SC 6 Obtain evaluate and communicate information about

SC 6. Obtain, evaluate, and communicate information about the properties that describe solutions and the nature of acids and bases. f. Use mathematics and computational thinking to compare, contrast, and evaluate the nature of acids and bases in terms of percent dissociation, hydronium ion concentration, and p. H. g. Ask questions to evaluate merits and limitations of the Arrhenius and Bronsted-Lowry models of acid and bases. h. Plan and carry out an investigation to explore acid-base neutralization.

PROPERTIES OF ACIDS AND BASES

Properties of Acids Taste sour Can be strong or weak electrolytes in aqueous solution

Properties of Acids React with metals to form a salt and hydrogen gas Zn + 2 HCl(aq) Zn. Cl 2 + H 2 http: //www. launc. tased. edu. au/online/sciences/Phys. Sci/pschem/acidbase/acid. Cu. swf React with carbonates and bicarbonates to produce a salt, water, and carbon dioxide gas 2 HCl + Ca. CO 3 Ca. Cl 2 + H 2 O + CO 2 http: //www. launc. tased. edu. au/online/sciences/Phys. Sci/pschem/acidbase/cuco 3. swf
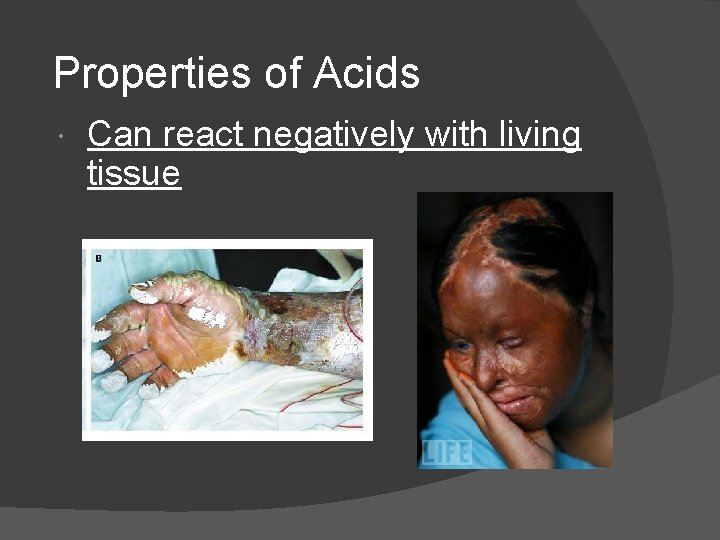
Properties of Acids Can react negatively with living tissue

Effects of Acid Rain on Marble (marble is calcium carbonate) George Washington: BEFORE acid rain George Washington: AFTER acid rain

n Change the color of indicators Blue litmus paper turns red in contact with acid (red paper stays red) Other liquid indicators are typically reddish when mixed with acid

Properties of Acids React with bases to form water and a salt (more about this a little later…) Have a p. H < 7 (more about this a little later…) How do you know if a chemical is an acid? It usually starts with hydrogen HCl, H 2 SO 4, HNO 3, etc. (but not water!)

Examples of Acids Ø sulfuric acid, H 2 SO 4 (battery acid) hydrochloric acid, HCl (stomach acid) Ø acetic acid, CH 3 COOH (vinegar) Ø nitric acid, HNO 3 (fertilizer production) Ø

Properties of Bases Taste bitter Most natural toxins are alkaline (basic): the bitter taste is a warning Can be strong or weak electrolytes in aqueous solution
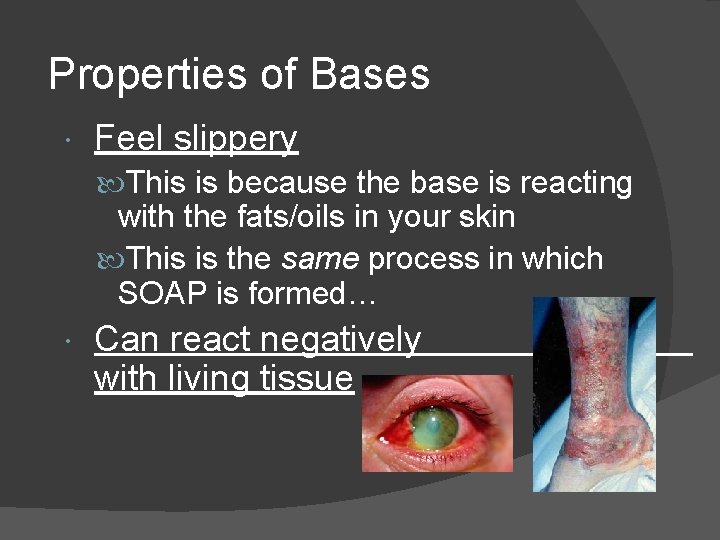
Properties of Bases Feel slippery This is because the base is reacting with the fats/oils in your skin This is the same process in which SOAP is formed… Can react negatively with living tissue

n Change the color of indicators Red litmus paper turns Other liquid indicators blue in contact with base are typically bluish (blue paper stays blue) when mixed with acid

