Saudi Arabia Veterinary Pharmaceutical Legislations Marketing Authorization Application

Saudi Arabia: Veterinary Pharmaceutical Legislations & Marketing Authorization Application (MAA) Presented by: Maher Aljaser, MHA. Section Head, Veterinary Products Licensing Brussels, Belgium 21 -22 June 2016

Agenda • • • Introduction Saudi Food & Drug Authority (SFDA) Veterinary Pharmaceutical Vision and Mission SFDA Roles and Responsibilities Drug Approvals

Introduction sssager@sfda. gov. sa 3

Introduction • Cooperation Council for the Arab States of the Gulf (GCC) • Population (2015) 45 M • Saudi Arabia accounts for 65% of all drug sales in the GCC region • Locally made pharmaceuticals supply only around 15% of the market sssager@sfda. gov. sa 4

Animal Resources Animal type Numbers Sheep 17. 5 million fish farming and aquaculture 99 project (15, 000) m³ Goat 6. 1 million Poultry 50 million bird beehive 38, 878 thousand (2 milloin ton/year) Camel 1. 4 million Cattle 1 million
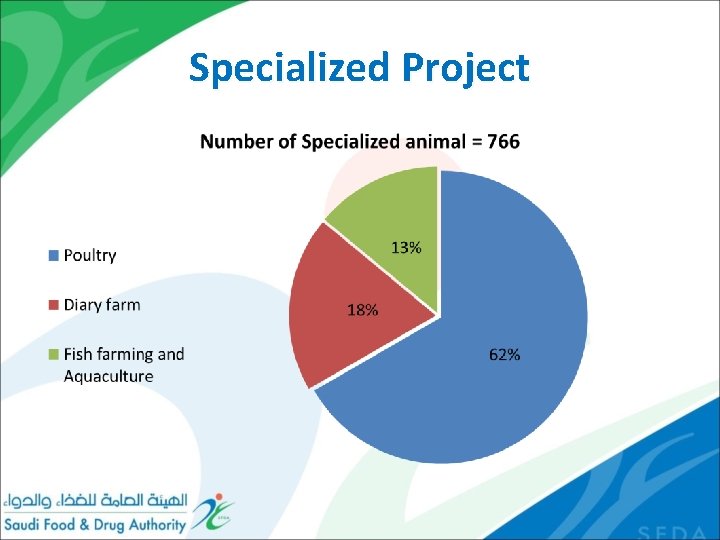
Specialized Project
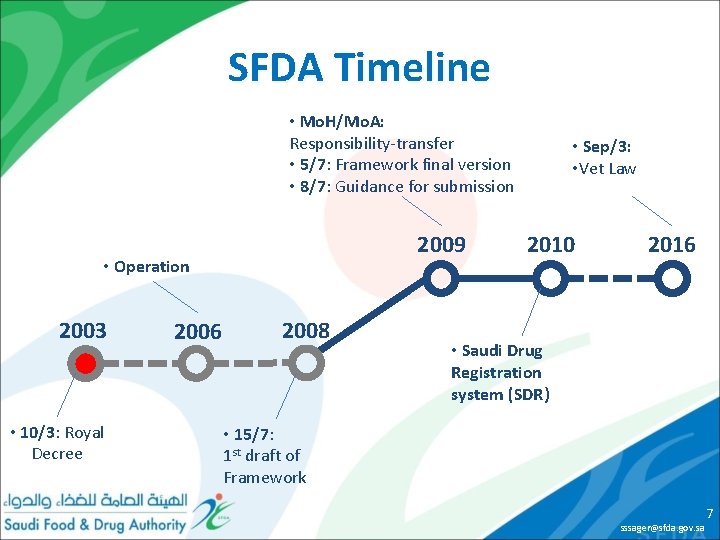
SFDA Timeline • Mo. H/Mo. A: Responsibility-transfer • 5/7: Framework final version • 8/7: Guidance for submission 2009 • Operation 2003 • 10/3: Royal Decree 2006 2008 • Sep/3: • Vet Law 2010 2016 • Saudi Drug Registration system (SDR) • 15/7: 1 st draft of Framework sssager@sfda. gov. sa 7

