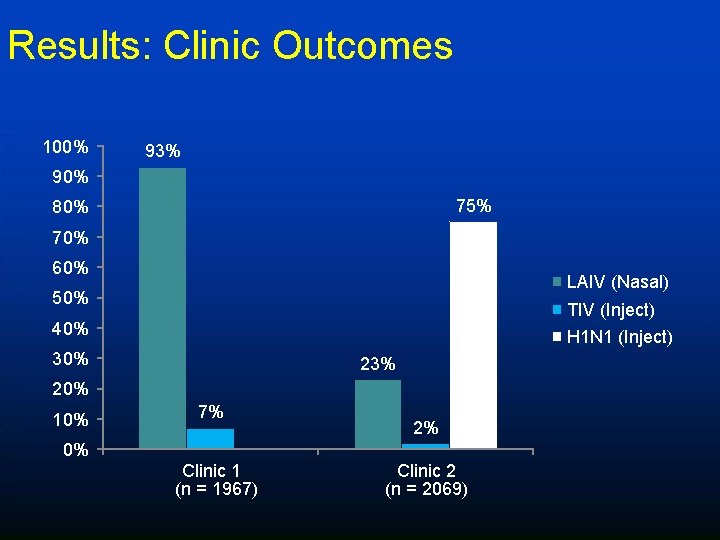
Results: Clinic Outcomes 100% 93% 90% 75% 80% 70% 60% LAIV (Nasal) 50% TIV (Inject) 40% H 1 N 1 (Inject) 30% 23% 20% 10% 7% 2% 0% Clinic 1 (n = 1967) Clinic 2 (n = 2069)

Results: Clinic Outcomes 26% (518) of clinic 1 participants required a 2 nd dose of seasonal influenza vaccine *Among clinic 1 participants who received their 1 st dose at clinic 1 and required a 2 nd dose, 82% (382/465) received a 2 nd dose 10% (53) received 2 nd dose at 1 st clinic 90% (465) received 1 st dose at 1 st clinic 79% (369) received a 2 nd seasonal influenza vaccine at 2 nd clinic 3% (13) received a 2 nd dose prior to 2 nd clinic elsewhere 18% (83) not vaccinated at 2 nd clinic; no record of vaccine received elsewhere
- Slides: 32