Properties of Bases React with acids to form water and a salt Have a p. H > 7 How do you know if a chemical is a base? It usually ends with hydroxide Na. OH, Ca(OH)2

Examples of Bases sodium hydroxide, Na. OH (drain cleaner) Ø potassium hydroxide, KOH (alkaline batteries, fertilizer) Ø calcium hydroxide, Ca(OH)2 (masonry, nolye relaxers) Ø ammonia, NH 3 (fertilizer, cleaning) Ø

Acids Neutralize Bases HCl + Na. OH → Na. Cl + H 2 O Neutralization reactions ALWAYS produce a salt (which is an ionic compound made of the base cation and the acid anion) and water.

Bases Neutralize Acids Milk of Magnesia contains magnesium hydroxide, Mg(OH)2, which neutralizes stomach acid, HCl. 2 HCl + Mg(OH)2 Mg. Cl 2 + 2 H 2 O

ACID-BASE THEORIES Arrhenius § Brønsted-Lowry § Lewis §

Svante Arrhenius (1859 -1927)

Svante Arrhenius Swedish physicist/chemist Nobel Prize in Chemistry (1903) for his theory of ionic dissociation Chemical reactions in aqueous solutions are actually among ions of salts One of the first chemists to explain the chemical theory of the behavior of acids and bases
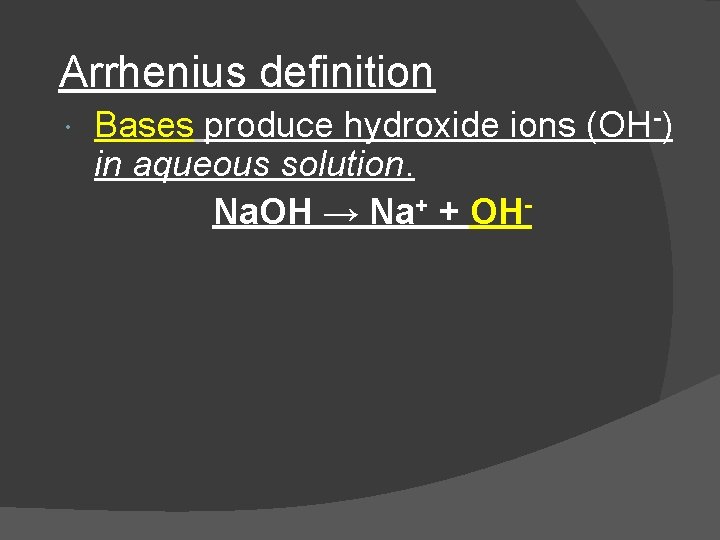
Arrhenius definition Bases produce hydroxide ions (OH-) in aqueous solution. Na. OH → Na+ + OH-

Arrhenius definition Acids produce hydrogen ions (H+) in aqueous solution HCl → H+ + Cl HOWEVER, H+ ions are never found alone in aqueous solution. They are ALWAYS bound to H 2 O H + + H 2 O H 3 O + So, the resulting equation looks like HCl + H 2 O → H 3 O+ + Cl H 3 O+ is called the hydronium ion.

Acids Not all compounds that have hydrogen are acids Also, not all the hydrogen in an acid may be released as ions H 2 SO 4 loses 1 H easily, but not the second

Organic Acids (those with carbon) Organic acids all contain the carboxyl group, (-COOH), sometimes several of them. CH 3 COOH – of the 4 hydrogen, only 1 ionizable (due to being bonded to the highly electronegative Oxygen) The carboxyl group is a poor H+ donor, so ALL organic acids are weak acids.

BREAKING NEWS! STRONG VERSUS WEAK

Strength of Acids and Bases Acids and bases are classified according to the degree to which they dissociate in water: Strong completely dissociate in aqueous solution; this means they ionize 100% HA H+ + ABOH B+ + OH Weak dissociate only slightly in aqueous solution HA + H+ + ABOH + B+ + OH-

Strength is different from concentration!