SFDA Vision To be the leading regional regulatory authority for food, drugs and medical devices with professional and excellent services that contributes to the protection and advancement of the health in Saudi Arabia 8

SFDA Mission To ensure the safety of food; the safety, quality and efficacy of drugs; and the safety and effectiveness of medical devices, by developing and enforcing an appropriate regulatory system 9
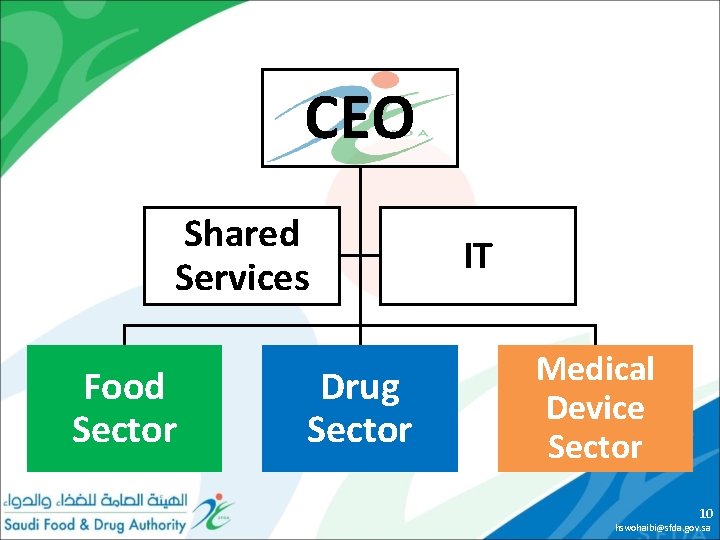
CEO Shared Services Food Sector Drug Sector IT Medical Device Sector 10 hswohaibi@sfda. gov. sa

Veterinary Pharmaceutical • • • Drug sector (Vision and mission) Law & Legislation Organogram Roles & Responsibilities Product Registration Website

Vision To be the leading regional Drug Regulatory Authority for pharmaceuticals and cosmetic products, with professional excellence and services that contribute to the protection and advancement of public health in the Kingdom of Saudi Arabia

Mission Protecting public health by ensuring safety, quality, efficacy and accessibility of human and veterinary drugs and biological products, and safety of cosmetics, through administration of a national regulatory system which is consistent with international best practice.

