sBlock Elements Group 1 Elements Alkali metals Electronic

s-Block Elements
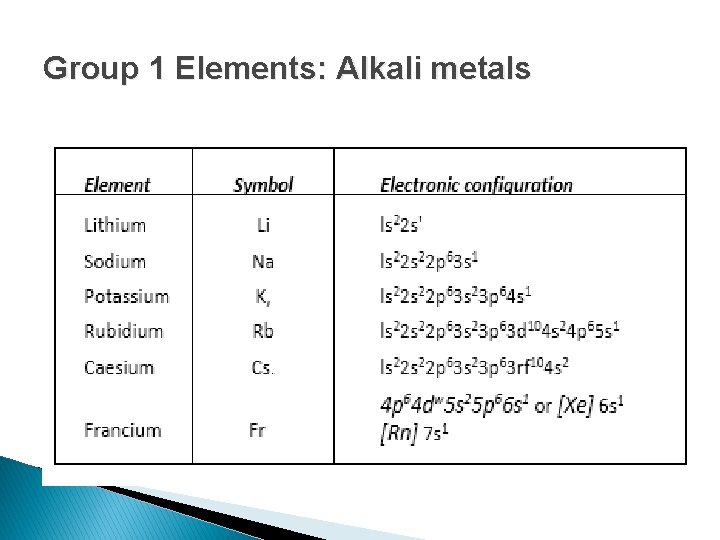
Group 1 Elements: Alkali metals

� Electronic Configuration, ns 1, where n represents the valence shell. � These elements are called alkali metals because they readily dissolve in water to form soluble hydroxides, which are strongly alkaline in nature

Atomic and Ionic Radii Atomic and ionic radii of alkali metals increase on moving down the group i. e. , they increase in size going from Li to Cs. Alkali metals form monovalent cations by losing one valence electron. Thus cationic radius is less as compared to the parent atom.

Diagonal Relationship of Lithium � Lithium shows diagonal resemblance with magnesium [the element of group 2] and this � resemblance is due to similar polarising power, i. e. , � [ionic charge / (ionic radius)2] of both these elements.

Lithium resembles magnesium in the following respects 1. The atomic radius of lithium is 1. 31 Å while that of magnesium is 1. 34 Å. � 2. The ionic radius of Li+i on is 0. 60 Å, which is very close to that of Mg 2+ ion (0. 65 Å). � 3. Lithium (1. 0) and magnesium (1. 2) have almost similar electronegativities. � 4. Both Li and Mg are hard metals. � 5. Li. F is partially soluble in water like Mg. F 2. � 6. Both combine with O 2 to form monoxides, e. g. , Li 2 O and Mg. O. � 7. Both Li. OH and Mg(OH)2 are weak bases. � 8. Both Li. CI and Mg. Cl 2 are predominantly covalent. � 9. Both Li and Mg combine with N 2 to form their respective nitrides, Li 3 N and Mg 3 N 2. � 10. Both lithium and magnesium nitrates on heating evolve NO 2 and O 2 leaving behind their oxides. �
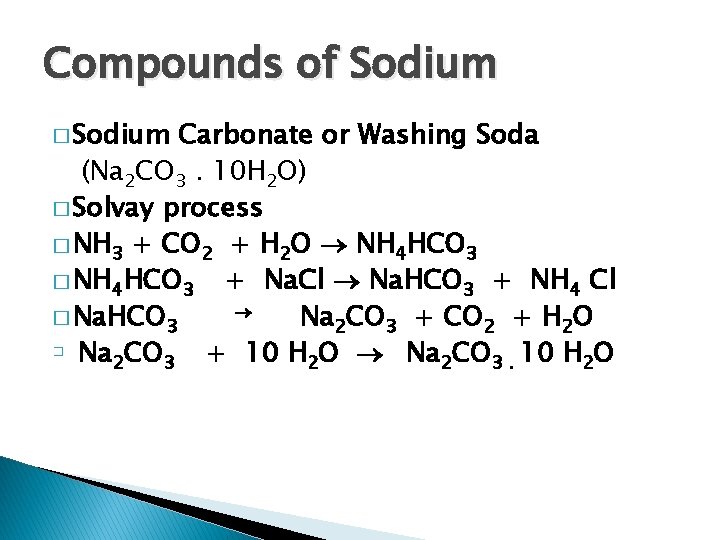
Compounds of Sodium � Sodium Carbonate or Washing Soda (Na 2 CO 3. 10 H 2 O) � Solvay process � NH 3 + CO 2 + H 2 O NH 4 HCO 3 � NH 4 HCO 3 + Na. Cl Na. HCO 3 + NH 4 Cl � Na. HCO 3 Na 2 CO 3 + CO 2 + H 2 O � Na CO + 10 H 2 O Na 2 CO 3. 10 H 2 O 2 3

Uses of sodium carbonate � 1. It is used in water softening, laundering and cleaning. � 2. It is used in paper, paints and textile industries
![� Sodium Chloride, Common Salt or Table Salt [Na. CI] Sea water contains 2. � Sodium Chloride, Common Salt or Table Salt [Na. CI] Sea water contains 2.](http://slidetodoc.com/presentation_image_h2/8db94490c529514d8c747beb48d4ee46/image-9.jpg)
� Sodium Chloride, Common Salt or Table Salt [Na. CI] Sea water contains 2. 7 to 2. 9%by mass of the salt. Sodium chloride is obtained by evaporation of sea water but due to the presence of impurities like Ca. Cl 2 and Mg. Cl 2 it has deliquescent nature. It is purified by passing HCI gas through the impure saturated solution of Na. Cl and due to common ion effect, pure Na. Cl gets precipitated. 28% Na. Cl solution is called brine.
![Sodium Hydroxide or Caustic Soda [Na. OH] � Methods of preparation � Lime –Caustic Sodium Hydroxide or Caustic Soda [Na. OH] � Methods of preparation � Lime –Caustic](http://slidetodoc.com/presentation_image_h2/8db94490c529514d8c747beb48d4ee46/image-10.jpg)
Sodium Hydroxide or Caustic Soda [Na. OH] � Methods of preparation � Lime –Caustic soda process � An aqueous solution of sodium carbonate is treated with hot milk of lime-Ca(OH)2 Na 2 CO 3 + Ca(OH)2 Ca. CO 3 + 2 Na. OH

Electrolytic process of preparation of Na. OH It involves Nelson cell and Castner-Kellner cell. A brine solution is electrolysed using a mercury cathode and a carbon anode. Sodium metal discharged at the cathode combines with Hg to form Na-amalgam. Chlorine gas is evolved at the anode. The amalgam is treated with water to give sodium hydroxide and hydrogen gas. 2 Na-Hg + 2 H 2 O → 2 Na. OH + 2 Hg + H 2

Physical properties of Na. OH � Sodium hydroxide is a white translucent solid. � It is readily soluble in water. � Crystals of Na. OH are deliquescent. � It is a hygroscopic, deliquescent white solid, absorbs CO 2 and moisture from the atmosphere

Sodium Bicarbonate or Baking Soda (Na. HCO 3) � Preparation � It is obtained as an intermediate product in Solvay process � Na 2 CO 3 + CO 2 + H 2 O Na. HCO 3

Uses of sodium bicarbonate � 1. It is used as a constituent of baking powder which is a mixture of sodium bicarbonate, starch � and potassium bitartrate or cream of tartar and in medicine to remove acidity of the stomach (as antacid). � 2. Na. HCO 3 is a mild antiseptic for skin infections. � 3 It is used in fire extinguisher.
- Slides: 14