Johannes Brønsted (1879 -1947) Denmark Thomas Lowry (1874 -1936) England

Brønsted-Lowry definition Acids are H+ (or proton) donors. Bases are H+ acceptors. Acids DONATE the protons TO the base ACCEPTORS. HCl(g) + H 2 O(l) ↔ H 3 O+(aq) + Cl-(aq) Which is the Brønsted-Lowry acid? Which is the Brønsted-Lowry base?

Why Ammonia is a Base Ammonia can be explained as a base by using Brønsted-Lowry: NH 3(g) + H 2 O(l) ↔ NH 4+(aq) + OH-(aq) Ammonia is the H+ acceptor (base), and water is the H+ donor (acid). This causes the OH- concentration to be greater than in pure water, and the ammonia solution is basic

Brønsted-Lowry acids and bases come in pairs A conjugate base is the remainder of the B-L acid, after it donates its H+ A conjugate acid is the substance formed when the B-L base gains a H+ Thus, a conjugate acid-base pair is related by the loss or gain of a single hydrogen ion.

Conjugate Acids & Bases HCl + NH 3 ↔ Cl- + NH 4+ BLA BLB CB CA HCl + H 2 O ↔ H 3 O+ + Cl. NH 3 + H 2 O ↔ NH 4+ + OHAmphoteric – a substance that can act as both an acid and base (Water is an example!)

Gilbert Lewis (1875 -1946)

Lewis definition Not only did he discover the covalent bond, but he also developed the dot diagram notation for valence electrons. Lewis Acid - electron pair acceptor Lewis Base - electron pair donor Most general of all 3 definitions— acids don’t even need hydrogen!

HYDROGEN IONS AND ACIDITY Self-ionization of water § Ion-product constant § p. H/p. OH § Indicators §

Hydrogen Ions from Water To a very small extent, water selfionizes: 2 H 2 O ↔ H 3 O+ + OH− The concentration (molarity) of H+ (H 3 O+) ions in water [H+] is equal to the molarity of OH− ions [OH−]: [H+ ] = [OH−] = 1 x 10 -7 M

Hydrogen Ions from Water Since the molarities of the “acidic” ion and the “alkaline” ion are equal, a neutral solution results from water An equilibrium constant (called the ion product constant) is established for the self-ionization of water: Kw = [H+ ] x [OH−] = 1 x 10 -14 M 2
![Ion Product Constant So, we can calculate one molarity from the other… If [H+] Ion Product Constant So, we can calculate one molarity from the other… If [H+]](http://slidetodoc.com/presentation_image_h2/5b3524dec46d0949d08768a31e8a8ed1/image-37.jpg)
Ion Product Constant So, we can calculate one molarity from the other… If [H+] > 10 -7 M, then [OH−] < 10 -7 M If [H+] < 10 -7 M, then [OH−] > 10 -7 M [H+] = 10 -3 M, [OH−] = ? [OH−] = 2. 4 x 10 -9 M, [H+] = ?
![The p. H concept p. H = power of hydrogen p. H = −log[H+] The p. H concept p. H = power of hydrogen p. H = −log[H+]](http://slidetodoc.com/presentation_image_h2/5b3524dec46d0949d08768a31e8a8ed1/image-38.jpg)
The p. H concept p. H = power of hydrogen p. H = −log[H+] in neutral solution, [H+] = 1 x 10 -7 M −log(1 x 10 -7) = ? in acidic solution, [H+] > 1 x 10 -7 M −log(2. 3 x 10 -4) = ? in alkaline solution, [H+] < 1 x 10 -7 M −log(5. 6 x 10 -9) = ?
![The p. H concept What is the maximum [H+] value? What is the minimum The p. H concept What is the maximum [H+] value? What is the minimum](http://slidetodoc.com/presentation_image_h2/5b3524dec46d0949d08768a31e8a8ed1/image-39.jpg)
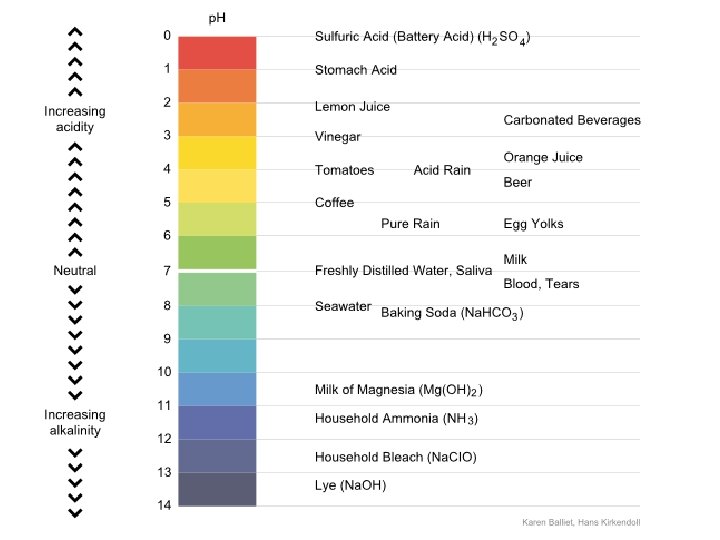
The p. H concept What is the maximum [H+] value? What is the minimum [H+] value? The p. H scale ranges from 0 -14, where 0 -6. 9 is acidic, 7. 0 is neutral, and 7. 1 -14 is alkaline
![The p. H concept You can determine the [H+] from p. H if you The p. H concept You can determine the [H+] from p. H if you](http://slidetodoc.com/presentation_image_h2/5b3524dec46d0949d08768a31e8a8ed1/image-40.jpg)
The p. H concept You can determine the [H+] from p. H if you use the p. H equation and solve for [H+]=log-1(-p. H) If the p. H = 2. 3, what is [H+]?