Legislations Law of the General Food and Drug Authority Law of Pharmaceutical Establishments and preparation Law of Veterinary Pharmaceutical for Gulf Council Countries(GCC) Executive Guidelines for Law of Veterinary Pharmaceutical for Gulf Council Countries(GCC) 14 MAJaser@sfda. gov. sa
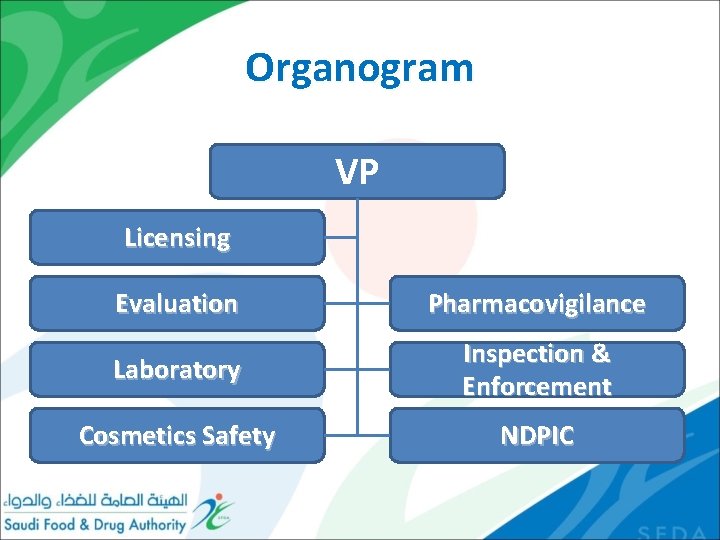
Organogram VP Licensing Evaluation Pharmacovigilance Laboratory Inspection & Enforcement Cosmetics Safety NDPIC
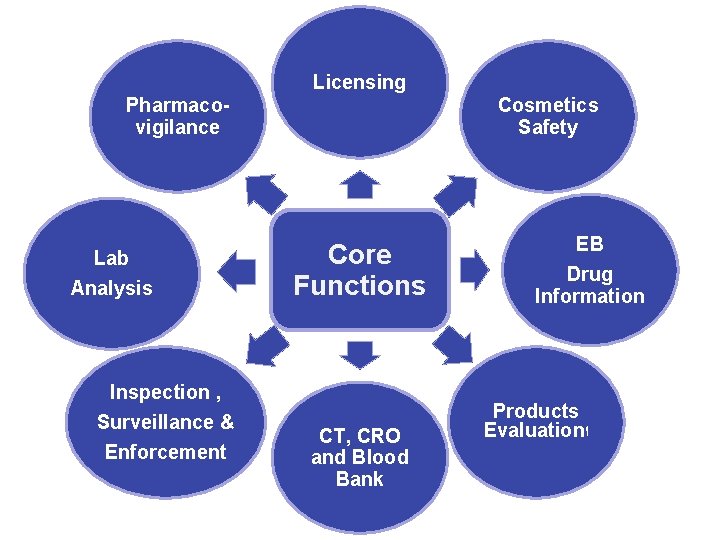
Licensing Pharmacovigilance Lab Analysis Inspection , Surveillance & Enforcement Cosmetics Safety Core Functions CT, CRO and Blood Bank EB Drug Information Products Evaluation ﺍ 16

Product Registration: 1. Guidelines 2. e-Services 17 MAJaser@sfda. gov. sa

18
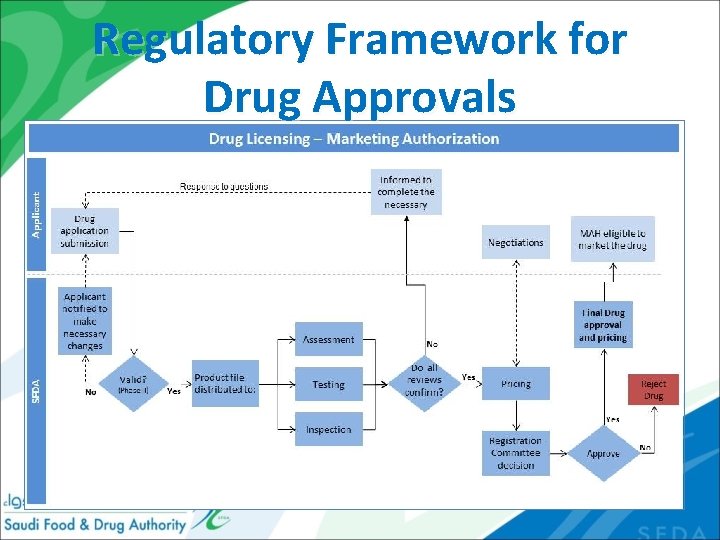
Regulatory Framework for Drug Approvals

Regulatory Framework for Drug Approvals (Time) 20

Regulatory Framework for Drug Approvals (Fees) 21

Veterinary Medicine Reviewed Number of reviewed years 842 2013 707 2014 684 2015 2197 total
http: //www. sfda. gov. sa

Thank You ﺷﻜﺮﺍ majaser@sfda. gov. sa Tel: +966112038222 Ext: 5739 Fax: +966112057643 24 MAJaser@sfda. gov. sa
- Slides: 24