Calculating p. OH p. H + p. OH = 14 p. OH = −log [OH−] A solution with a p. OH > 7 is acidic. p. H = 3. 5, p. OH = ? , [H+] = ? , [OH−] = ? A solution with a p. OH < 7 is basic. p. H = 10. 2, p. OH = ? , [H+] = ? , [OH−] = ?

Why measure p. H? Everyday solutions we use…swimming pools, soil conditions for plants, medical diagnoses, soaps and shampoos, etc. Sometimes we can use indicators, other times we might need a p. H meter

Indicators are weak acids or bases that are differently colored than their conjugate. Phenolphthalein (a weak acid) is colorless. When it is combined with a base, the weak acid loses H+, and its conjugate base is pink.

Some of the many p. H indicators and their p. H range

Acid-Base Indicators Although useful, there are limitations to indicators: usually given for a certain temperature (25 o. C), thus may change at different temperatures what if the solution already has a color, like paint? the ability of the human eye to distinguish colors is limited

Acid-Base Indicators A p. H meter may give more definitive results some are large, others portable works because of electrolytes; typically accurate to within 0. 01 p. H unit of the true p. H instruments need to be calibrated often

NEUTRALIZATION REACTIONS Reaction Products § Titration § Buffers §

The general reaction is… Acid + Base Salt + Water This type of reaction is common: Taking antacids for tummy trouble Adjusting soil p. H for crops Natural cave structures Formation of kidney/gall stones

Titration is a laboratory technique used to determine the concentration (molarity) of an unknown analyte The concentration of acid (or base) in solution can be determined by performing a neutralization reaction

Titration An indicator is used to show when neutralization has occurred Often we use phenolphthalein because it is colorless in neutral and acid; turns pink in base The equivalence point is when the moles of hydrogen ions are equal to the moles of hydroxide ions

Titration Remember we did this (sort of…) via calculation with solutions – M 1 V 1 = M 2 V 2 known unknown This time, we know the volume of the acid and the molarity and volume of the base used to neutralize it. M 1 V 1 = M 2 V 2 acid base

Steps of a Titration 1. A measured volume of acid of unknown concentration is added to a flask 2. Indicator is added to the flask 3. A base of known concentration is slowly added via a buret, until the indicator changes color (the endpoint) 4. Measure the volume of base used

A Titration Problem…

Buffers are solutions in which the p. H remains relatively constant, even when small amounts of acid or base are added Buffer solutions use a pair of chemicals: a weak acid and one of its salts or a weak base and one of its salts

Buffers A buffer system is better able to resist changes in p. H than pure water Since it is a pair of chemicals: one chemical neutralizes any acid added, while the other chemical would neutralize any additional base AND, they produce each other in the process!

Buffers Example: Ethanoic (acetic) acid and sodium ethanoate (also called sodium acetate) The buffer capacity is the amount of acid or base that can be added before a significant change in p. H

Aspirin contains acetylsalicylic acid. A sideeffect of the chronic use of aspirin is stomach ulcers. Buffered aspirin is coated with a substance that helps neutralize the acetylsalicylic acid as it passes through the stomach.
- Slides: 58